Abstract
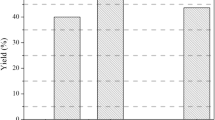
To increase hydroxyl content in lignin, demethylation of enzymatic hydrolysis lignin (DML) was conducted using 1-dodecanethiol as nucleophilic reagent and sodium methoxide as catalyst in this work. Under alkaline conditions, the demethylated lignin–epoxy resin (DML-EP) was synthesized by demethylated lignin reacted with epichlorohydrin (ECH). The optimum conditions for the synthesis of DML-EP were: 80 °C, 2.5 h, molar ratio of ECH and NaOH to phenolic hydroxyl of 7.5 and 1.2, respectively. FTIR spectrum showed the epoxy group appeared in the product. Then, the lignin-based epoxy composites were produced by cast molding method. Tensile strength test reflects that DML-EP-reinforced epoxy composites have an obvious improvement in tensile strength and elongation of about 27 and 34% compared to the pure epoxy resin with 5% content. Scanning electron microscopy images showed the fractured surface of DML-EP/epoxy composites was rough. Rheological analysis showed that the cured DML-EP/E-51 epoxy resin composites had better elasticity and bonding effect. TGA experiment showed the thermal stability of DML-EP/epoxy composites was also improved. Non-isothermal method was used to analyze the curing kinetics of E-51 resin and DML-EP/E-51 composites, and the curing apparent activation energy was calculated using the Kissinger and Ozawa methods.








Similar content being viewed by others
References
Asada C, Basnet S, Otsuka M, Sasaki C, Nakamura Y (2015) Epoxy resin synthesis using low molecular weight lignin separated from various lignocellulosic materials. Int J Biol Macromol 74:413–419
Auvergne R, Caillol S, David G, Boutevin B, Pascault JP (2014) Biobased thermosetting epoxy: present and future. Chem Rev 114(2):1082–1115
Chung H, Washburn NR (2012) Chemistry of lignin-based materials. Green Mater 1(3):137–160
Delmas GH, Benjelloun-Mlayah B, Bigot YL, Delmas M (2013) Biolignin (TM) based epoxy resins. J Appl Polym Sci 127(3):1863–1872
Ding N, Wang X, Tian Y, Yang L, Chen H, Wang Z (2015) A renewable agricultural waste material for the synthesis of the novel thermal stability epoxy resins. Polym Eng Sci 54(12):2777–2784
Ferdosian F, Yuan Z, Anderson M, Xu C (2014) Synthesis of lignin-based epoxy resins: optimization of reaction parameters using response surface methodology. RSC Adv 4(60):31745–31753
Hofmann K, Glasser WG (1993a) Engineering plastics from lignin. 21. Synthesis and properties of epoxidized lignin-poly(propylene oxide) copolymers. J Wood Chem Technol 13(1):73–95
Hofmann K, Glasser WG (1993b) Engineering plastics from lignin. 22. Cure of lignin based epoxy resins. J Adhes 40(2–4):229–241
Hu L, Pan H, Zhou Y, Hse C, Liu C, Zhang B, Xu B (2014) Chemical groups and structural characterization of lignin via thiol-mediated demethylation. J Wood Chem Technol 34(2):122–134
Jin FL, Li X, Park SJ (2015) Synthesis and application of epoxy resins: a review. J Ind Eng Chem 29:1–11
Kaelble DH, Smith T (1974) Analysis of curing kinetics in polymer composites. J Polym Sci Part A Polym Chem 11(8):533–540
Laurichesse S, Avérous L (2014) Chemical modification of lignins: towards biobased polymers. Prog Polym Sci 39(7):1266–1290
Li J, Wang W, Zhang S, Gao Q, Zhang W, Li J (2016) Preparation and characterization of lignin demethylated at atmospheric pressure and its application in fast curing biobased phenolic resins. RSC Adv 6(71):67435–67443
Llano T, Alexandri M, Koutinas A, Gardeli C, Papapostolou H, Coz A, Quijorna N, Andres A, Komaitis M (2015) Liquid–liquid extraction of phenolic compounds from spent sulphite liquor. Waste Biomass Valorization 6(6):1149–1159
Ma S, Liu X, Fan L, Jiang Y, Cao L, Tang Z, Zhu J (2014) Synthesis and properties of a bio-based epoxy resin with high epoxy value and low viscosity. Chemsuschem 7(2):555–562
Mansouri NEE, Yuan Q, Mail FH (2011) Synthesis and characterization of kraft based epoxy resins. BioResources 6(3):2492–2503
Ozawa T (1965) A new method of analyzing thermogravimetric data. Bullchemsocjpn 38(11):1881–1886
Podschun J, Saake B, Lehnen R (2015) Reactivity enhancement of organosolv lignin by phenolation for improved bio-based thermosets. Eur Polymer J 67:1–11
Rahimi A, Ulbrich A, Coon JJ, Stahl SS (2014) Formic-acid-induced depolymerization of oxidized lignin to aromatics. Nature 515(7526):249–252
Sasaki C, Wanaka M, Takagi H, Tamura S, Asada C, Nakamura Y (2013) Evaluation of epoxy resins synthesized from steam-exploded bamboo lignin. Ind Crops Prod 43(1):757–761
Song Y, Wang Z, Yan N, Zhang R, Li J (2016) Demethylation of wheat straw alkali lignin for application in phenol formaldehyde adhesives. Polymers 8(6):209
Sun QN, Qin TF, Li GY (2009) Chemical groups and structural characterization of brown-rotted lignin. Int J Polym Anal Charact 14(1):19–33
Upton BM, Kasko AM (2015) Strategies for the conversion of lignin to high-value polymeric materials: review and perspective. Chem Rev 116(4):2275–2306
Wang F, Li SQ, Wang JW, Xiao J (2012) Effect of functionalized multi-walled carbon nanotubes on the curing behavior and thermal stability of epoxy resin. High Perform Polym 24(2):97–104
Wei Q, Li S, Guo G, Han S, Ren S, Ma Y (2015) Synthesis and characterization of phenol-formaldehyde resin using enzymatic hydrolysis lignin. J Ind Eng Chem 21(1):1417–1422
Xin J, Li M, Li R, Wolcott MP, Zhang J (2016) Green epoxy resin system based on lignin and tung oil and its application in epoxy asphalt. ACS Sustain Chem Eng 4(5):2754–2761
Xu C, Arancon RA, Labidi J, Luque R (2014) Lignin depolymerisation strategies: towards valuable chemicals and fuels. Chem Soc Rev 43(22):7485–7500
Yelle DJ, Kaparaju P, Hunt CG, Hirth K, Kim H, Ralph J, Felby C (2013) Two-dimensional NMR evidence for cleavage of lignin and xylan substituents in wheat straw through hydrothermal pretreatment and enzymatic hydrolysis. Bioenergy Res 6(1):211–221
Funding
This study was supported by the National Natural Science Foundation of China (No. 31200451). And we also thank Advanced Analysis and Testing Center, Nanjing Forestry University, for SEM characterization.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Wang, F., Kuai, J., Pan, H. et al. Study on the demethylation of enzymatic hydrolysis lignin and the properties of lignin–epoxy resin blends. Wood Sci Technol 52, 1343–1357 (2018). https://doi.org/10.1007/s00226-018-1024-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00226-018-1024-z