Abstract
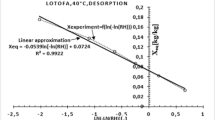
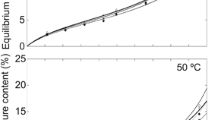
Water vapor sorption surface areas and sorption energies of untreated and thermally modified Norway spruce [Picea abies (L.) Karst.], sycamore maple (Acer pseudoplatanus L.) and European ash (Fraxinus excelcior L.) were investigated by means of dynamic vapor sorption (DVS) measurements and excess surface work (ESW) evaluation method, respectively. Adsorption and desorption experiments in the hygroscopic range and desorption tests from water saturation were conducted. Thermodynamically, ESW is the sum of the surface free energy and the isothermal isobaric work of sorption. From the amount adsorbed in the first minimum a specific surface area similar to the BET surface area can be obtained. The results show that untreated spruce has a significantly higher specific water vapor sorption surface and sorption energy compared to both hardwoods maple and ash. Thermal modification of the woods leads to a significant reduction of water vapor sorption surface and sorption energy. The determined surface area and energy are higher in desorption direction than in adsorption direction, whereby the highest values in desorption direction from water saturation, especially for maple and ash, were obtained. The surface areas calculated by means of the ESW method are similar to the surface areas calculated by means of the BET method, particularly in adsorption direction.


Similar content being viewed by others
References
Adolphs J (2007) Excess surface work-A modelless way of getting surface energies and specific surface areas directly from sorption isotherms. Appl Sur Sci 253:5645–5649
Adolphs J, Setzer MJ (1996a) A model to describe adsorption isotherms. J Colloid Interface Sci 180:70–76
Adolphs J, Setzer MJ (1996b) Energetic Classification of Adsorption Isotherms. J Colloid Interface Sci 184:443–448
Adolphs J, Setzer MJ (1998) Description of gas adsorption isotherms on porous and dispersed systems with the excess surface work model. J Colloid Interface Sci 207:349–354
Bächle H, Zimmer B, Windeisen E, Wegener G (2010) Evaluation of thermally modified beech and spruce wood and their properties by FT-NIR spectroscopy. Wood Sci Technol 44:421–433
Borrega M, Niemelä K, Sixta H (2013) Effect of hydrothermal treatment intensity on the formation of degradation products from birchwood. Holzforschung 67:871–879
Bourgois J, Guyonnet R (1988) Characterization and analysis of torrefied wood. Wood Sci Technol 22:143–155
Brunauer S, Emmet PH, Teller E (1938) Adsorption of gases in multimolecular layers. J Am Chem Soc 60:309–319
Churaev NV, Setzer MJ, Adolphs J (1998) A model to describe adsorption isotherms. J Colloid Interface Sci 197:327–333
De Boer JH, Zwicker CZ (1929) Adsorption als folge von polarisation. Die adsorptionsisotherme (adsorption as a result of polarization. The adsorption isotherms). Z Physik Chem B3:407–418 (In German)
Derjaguin BV, Churaev NV, Muller VM (1987) Surface forces. Consultants Bureau, New York
Engelund ET, Thygesen LG, Svensson S, Hill CAS (2013) A critical discussion of the physics of wood-water interactions. Wood Sci Technol 47:141–161
Engelund ET, Thygesen LG, Burgert I (2017) Hydroxyl accessibility in wood cell walls as affected by drying and re-wetting procedures. Cellulose 24:2375–2384
Enke D, Rückriem M, Schreiber A, Adolphs J (2010) Water vapor sorption on hydrophilic and hydrophobic nanoporous materials. Appl Sur Sci 256:5482–5485
Fengel D, Wegener G (2003) Wood: chemistry, ultrastructure. Verlag Kessel, München, Reaction
Fernandes Diniz JMB, Gil MH, Castro JAAM (2004) Hornification-its origin and interpretation in wood pulps. Wood Sci Technol 37:489–494
Herrera R, Erdocia X, Labidi J, Llano-Ponte R (2015a) Chemical analysis of industrial-scale hydrothermal wood degraded by wood-rotting basidiomycetes and its action mechanisms. Polym Degrad Stab 117:37–45
Herrera R, Muszynska M, Krystofiak T, Labidi J (2015b) Comparative evaluation of different thermally modified wood samples finishing with UV-curable and water borne coatings. Appl Sur Sci 357:1444–1453
