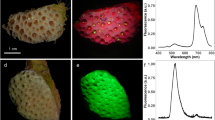
Abstract
Scleractinian symbiotic corals living in the Ligurian Sea (NW Mediterranean Sea) have experienced warm summers during the last decade, with temperatures rapidly increasing, within a few days, to 3–4°C above the mean value of 24°C. The effect of elevated temperatures on the photosynthetic efficiency of zooxanthellae in symbiosis with temperate corals has not been well investigated. In this study, the corals, Cladocora caespitosa and Oculina patagonica were collected in the Ligurian Sea (44°N, 9°E), maintained during 2 weeks at the mean summer temperature of 24°C and then exposed during 48 h to temperatures of 24 (control), 27, 29 and 32°C. Chlorophyll (chl) fluorescence parameters [F v/F m, electron transport rate (ETR), non-photochemical quenching (NPQ)] were measured using pulse amplitude modulated (PAM) fluorimetry before, during the thermal increase, and after 1 and 7 days of recovery (corals maintained at 24°C). Zooxanthellae showed a broad tolerance to temperature increase, since their density remained unchanged and there was no significant reduction in their maximum quantum yield (F v/F m) or ETR up to 29°C. This temperature corresponded to a 5°C increase compared to the mean summer temperature (24°C) in the Ligurian Sea. At 32°C, there was a significant decrease in chl contents for both corals. This decrease was due to a reduction in the chl/zooxanthellae content. For C. caespitosa, there was also a decrease in ETRmax, not associated with a change in F v/F m or in the non-photochemical quenching (NPQ); for O. patagonica, both ETRmax and F v/F m significantly decreased, and NPQmax showed a significant increase. Damages to the photosystem II appeared to be reversible in both corals, since F v/F m values returned to normal after 1 day at 24°C. Zooxanthellae in symbiosis with the Mediterranean corals investigated can therefore be considered as resistant to short-term increases in temperature, even well above the maximum temperatures experienced by these corals in summer.






Similar content being viewed by others
References
Ben-Haim Y, Banim E, Kushmaro A, Loya Y, Rosenberg E (1999) Inhibition of photosynthesis and bleaching of zooxanthellae by the coral pathogen Vibrio shiloi. Environ Microbiol 1:223–229
Bethoux JP, Gentili B, Raunet J, Tailliez D (1990) Warming trend in the western Mediterranean deep water. Nature 347:660–662
Bhagooli R, Hidaka M (2004) Photoinhibition, bleaching susceptibility and mortality in two scleractinian corals, Platygyra ryukuensis and Stylophora pistillata, in response to thermal and light stresses. Comp Biochem Physiol A 137:547–555
Brown BE, Downs CA, Dunne RP, Gibbs SW (2002) Exploring the basis of thermotolerance in the reef coral Goniastrea aspera. Mar Ecol Prog Ser 242:119–129
Buchel C, Wilhelm C (1993) In vivo analysis of slow chlorophyll fluorescence induction kinetics in algae: progress, problems and perspectives. Photochem Photobiol 58:137–148
Cerrano C, Bavestrello G, Bianchi CN, Cattaneo-Vietti R, Bava S, Morganti C, Morri C, Picco P, Sara G, Schiaparelli S, Siccardi A, Sponga F (2000) A catastrophic mass-mortality episode of gorgonians and other organisms in the Ligurian Sea (NW Mediterranean), summer 1999. Ecol Lett 3:284–293
Coma R, Ribes M, Gili JM (2000) Seasonality in coastal benthic ecosystems. Trend Ecol Evol 15:448–453
Fine M, Zibrowius H, Loya Y (2001) Oculina patagonica: a non-lessepsian scleractinian coral invading the Mediterranean Sea. Mar Biol 138:1195–1203
Fitt WK, Brown BE, Warner ME, Dunne RP (2001) Coral bleaching: interpretation of thermal tolerance limits and thermal thresholds in tropical corals. Coral Reefs 20:51–65
Garrabou J, Perez T, Sartoretto S, Harmelin JG (2001) Mass mortality event in Red coral Corallium Rubrum populations in the Provence regions (France, NW Mediterranean). Mar Ecol Prog Ser 217:263–272
Harmelin J-G (2004) Environnement thermique du benthos côtier de l’ile de Port-Cros (parc national, France, Méditerranée nord-occidentale) et implications biogéographiques. Sci Rep Port-Cros natl Park, Fr 20:173–194
