Abstract
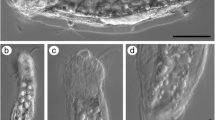
The rapid identification of hydrothermal vent-endemic larvae to the species level is a key limitation to understanding the dynamic processes that control the abundance and distribution of fauna in such a patchy and ephemeral environment. Many larval forms collected near vents, even those in groups such as gastropods that often form a morphologically distinct larval shell, have not been identified to species. We present a staged approach that combines morphological and molecular identification to optimize the capability, efficiency, and economy of identifying vent gastropod larvae from the northern East Pacific Rise (NEPR). With this approach, 15 new larval forms can be identified to species. A total of 33 of the 41 gastropod species inhabiting the NEPR, and 26 of the 27 gastropod species known to occur specifically in the 9° 50′ N region, can be identified to species. Morphological identification efforts are improved by new protoconch descriptions for Gorgoleptis spiralis, Lepetodrilus pustulosus, Nodopelta subnoda, and Echinopelta fistulosa. Even with these new morphological descriptions, the majority of lepetodrilids and peltospirids require molecular identification. Restriction fragment length polymorphism digests are presented as an economical method for identification of five species of Lepetodrilus and six species of peltospirids. The remaining unidentifiable specimens can be assigned to species by comparison to an expanded database of 18S ribosomal DNA. The broad utility of the staged approach was exemplified by the revelation of species-level variation in daily planktonic samples and the identification and characterization of egg capsules belonging to a conid gastropod Gymnobela sp. A. The improved molecular and morphological capabilities nearly double the number of species amenable to field studies of dispersal and population connectivity.







Similar content being viewed by others
References
Adams DK, Mullineaux LS (2008) Supply of gastropod larvae to hydrothermal vents reflects transport from local larval sources. Limnol Oceanogr 53:1945–1955
Comtet T, Jollivet D, Khripounoff A, Segonzac M, Dixon DR (2000) Molecular and morphological identification of settlement-stage vent mussel larvae, Bathymodiolus azoricus (Bivalvia: Mytilidae), preserved in situ at active vent fields on the Mid-Atlantic Ridge. Limnol Oceanogr 45:1655–1661
Dawson MN, Raskoff KA, Jacobs DK (1998) Field preservation of marine invertebrate tissue for DNA analyses. Mol Mar Biol Biotechnol 7:145–152
Douris V, Cameron RAD, Rodakis GC, Lecanidou R (1998) Mitocondrial phylogeography of the land snail Albinaria in Crete: long-term geological and short-term vicariance effects. Evolution 52:116–125
Dreyer J, Knick KE, Flickinger WB, Van Dover CL (2005) Development of macrofaunal community structure in mussel beds on the northern East Pacific Rise. Mar Ecol Prog Ser 302:121–134
Epifanio CE, Perovich G, Dittel AI, Cary SC (1999) Development and behavior of megalopa larvae and juveniles of the hydrothermal vent crab Bythograea thermydron. Mar Ecol Prog Ser 185:147–154
Govenar B, Freeman M, Bergquist DC, Johnson GA, Fisher CR (2004) Composition of a one-year-old Riftia pachyptila community following a clearance experiment: Insight to succession patterns at deep-sea hydrothermal vents. Biol Bull 207:177–182
Gustafson RG, Littlewood DTJ, Lutz RA (1991) Gastropod egg capsules and their contents from deep-sea hydrothermal vent environments. Biol Bull 180:34–55
Harasewych MG, McArthur AG (2000) A molecular phylogeny of the Patellogastropoda (Mollusca: Gastropoda). Mar Biol 137:183–194
Johnson SB, Warén A, Vrijenhoek RC (2008) DNA barcoding of Lepetodrilus limpets reveals cryptic species. J Shellfish Res 27:43–51
Khripounoff A, Comtet T, Vangriesheim A, Crassous P (2000) Near-bottom biological and mineral particle flux in the Lucky Strike hydrothermal vent area (Mid-Atlantic Ridge). J Mar Syst 25:101–118
Lutz R, Jablonski D, Rhoads D, Turner R (1980) Larval dispersal of a deep-sea hydrothermal vent bivalve from the Galápagos Rift. Mar Biol 57:127–133
Lutz RA, Jablonski D, Turner RD (1984) Larval development and dispersal at deep-sea hydrothermal vents. Science 226:1451–1454
Lutz RA, Bouchet P, Jablonski D, Turner RD, Warén A (1986) Larval ecology of mollusks at deep-sea hydrothermal vents. Am Malacol Bull 4:49–54
Maddison DR, Maddison WP (2000) MacClade. Sinauer Associates, Sunderland, Mass
Marsh AG, Mullineaux LS, Young CM, Manahan DT (2001) Larval dispersal potential of the tubeworm Riftia pachyptila at deep-sea hydrothermal vents. Nature 411:77–80
Matabos M, le Bris N, Pendlebury SJD, Thiebaut E (2008a) Role of physico-chemical environment on gastropod assemblages at hydrothermal vents on the East Pacific Rise (13° N/EPR). J Mar Biol Assoc UK 88:995–1008
Matabos M, Thiebaut E, Le Guen D, Sadosky F, Jollivet D, Bonhomme F (2008b) Geographic clines and stepping-stone patterns detected along the East Pacific Rise in the vetigastropod Lepetodrilus elevatus reflect species crypticism. Mar Biol 153:545–563
McLean JH (1988) New archaeogastropod limpets from hydrothermal vents—superfamily Lepetodrilacea. 1. Systematic descriptions. Philos Trans R Soc Lond B Biol Sci 319:1–32