Hill CAS (2006) Wood modification: chemical, thermal and other processes. Wiley, Chichester
Hill CAS, Norton AJ, Newmann G (2010) The water vapour sorption properties of Sitka spruce determined using a dynamic vapour sorption apparatus. Wood Sci Technol 44:497–514
Hill CAS, Ramsay J, Keating B, Laine K, Rautkari L, Hughes M, Constant B (2012) The water vapour sorption properties of thermally modified and densified wood. J Mater Sci 47:3191–3197
Himmel S, Mai C (2016) Water vapour sorption of wood modified by acetylation and formalization-analysed ba a sorption kinetics model and thermodynamic considerations. Holzforschung 70:203–213
Hosseinpourpia R, Adamopoulos S, Holstein N, Mai C (2017) Dynamic vapour sorption and water-related properties of thermally modified Scots pine (Pinus sylvestris L.) wood pre-treated with proton acid. Polym Degrad Stab 138:161–168
ISO 9277 (2010) Determination of the specific area of solids by gas adsorption—BET method. Beuth Verlag, Berlin
Olek W, Bonarski JT (2014) Effects of thermal modification on wood ultrastructure analyzed with crystallographic texture. Holzforschung 68:721–726
Olek W, Majka J, Czaikowski Ł (2013) Sorption isotherms of thermally modified wood. Holzforschung 67:183–191
Peschel G, Adlfinger KH (1971) Thermodynamic investigations of thin layers between solid surfaces. Zeit Naturforsch 26A:705–715
Pètrissans A, Younsi R, Chaoch M, Gèrardin P, Pètrissans M (2012) Experimental and numerical analysis of wood thermodegradation: mass loss kinetics. J Therm Anal Calorim 109:907–914
Pfriem A, Zauer M, Wagenführ A (2010) Alteration of the unsteady sorption behaviour of maple (Acer pseudoplatanus L.) and spruce (Picea abies (L.) Karst.) due to thermal modification. Holzforschung 64:235–241
Popescu CM, Hill CAS (2013) The water vapour adsorption-desorption behaviour of naturally aged Tilia codorata Mill. wood. Polym Degrad Stab 98:1804–1813
Repellin V, Guyonnet R (2005) Evaluation of heat-treated wood swelling by differential scanning calorimetry in relation to chemical composition. Holzforschung 59:28–34
Roffael E, Kraft R (2012) Influence of thermal wood modification on the water retention value (WRV). Eur J Wood Prod 70:393–395
Rübner K, Prinz C, Adolphs J, Hempel S, Schell A (2015) Microstructural characterisation of lightweight granules made from masonry rubble. Micropor Mesopor Mater 209:113–121
Salmén L, Bergström E (2009) Cellulose structural arrangement in relation to spectral changes in tensile loading FTIR. Cellulose 16:975–982
Sandermann W, Augustin H (1963) Chemical investigations on the thermal decomposition of wood. Part I: stand of research. Holz Roh Werkst 21:256–265
Skaar C (1988) Wood-water relations. Springer, Berlin
Weiland JJ, Guyonnet R (2003) Study of chemical modification and fungi degradation of thermally modified wood using DRFIT spectroscopy. Holz Roh Werkst 61:216–220
Windeisen E, Strobel C, Wegener G (2007) Chemical changes during the production of thermo- treated beech wood. Wood Sci Technol 41:523–536
Zaman A, Alèn R, Kotilainen R (2000) Thermal behavior of Scots pine (Pinus sylvestris) and silver birch (Betula pendula) at 200–230°C. Wood Fiber Sci 32:138–143
Zauer M, Kowalewski A, Oberer I, Sproßmann R, Wagenführ A (2015) Development of thermally modified wood to substitute tropical hardwood for the use in acoustic guitars. Proceedings of the Eighth European Conference on Wood Modification 8:96–99
Zauer M, Kowalewski A, Sproßmann R, Stonjek H, Wagenführ A (2016) Thermal modification of European beech at relatively mild temperatures for the use in electric bass guitars. Eur J Wood Prod 74:43–48
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zauer, M., Prinz, C., Adolphs, J. et al. Sorption surfaces and energies of untreated and thermally modified wood evaluated by means of excess surface work (ESW). Wood Sci Technol 52, 957–969 (2018). https://doi.org/10.1007/s00226-018-1021-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00226-018-1021-2