Abstract
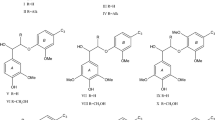
Natural lignins are one of the most widespread biopolymers. The main active centers in the lignin macromolecules are phenolic hydroxyl groups. They form lignin macromolecules reactivity. The lignin is considered as a natural polyphenol. The main characteristics of the lignin reactivity are their acid–base and redox properties. Acid–base properties of lignins are characterized by pKa values of basic types of phenolic structures. The redox properties are characterized by effective oxidation potential and the Gibbs energy of the phenolic structures. This paper studies the acid–base and redox properties of the phenolic structures of coniferous and deciduous natural lignins. The authors analyze the representative slightly altered preparations of dioxane lignin extracted from coniferous wood (spruce, common juniper) and deciduous wood (common aspen). The study reveals the differences in the reaction properties of coniferous and deciduous lignins due to their functional nature and macromolecular properties.



Similar content being viewed by others
References
Adams RP, Pandey RN, Leverenz JW, Dignard N, Hoegh K, Thorfinnsson T (2003) Pan-arctic variation in Juniperus communis: historical biogeography based on DNA fingerprinting. Biochem Syst Ecol 31(2):181–192
Afanas’ev NI, Fesenko AV, Vishnyakova AP, Chainikov AN (2008) Macromolecular properties and topological structure of spruce dioxane lignin. Polym Sci A 50(2):190–197
Bogolitsyn KG, Lunin VV et al (2010) Physical chemistry of lignin. Academkniga, Moscow
Bogolitsyn KG, Gusakova MA, Khviyuzov SS, Zubov IN (2014) Physicochemical properties of conifer lignins using Juniperus communis as an example. Chem Nat Compd 50(2):337–341
Brauns PE (1952) The chemistry of lignin. Academic Press, New York
Djikanović D, Simonović J, Savić A, Ristić I et al (2012) Structural differences between lignin model polymers synthesized from various monomers. J Polym Environ 20:607–617
Hatakeyama H, Hatakeyama T (2010) Lignin structure, properties, and applications. Adv Polym Sci 232:1–63
Heitner C, Dimmel DR, Schmidt JA (2010) Lignin and lignans: advances in chemistry. Taylor & Francis Group, Boca Raton
Karmanov AP (2004) Self-organization and structural organization of lignin. UB RAS, Ekaterinburg
Kosyakov DS, Khviyuzov SS, Gorbova NS, Bogolitsyn KG (2013a) Protolytic properties of lignin in binary mixtures of water with aprotic solvents. Russ J Appl Chem 86(7):1131–1136
Kosyakov DS, Panfilova MV, Gorbova NS, Bogolitsyn KG (2013b) Acidity of phenols of syringyl series in binary mixtures of water with 1,4-dioxane and acetonitrile. Chem Chem Technol 56(6):32–37
Lehto J, Pakkanen H, Alen R (2015) Characterization of lignin dissolved during alkaline pretreatment of softwood and hardwood. J Wood Chem Technol 35:337–347
Levin ED, Rubchevskaya LP (1980) About the representativeness of samples in the study of the chemical composition of wood. Wood Chem 4:103–106
Lin SY, Dence CW (1992) Methods in lignin chemistry. Springer, Berlin
Maksimov IV, Cherepanova EA, Burkhanova GF, Sorokan’ AV, Kuzmina OI (2011) Structural–functional features of plant isoperoxidases. Biochemistry 76(6):609–621
Obolenskaya AV, El’nitskaya ZP, Leonovich AL (1991) Laboratory studies of wood and cellulose chemistry. Ecology, Moscow
Ragnar M, Lindgren CT, Nilvebrant NO (2000) pKa-values of guaiacyl and syringyl phenols related to lignin. J Wood Chem Technol 20(3):277–305
Samylova OA, Aizenshtadt AM, Bogolitsyn KG, Bogdanov MV, Chukhchin DG, Morozova YuG (2002) Effective potential of coniferous native lignin preparations in aqueous-alkaline medium. For J 6:98–107
Sarkanen KV, Ludwig CH (1971) Lignins: occurrence, formation, structure and reactions. Wiley, New York
Tejado A, Pena C, Labidi J, Echeverria JM, Mondragon I (2007) Physico-chemical characterization of lignins from different sources for use in phenol–formaldehyde resin synthesis. Bioresour Technol 98:1655–1663
Vanholme R, Demedts B, Morreel K, Ralph J, Boerjan W (2010) Lignin biosynthesis and structure. Plant Physiol 153:895–905
Zakis GF (1994) Functional analysis of lignins and their derivatives. GaTAPPI Press, Atlanta
Zubov IN, Khviyuzov SS, Lobanova MA, Gusakova MA, Bogolitsyn KG (2012) Influence of abiotic factors on the formation of carbohydrate matrix of juniper wood. For J 1:113–120
Acknowledgements
This research was funded by the Federal Agency of Scientific Organizations of Russia under the project agreement No. AAAA-A18-118012390231-9 and the Russian Foundation for Basic Research, project No. 14-03-31551 mol_a. We used the equipment of the “Critical Technologies of Russian Federation in the Field of Environmental Safety of Arctic” Centre of Collective Use of Scientific Equipment (Federal Center for Integrated Arctic Research, Russia) and the instrumentation of the Core Facility Center “Arktika” of Northern (Arctic) Federal University, Russia (unique identifier RFMEFI59417X0013).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bogolitsyn, K.G., Khviuzov, S.S., Gusakova, M.A. et al. The differences between acid–base and redox properties of phenolic structures of coniferous and deciduous native lignins. Wood Sci Technol 52, 1153–1164 (2018). https://doi.org/10.1007/s00226-018-1008-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00226-018-1008-z