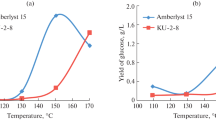
Abstract
Hydrothermal transformation of microcrystalline cellulose (isolated from birch wood) under the influence of Al2O3-B2O3 mixed oxide solid acid catalysts was studied at temperatures of 180 and 215 °C (catalyst and cellulose were mix-milled in an AGO-2 planetary mill). At 180 °C, during the first hour, the average accumulation rates of glucose and 5 hydroxymethylfurfural attain 1030 and 320 mg of product per gram catalyst per hour, respectively. This activity exceeds typical values for sulfonated carbons bearing a similar concentration of acid groups (2.5 times faster glucose accumulation, 4.4 times faster 5-hydroxymethylfurfural accumulation). However, rapid accumulation of unhydrolyzed cellooligosaccharides in the reaction solution causes inhibition of the hydrolysis process after the first hour, probably via oligosaccharide chemisorption on the catalytically active acid sites, which leads to their deactivation. After the process at 180 °C, the catalysts can be washed with water and used again without significant loss of the catalytic performance; after four consecutive runs with the same catalyst, the combined yield of glucose, 5-hydroxymethylfurfural and furfural decreases by 15% compared with the first run. While the catalysts show considerable activity toward acid conversion of glucose into 5-hydroxymethyl furfural, the commonly observed next stage of such conversion into levulinic acid notably does not occur at 180 °C and proceeds at 215 °C.





Similar content being viewed by others
Abbreviations
- COS:
-
Cellooligosaccharide
- 5-HMF:
-
5-Hydroxymethylfurfural
- LA:
-
Levulinic acid
- MCC:
-
Microcrystalline cellulose
- TOF:
-
Turnover frequency
- BA:
-
Boron-aluminum mixed oxide
References
Abasaeed AE, Lee YY, Watson JR (1991) Effect of transient heat transfer and particle size on acid hydrolysis of hardwood cellulose. Biores Technol 35:15–21. https://doi.org/10.1016/0960-8524(91)90077-W
Aymonier C, Gromov NV, Taran OP, Parmon VN (2021) Hydrolysis–dehydration of cellulose to glucose and 5-hydroxymethylfurfural over Sibunit solid acid carbon catalysts under semi-flow conditions. Wood Sci Technol 55:607–624. https://doi.org/10.1007/s00226-021-01271-z
Baek IG, You SJ, Park ED (2012) Direct conversion of cellulose into polyols over Ni/W/SiO2-Al2O3. Biores Technol 114:684–690. https://doi.org/10.1016/j.biortech.2012.03.059
Ermolin VN, Bayandin MA, Kazitsin SN (2016) Mechanical activation of wood for adhesive-free board production. IOP CS Mat Sci Eng 155:012038. https://doi.org/10.1088/1757-899X/155/1/012038
Bautista FM, Campelo JM, Garcia A, Luna D, Marinas JM, Moreno MC, Romero AA (1998) Acidity and catalytic activity of AlPO4–B2O3 and Al2O3–B2O3 (5–30wt% B2O3) systems prepared by impregnation. Appl Cat a: Gen 170:159–168. https://doi.org/10.1016/S0926-860X(98)00046-5
Bustos G, Ramírez JA, Garrote G, Vázquez M (2003) Modeling of the hydrolysis of sugar cane bagasse with hydrochloric acid. Appl Biochem Biotechnol 104:51–68. https://doi.org/10.1385/abab:104:1:51
Chung NH, Dien LQ, Que NT, Thanh NT, Ly GTP (2021) Comparative study on the conversion of Acacia mangium wood sawdust-derived xylose-containing acid hydrolysate to furfural by sulfonated solid catalysts prepared from different lignocellulosic biomass residues. Wood Sci Technol 55:659–679. https://doi.org/10.1007/s00226-021-01284-8
Fukuoka A, Dhepe PL (2006) Catalytic conversion of cellulose into sugar alcohols. Angew Chem Int Ed 45:5161–5163. https://doi.org/10.1002/anie.200601921