McLean JH (1989a) New archaeogastropod limpets from hydrothermal vents—new family Peltospiridae, new superfamily Peltospiracea. Zool Scr 18:49–66
McLean JH (1989b) New slit-limpets (Scissurellacea and Fissurellacea) from hydrothermal vents. Part 1. Systematic descriptions and comparisons based on shell and radular characters. Contrib Sci 407:1–31
McLean JH (1990) Neolepetopsidae, a new docoglossate limpet family from hydrothermal vents and its relevance to patellogastropod evolution. J Zool 222:485–528
Metaxas A (2004) Spatial and temporal patterns in larval supply at hydrothermal vents in the northeast Pacific Ocean. Limnol Oceanogr 49:1949–1956
Micheli F, Peterson CH, Johnson GA, Mullineaux LS, Mills SW, Sancho G, Fisher CR, Lenihan HS (2002) Predation structures communities at deep-sea hydrothermal vents. Ecol Monogr 72:365–382
Mills SW, Mullineaux LS, Tyler PA (2007) Habitat associations in gastropod species at East Pacific Rise hydrothermal vents (9° 50′N). Biol Bull 212:185–194
Mullineaux LS, Kim SL, Pooley A, Lutz RA (1996) Identification of archaeogastropod larvae from a hydrothermal vent community. Mar Biol 124:551–560
Mullineaux LS, Peterson CH, Micheli F, Mills SW (2003) Successional mechanism varies along a gradient in hydrothermal fluid flux at deep-sea vents. Ecol Monogr 73:523–542
Mullineaux LS, Mills SW, Sweetman AK, Beaudreau AH, Metaxas A, Hunt HL (2005) Vertical, lateral and temporal structure in larval distributions at hydrothermal vents. Mar Ecol Prog Ser 293:1–16
Palumbi SR (1996) Nucleic acids II: the polymerase chain reaction. In: Hillis DM, Mortiz C, Mable BK (eds) Molecular systematics. Sinauer Associates, Sunderland, Mass., pp 205–248
Pardo LM, Ampuero D, Veliz D (2009) Using morphological and molecular tools to identify megalopae larvae collected in the field: the case of sympatric Cancer crabs. J Mar Biol Assoc UK 89:481–490
Pradillon F, Shillito B, Chervin JC, Hamel G, Gaill F (2004) Pressure vessels for in vivo studies of deep-sea fauna. High Press Res 24:237–246
Pradillon F, le Bris N, Shillito B, Young CM, Gaill F (2005) Influence of environmental conditions on early development of the hydrothermal vent polychaete Alvinella pompejana. J Exp Biol 208:1551–1561
Pradillon F, Schmidt A, Peplies J, Dubilier N (2007) Species identification of marine invertebrate early stages by whole-larvae in situ hybridisation of 18S ribosomal RNA. Mar Ecol Prog Ser 333:103–116
Reid DG, Rumbak E, Thomas RH (1996) DNA, morphology and fossils: phylogeny and evolutionary rates of the gastropod genus Littorina. Philos Trans R Soc Lond B Biol Sci 351:877–895
Savolainen V, Cowan RS, Vogler AP, Roderick GK, Lane R (2005) Towards writing the encyclopaedia of life: an introduction to DNA barcoding. Philos Trans R Soc Lond B Biol Sci 360:1805–1811
Sawada H, Saito H, Hosoi M, Toyohara H (2008) Evaluation of PCR methods for fixed bivalve larvae. J Mar Biol Assoc UK 88:1441–1449
Swofford DL (2003) PAUP* phylogenetic analysis using parsimony (*and other methods). version 4.0. Sinauer Associates, Sunderland, Massachusetts
Turner RD, Lutz R, Jablonski D (1985) Modes of molluscan larval development at deep-sea hydrothermal vents. In, Jones ML (ed) Bulletin of the Biological Society of Washington, pp 167–184
Van Dover CL (2003) Variation in community structure within hydrothermal vent mussel beds of the East Pacific Rise. Mar Ecol Prog Ser 253:55–66
Walsh PS, Metzger DA, Higuchi R (1991) Chelex 100 as a medium for simple extraction of DNA for PCR-based typing from forensic material. BioTechniques 10:506–513
Warén A, Bouchet P (1989) New gastropods from East Pacific hydrothermal vents. Zool Scr 18:67–102
Warén A, Bouchet P (1993) New records, species, genera, and a new family of gastropods from hydrothermal vents and hydrocarbon seeps. Zool Scr 22:1–90
Warén A, Bouchet P (2001) Gastropoda and Monoplacophora from hydrothermal vents and seeps; New taxa and records. Veliger 44:116–231
Acknowledgments
We are grateful to the chief scientists, captains and crews of the R/V Atlantis, the shipboard scientific support group, and the Alvin group for their support during multiple collection cruises. We thank Amy Baco-Taylor, Kate Buckman, Abigail Fusaro, and Robert Jennings for training and assistance with molecular techniques. Louis Kerr supported the scanning electron microscopy. Carly Strasser, Stace Beaulieu, Robert Jennings, Jesús Pineda, and Glenn Flierl contributed to discussions at various stages of research and manuscript preparation. We thank two anonymous reviewers for their comments.
Funding was provided by as Woods Hole Oceanographic Institution Deep Ocean Exploration Institute grant to L. M and S. Beaulieu, National Science Foundation grants OCE-0424953, OCE-9712233, and OCE-9619605 to L.M, OCE-0327261 to T.S., and OCE-0002458 to K. Von Damm, and a National Defense Science and Engineering Graduate fellowship to D.A. The experiments comply with the current laws of the United States of America.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by C. Riginos.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Adams, D.K., Mills, S.W., Shank, T.M. et al. Expanding dispersal studies at hydrothermal vents through species identification of cryptic larval forms. Mar Biol 157, 1049–1062 (2010). https://doi.org/10.1007/s00227-009-1386-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-009-1386-8