Hill R, Schreiber U, Gademann R, Larkum AWD, Kühl M, Ralph PJ (2004) Spatial heterogeneity of photosynthesis and the effect of temperature-induced bleaching conditions in three species of corals. Mar Biol 144:633–640
Hoegh-Guldberg O (1999) Climate change, coral bleaching and the future of the world’s coral reefs. Mar Freshwater Res 50:839–866
Hoegh-Guldberg O, Smith GJ (1989) The effect of sudden changes in temperature, light and salinity on the population density and export of zooxanthellae from the reef coral Stylophora pistillata (Esper) and Seriatopora hystrix (Dana). J Exp Mar Biol Ecol 129:279–303
Houlbrèque F, Tambutté E, Ferrier-Pagès C (2003) Effect of zooplankton availability on the rates of photosynthesis and tissue and skeletal growth of the scleractinian coral Stylophora pistillata. J Exp Mar Biol Ecol 296(2):145–166
Howe SA, Marshall AT (2001) Thermal compensation of metabolism in the temperate coral Plesiastrea versipora (Lamarck 1816). J Exp Mar Biol Ecol 259(2):231–248
Jacques TG, Marshall N, Pilson MEQ (1983) Experimental ecology of the temperate scleractinian coral Astrangia danae. II. Effect of temperature, light intensity and symbiosis with zooxanthellae on metabolic rate and calcification. Mar Biol 76:135–148
Jeffrey SW, Humphrey GF (1975) New spectrophotometric equations for determining chlorophylls a, b, c1 and c2in higher plants, algae and natural phytoplankton. Biochem Physiol Pflanz 167:191–194
Jones RJ, Hoegh-Guldberg O, Larkum AW, Schreiber U (1998) Temperature-induced bleaching of corals begins with impairment of the CO2 fixation mechanism in zooxanthellae. Plant Cell Environ 21:1219–1230
Jones RJ, Ward S, Amri AY, Hoegh-Guldberg O (2000) Changes in quantum efficiency of photosystem II of symbiotic dinoflagellates of corals after heat stress, and of bleached corals sampled after the 1998 Great Barrier Reef mass bleaching event. Mar Freshwater Res 51:63–71
Kružić P, Požar-Domac A (2003) Banks of the coral Cladocora caespitosa (Anthozoa, Scleractinia) in the Adriatic Sea. Coral Reefs 22:536–536
Kushmaro A, Rosenberg E, Fine M, Ben-Haim Y, Loya L (1996) Bacterial infection and coral bleaching. Nature 380:396
Linares C, Coma R, Diaz D, Zabala M, Hereu B, Dantart L (2005) Immediate and delayed effects of a mass mortality event on gorgonian dynamics and benthic community structure in the NW Mediterranean Sea. Mar Ecol Prog Ser 305:127–137
Marsh YA (1970) Primary productivity of reef-building calcareous and red algae. Ecology 55:225–263
Morri C, Peirano A, Bianchi CN, Sassarini M (1994) Present-day bioconstructions of the hard coral, Cladocora caespitosa (L.) (Anthozoa, Scleractinia), in the eastern Ligurian Sea (NW Mediterranean). Biol Mar Medit 1(1):371-372
Muller-Parker G, Davy SK (2001) Temperate and tropical algal-sea anemone symbiosis. Invert Biol 120(2):104–123
Nakamura E, Yokohama Y, Tanaka J (2003) Photosynthetic activity of a temperate coral Acropora pruinosa (Scleractinia, Anthozoa) with symbiotic algae in Japan. Phycol Res 51:38–44
Peirano A, Abbate M, Cerrati G, Difesca V, Peroni C, Rodolfo-Metalpa R (2005) Monthly variations in calyx growth, polyp tissue, and density banding of the Mediterranean scleractinian Cladocora caespitosa (L.). Coral Reefs 24(3):404–409
Peirano A, Morri C, Bianchi CN (1999) Skeleton growth and density pattern of the temperate, zooxanthellate scleractinian Cladocora caespitosa from the Ligurian Sea (NW Mediterranean). Mar Ecol Prog Ser 185:195–201
Peirano A, Morri C, Mastronuzzi G, Bianchi CN (1998) The coral Cladocora caespitosa (Anthozoa, Scleractinia) as a bioherm builder in the Mediterranean Sea. Mem Descr Carta Geol d’It 52(1994):59–74
Perez T, Garrabou J, Sartoretto S, Harmelin J-G, Francour P, Vacelet J (2000) Mortalité massive d’invertébrés marins: un événement sans précédent en Méditerranée nord-occidentale. CR Acad Sci Paris 323:853–865
Picco P (1990) Climatological atlas of the Western Mediterranean. ENEA, Centre S. Teresa, La Spezia
Piniak GA (2002) Effects of symbiotic status, flow speed, and prey type on prey capture by the facultatively symbiotic temperate coral Oculina arbuscula. Mar Biol 141:449–455