Gallezot P (2012) Conversion of biomass to selected chemical products. Chem Soc Rev 41:1538–1558. https://doi.org/10.1039/c1cs15147a
Garyntseva NV, Sudakova IG, Kondrasenko AA, Skripnikov AM, Kuznetsov BN, Taran OP, Agabekov VE (2015) Composition of the products of delignification of birch wood with hydrogen peroxide in the medium “acetic acid - water - catalyst TiO2.” J Sib Fed Univ Chem 8:450–464
Geboers JA, Van de Vyver S, Ooms R, Op de Beeck B, Jacobs PA, Sels BF (2011) Chemocatalytic conversion of cellulose: opportunities, advances and pitfalls. Catal Sci Technol 1:714–726. https://doi.org/10.1039/c1cy00093d
Gromov NV, Medvedeva TB, Sorokina KN, Samoylova YV, Rodikova YA, Parmon VN (2020) Direct conversion of microalgae biomass to formic acid under an air atmosphere with soluble and solid Mo–V–P heteropoly acid catalysts. ACS Sust Chem Eng 8:18947–18956. https://doi.org/10.1021/acssuschemeng.0c06364
Gromov NV, Medvedeva TB, Taran OP, Bukhtiyarov AV, Aymonier C, Prosvirin IP, Parmon VN (2018) Hydrothermal solubilization–hydrolysis–dehydration of cellulose to glucose and 5-hydroxymethylfurfural over solid acid carbon catalysts. Top in Catal 61:1912–1927. https://doi.org/10.1007/s11244-018-1049-4
Hick SM, Griebel C, Restrepo DT, Truitt JH, Buker EJ, Bylda C, Blair RG (2010) Mechanocatalysis for biomass-derived chemicals and fuels. Green Chem 12:468–474. https://doi.org/10.1039/b923079c
James SL, Adams CJ, Bolm C, Braga D, Collier P, Friščić T, Grepioni F, Harris KDM, Hyett G, Jones W, Krebs A, Mack J, Maini L, Orpen AG, Parkin IP, Shearouse WC, Steed JW, Waddell DC (2012) Mechanochemistry: opportunities for new and cleaner synthesis. Chem Soc Rev 41:413–447. https://doi.org/10.1039/c1cs15171a
Kim JS, Lee YY, Torget RW (2001) Cellulose hydrolysis under extremely low sulfuric acid and high-temperature conditions. Appl Biochem Biotechnol 91:331–340. https://doi.org/10.1385/abab:91-93:1-9:331
Kitano M, Yamaguchi D, Suganuma S, Nakajima K, Kato H, Hayashi S, Hara M (2009) Adsorption-enhanced hydrolysis of β-1,4-glucan on graphene-based amorphous carbon bearing SO3H, COOH, and OH groups. Langmuir 25:5068–5075. https://doi.org/10.1021/la8040506
Klemm D, Heublein B, Fink H, Bohn A (2005) Cellulose: fascinating biopolymer and sustainable raw material. Angew Chem 44(22):3358–3393
Kobayashi H, Yabushita M, Hasegawa J-y, Fukuoka A (2015) Synergy of vicinal oxygenated groups of catalysts for hydrolysis of cellulosic molecules. J Phys Chem C 119:20993–20999. https://doi.org/10.1021/acs.jpcc.5b06476
Kuznetsov BN, Sharypov VI, Kuznetsova SA, Baryshnikov SV, Danilov VG, Yatsenkova OV, Ivanchenko NM (2012) Effect of different activation methods on the composition, structure and reactivity of aspen wood. Chem Sust Dev 20:507–513
Kuznetsov BN, Kuznetsova SA, Levdansky VA, Levdansky AV, Vasil’eva NYu, Chesnokov NV, Ivanchenko NM, Djakovitch L, Pinel C, (2015) Optimized methods for obtaining cellulose and cellulose sulfates from birch wood. Wood Sci Technol 49:825–843. https://doi.org/10.1007/s00226-015-0723-y
Lamberov AA, Sitnikova EY, Gilmanov RR, Sidorov NA (2010) Effect of boron oxide addition on the structure and activity of an alumina catalyst for skeletal isomerization ofin-butylenes. Catal Ind 3:55
Lanzafame P, Temi DM, Perathoner S, Spadaro AN, Centi G (2012) Direct conversion of cellulose to glucose and valuable intermediates in mild reaction conditions over solid acid catalysts. Cat Today 179:178–184. https://doi.org/10.1016/j.cattod.2011.07.018
Lavrenov AV, Buluchevsky EA, Karpova TR, Moiseenko MA, Mikhailova MS, Chumachenko YA, Skorplyuk AA, Gulyaeva TI, Arbuzov AB, Leontyeva NN, Drozdov VA (2011) Synthesis, structure and properties of borate-containing oxide catalysts for petrochemical processes and synthesis of motor fuel components. Chem Sust Develop 19:87–95
Lenihan P, Orozco A, O’Neill E, Ahmad MNM, Rooney DW, Walker GM (2010) Dilute acid hydrolysis of lignocellulosic biomass. Chem Eng J 156:395–403. https://doi.org/10.1016/j.cej.2009.10.061
Liu J, Li H, Liu Y-C, Lu Y-M, He J, Liu X-F, Wu Z-B, Yang S (2015) Catalytic conversion of glucose to 5-hydroxymethylfurfural over nano-sized mesoporous Al2O3–B2O3 solid acids. Cat Comm 62:19–23. https://doi.org/10.1016/j.catcom.2015.01.008
Marianou AA, Michailof CC, Ipsakis D, Triantafyllidis K, Lappas AA (2019) Cellulose conversion into lactic acid over supported HPA catalysts. Green Chem 21:6161–6178. https://doi.org/10.1039/c9gc02622c
Neimark AV, Lin Y, Ravikovitch PI, Thommes M (2009) Quenched solid density functional theory and pore size analysis of micro-mesoporous carbons. Carbon 47(7):1617–1628
Onda A, Ochi T, Yanagisawa K (2008) Selective hydrolysis of cellulose into glucose over solid acid catalysts. Green Chem 10:1033–1037. https://doi.org/10.1039/b808471h
Palai YN, Shrotri A, Asakawa M, Fukuoka A (2021) Silica supported Sn catalysts with tetrahedral Sn sites for selective isomerization of glucose to fructose. Cat Today 365:241–248. https://doi.org/10.1016/j.cattod.2020.04.032
Pham ST, Nguyen BM, Le GH, Sapi A, Mutyala S, Szenti I, Konya Z, Vu TA (2020) Role of Brønsted and Lewis acidic sites in sulfonated Zr-MCM-41 for the catalytic reaction of cellulose into 5-hydroxymethyl furfural. React Kin Mech Cat 130:825–836. https://doi.org/10.1007/s11144-020-01799-4
Prasetyoko D, Ramli Z, Endud S, Nur H (2008) Characterization and catalytic performance of niobic acid dispersed over titanium silicalite. Adv Mat Sci Eng 2008:345895. https://doi.org/10.1155/2008/345895
Qi T, He M-F, Zhu L-F, Lyu Y-J, Yang H-Q, Hu C-W (2019) Cooperative catalytic performance of Lewis and Brønsted acids from AlCl3 salt in aqueous solution toward glucose-to-fructose isomerization. J Phys Chem C 123:4879–4891. https://doi.org/10.1021/acs.jpcc.8b11773
Ruiz-Matute AI, Hernández-Hernández O, Rodríguez-Sánchez S, Sanz ML, Martínez-Castro I (2011) Derivatization of carbohydrates for GC and GC–MS analyses. J Chromat B 879:1226–1240. https://doi.org/10.1016/j.jchromb.2010.11.013
Sharypov VI, Beregovtsova NG, Baryshnikov SV, Zhizhaev AM, Vosmerikov AV, Kuznetsov BN (2015) Study of thermal transformation of mechanochemically activated aspen wood in the presence of a high-silica zeolite catalyst. J Sib Fed Univ Chem 8:190–201
Shi N, Liu Q, Liu Y, Chen L, Zhang H, Ma L (2020) Identification of the soluble byproducts formed during the hydrothermal conversion of cellulose catalyzed by solid tungstated alumina. ACS Omega 5:19140–19150. https://doi.org/10.1021/acsomega.0c02491
Sluiter JB, Ruiz RO, Scarlata CJ, Sluiter AD, Templeton DW (2010) Compositional analysis of lignocellulosic feedstocks. 1. Review and description of methods. J Agr Food Chem 58:9043–9053. https://doi.org/10.1021/jf1008023
Suganuma S, Nakajima K, Kitano M, Yamaguchi D, Kato H, Hayashi S, Hara M (2008) Hydrolysis of cellulose by amorphous carbon bearing SO3H, COOH, and OH groups. J Amer Chemi Soc 130:12787–12793. https://doi.org/10.1021/ja803983h
Tarabanko N, Tarabanko VE, Kukhtetskiy SV, Taran OP (2019) Electrical double layer as a model of interaction between cellulose and solid acid catalysts of hydrolysis. Chem Phys Chem 20:706–718. https://doi.org/10.1002/cphc.201801160