Rodolfo-Metalpa R, Bianchi CN, Peirano A, Morri C (2000) Coral Mortality in NW Mediterranean. Coral Reefs 19(1):24–24
Rodolfo-Metalpa R, Bianchi CN, Peirano A, Morri C (2005) Tissue necrosis and mortality of the temperate coral Cladocora caespitosa. Ital J Zool 72:271–276
Rodriguez-Lanetty M, Chang SJ, Song JI (2003) Specificity of two temperate dinoflagelate-anthozoan associations from the north-western Pacific Ocean. Mar Biol 143:1193–1199
Romano JC, Bensoussan N, Younes W, Arlhac D (2000) Anomalies thermiques dans les eaux du golfe de Marseille durant l’été 1999. Une explication partielle de la mortalité d’invertébrés fixés. CR Acad Sci Paris 323:415–427
Rosenberg E, Falkovitz L (2004) The Vibrio shiloi/Oculina patagonica model system of coral bleaching. Annu Rev Microbiol 58:143–159
Rowan R, Knowlton N, Baker A, Jara J (1997) Landscape ecology of algal symbionts creates variation in episodes of coral bleaching. Nature 388:265–269
Savage AM, Goodson MS, Visram S, Trapido-Rosenthal H, Wiedenmann J, Douglas AE (2002) Molecular diversity of symbiotic algae at the latitudinal margins of their distribution: dinoflagellates of the genus Symbiodinium in corals and sea anemones. Mar Ecol Prog Ser 244:17–26
Schiller C (1993) Ecology of the symbiotic coral Cladocora caespitosa (L) (Favidae, Scleractinia) in the Bay of Piran (Adriatic Sea): II. Energy budget. PSZN I: Mar Ecol 14(3):221–238
Schreiber U, Schliwa U, Bilger W (1986) Continuous recordings of photochemical and non photochemical chlorophyll fluorescence quenching with a new type of modulation fluorimetry. Photosynth Res 10:51–62
Schumacher H, Zibrowius H (1985) What is hermatipic? A redefinition of ecological groups in corals and other organisms. Coral Reefs 4:1–9
Shenkar N, Fine M, Loya M (2005) Size matters: bleaching dynamics of the coral Oculina patagonica. Mar Ecol Prog Ser 294:181–188
Smith PK, Khrohn RI, Hermanson GT, Malia AK, Gartner FH, Provenzano MD, Fujimoto EK, Goeke NM, Olson BJ, Klenk DC (1985) Measurement of protein using bicinchoninic acid. Anal Biochem 150:76–85
Stimson J, Sakai K, Sembali H (2002) Interspecific comparison of the symbiotic relationship in corals with high and low rates of bleaching-induced mortality. Coral Reefs 21:409–421
Walther G-R, Post E, Convey P, Menzel A, Parmesan C, Beebee JC, Fromentin J-M, Hoegh-Guldberg O, Bairlein F (2002) Ecological responses to recent climate change. Nature 416:389–395
Warner ME, Fitt WK, Schmidt GW (1996) The effects of elevated temperature on the photosynthetic efficiency of zooxanthellae in hospite from four different species of reef coral: a novel approach. Plant Cell Environ 19:291–299
Zibrowius H (1974) Oculina patagonica, scleractiniaire hermatypique introduit en Méditerranée. Helgol Wiss Meeresunters 26:153–173
Zibrowius H (1980) Les Scléractiniaires de la Méditerranée et de l’Atlantique nord-oriental. Mem Inst Oceanogr Monaco 11:1–284
Acknowledgements
The work presented here is part of the PhD thesis of R. Rodolfo-Metalpa, supported by the Centre Scientifique de Monaco. Thanks are due to Drs. R. Delfanti, and A. Peirano (Marine Environmental Research Centre ENEA, Spezia, Italy) for their support during sample collection, to Prof. G. Fierro (University of Genoa, Italy) and Dr. D. Rubeo (France). Temperature data were either provided by SOMLIT (Service d’Observation en Milieu Littoral, CNRS, INSU, Villefranche-sur-Mer) or by the Oceanographic Museum in Monaco (thanks to T. Thèvenin). Zooxanthellae genotype of the coral C. caespitosa was gently provided by Dr. Didier Forcioli (University of Nice-Sophia-Antipolis). We also thank C. Matthew Moy (Stantford, USA) for English revision and three anonymous reviewers for an improvement of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by R. Cattaneo-Vietti, Genova
Rights and permissions
About this article
Cite this article
Rodolfo-Metalpa, R., Richard, C., Allemand, D. et al. Response of zooxanthellae in symbiosis with the Mediterranean corals Cladocora caespitosa and Oculina patagonica to elevated temperatures. Mar Biol 150, 45–55 (2006). https://doi.org/10.1007/s00227-006-0329-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-006-0329-x