Tarabanko N, Tarabanko VE, Taran OP (2020) Unidimensional approximation of the diffuse electrical layer in the inner volume of solid electrolyte grains in the absence of background ions. Chem Phys Chem 21:1925–1933. https://doi.org/10.1002/cphc.202000455
Tarabanko VE, Chernyak MY, Aralova SV, Kuznetsov BN (2002) Kinetics of levulinic acid formation from carbohydrates at moderate temperatures. Reaction Kinetics Cat Lett 75:117–126. https://doi.org/10.1023/a:1014857703817
Tempelman C, Jacobs U, Hut T, Pereira de Pina E, van Munster M, Cherkasov N, Degirmenci V (2019) Sn exchanged acidic ion exchange resin for the stable and continuous production of 5-HMF from glucose at low temperature. Appl Cat a Gen 588:117267. https://doi.org/10.1016/j.apcata.2019.117267
Tian J, Fang C, Cheng M, Wang X (2011) Hydrolysis of cellulose over CsxH3–xPW12O40 (X = 1–3) Heteropoly acid catalysts. Chem Eng Technol 34:482–486. https://doi.org/10.1002/ceat.201000409
Tsai MC, Chen YW (1990) Hydrothermal stability of aluminum borate. Cat Lett 6:225–230. https://doi.org/10.1007/bf00774724
Xi J, Zhang Y, Xia Q, Liu X, Ren J, Lu G, Wang Y (2013) Direct conversion of cellulose into sorbitol with high yield by a novel mesoporous niobium phosphate supported Ruthenium bifunctional catalyst. Appl Cat a Gen 459:52–58. https://doi.org/10.1016/j.apcata.2013.03.047
Yabushita M, Kobayashi H, Hara K, Fukuoka A (2014a) Quantitative evaluation of ball-milling effects on the hydrolysis of cellulose catalysed by activated carbon. Cat Sci Technol 4:2312–2317. https://doi.org/10.1039/c4cy00175c
Yabushita M, Kobayashi H, Hasegawa J-y, Hara K, Fukuoka A (2014b) Entropically favored adsorption of cellulosic molecules onto carbon materials through hydrophobic functionalities. Chem Sus Chem 7:1443–1450. https://doi.org/10.1002/cssc.201301296
Yang B, Dai Z, Ding S-Y, Wyman CE (2011) Enzymatic hydrolysis of cellulosic biomass. Biofuels 2:421–449. https://doi.org/10.4155/bfs.11.116
Yasuda H, Kameoka T, Sato T, Kijima N, Yoshimura Y (1999) Sulfur-tolerant Pd–Pt/Al2O3–B2O3 catalyst for aromatic hydrogenation. Appl Cat a Gen 185:L199–L201. https://doi.org/10.1016/S0926-860X(99)00193-3
Yurpalov VL, Fedorova ED, Drozdov VA, Lavrenov AV (2016) Evaluation of the acidic properties of the B2O3–Al2O3 and Pt/B2O3–Al2O3 systems by spin probe EPR spectroscopy and their correlation with the occurrence of the joint hydroisomerization of heptane and benzene. Kinet Cat 57:540–545. https://doi.org/10.1134/s0023158416040169
Zeng M, Pan X (2020) Insights into solid acid catalysts for efficient cellulose hydrolysis to glucose: progress, challenges, and future opportunities. Catal Rev. https://doi.org/10.1080/01614940.2020.1819936
Acknowledgements
This research was accomplished with financing from the Russian Science Foundation (Project No. 21-13-00250), and using the equipment of Krasnoyarsk Regional Research Equipment Centre of SB RAS.
Funding
This research was funded by the Russian Science Foundation (Project No. 21–13-00250).
Author information
Authors and Affiliations
Contributions
All the authors contributed equally to this work.
Corresponding authors
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Ethics approval
The authors did not conduct research on animals or humans in any form.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tarabanko, N., Baryshnikov, S.V., Kazachenko, A.S. et al. Hydrothermal hydrolysis of microcrystalline cellulose from birch wood catalyzed by Al2O3-B2O3 mixed oxides. Wood Sci Technol 56, 437–457 (2022). https://doi.org/10.1007/s00226-022-01363-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00226-022-01363-4