Abstract
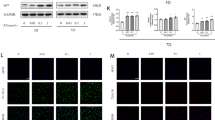
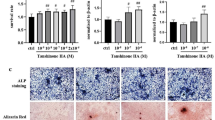
Monotropein is one of the active ingredients in Morinda Officinalis, which has been used for the treatment in multiple bone and joint diseases. This study aimed to observe the in vitro effects of Monotropein on osteogenic differentiation of lipopolysaccharide treated bone marrow mesenchymal stem cells (bMSCs), and the in vivo effects of local application of Monotropein on bone fracture healing in ovariectomized mice. Lipopolysaccharide was used to set up the inflammatory model in bMSCs, which were treated by Monotropein. Molecular docking analysis was performed to evaluate the potential interaction between Monotropein and p65. Transverse fractures of middle tibias were established in ovariectomized mice, and Monotropein was locally applied to the fracture site using injectable hydrogel. Monotropein enhanced the ability of primary bMSCs in chondro-osteogenic differentiation. Furthermore, Monotropein rescued lipopolysaccharide-induced osteogenic differentiation impairment and inhibited lipopolysaccharide-induced p65 phosphorylation in primary bMSCs. Docking analysis showed that the binding activity of Monotropein and p65/14-3-3 complex is stronger than the selective inhibitor of NF-κB (p65), DP-005. Local application of Monotropein partially rescued the decreased bone mass and biomechanical properties of callus or healed tibias in ovariectomized mice. The expressions of Runx2, Osterix and Collagen I in the 2-week callus were partially restored in Monotropein-treated ovariectomized mice. Taking together, local application of Monotropein promoted fracture healing in ovariectomized mice. Inhibition of p65 phosphorylation and enhancement in osteogenesis of mesenchymal stem cells could be partial of the effective mechanisms.






Similar content being viewed by others
References
Cheung WH, Miclau T, Chow SK, Yang FF, Alt V (2016) Fracture healing in osteoporotic bone. Injury 47(Suppl 2):S21-26
Cauley JA, Thompson DE, Ensrud KC, Scott JC, Black D (2000) Risk of mortality following clinical fractures. Osteoporosis Int 11:556–561
Bliuc D, Nguyen ND, Nguyen TV, Eisman JA, Center JR (2013) Compound risk of high mortality following osteoporotic fracture and refracture in elderly women and men. J Bone Min Res 28:2317–2324
Si L, Winzenberg TM, Jiang Q, Chen M, Palmer AJ (2015) Projection of osteoporosis-related fractures and costs in China: 2010–2050. Osteoporosis Int 26:1929–1937
Zhang JH, Xin HL, Xu YM, Shen Y, He YQ, Hsien-Yeh LB, Song HT, Juan-Liu YHY, Qin LP, Zhang QY, Du J (2018) Morinda officinalis how—a comprehensive review of traditional uses, phytochemistry and pharmacology. J Ethnopharmacol 213:230–255
He YQ, Yang H, Shen Y, Zhang JH, Zhang ZG, Liu LL, Song HT, Lin B, Hsu HY, Qin LP, Han T, Xin HL, Zhang QY (2018) Monotropein attenuates ovariectomy and LPS-induced bone loss in mice and decreases inflammatory impairment on osteoblast through blocking activation of NF-kappaB pathway. Chem Biol Interact 291:128–136
Zhang Z, Zhang Q, Yang H, Liu W, Zhang N, Qin L, Xin H (2016) Monotropein isolated from the roots of Morinda officinalis increases osteoblastic bone formation and prevents bone loss in ovariectomized mice. Fitoterapia 110:166–172
Xia T, Dong X, Lin L, Jiang Y, Ma X, Xin H, Zhang Q, Qin L (2019) Metabolomics profiling provides valuable insights into the underlying mechanisms of Morinda officinalis on protecting glucocorticoid-induced osteoporosis. J Pharm Biomed Anal 166:336–346
Balakrishnan B, Soman D, Payanam U, Laurent A, Labarre D, Jayakrishnan A (2017) A novel injectable tissue adhesive based on oxidized dextran and chitosan. Acta Biomater 53:343–354
Jiang F, Tang ZM, Zhang YH, Ju YH, Gao HQ, Sun N, Liu F, Gu P, Zhang WA (2019) Enhanced proliferation and differentiation of retinal progenitor cells through a self-healing injectable hydrogel. Biomater Sci-Uk 7:2335–2347
Shu B, Zhao Y, Zhao S, Pan H, Xie R, Yi D, Lu K, Yang J, Xue C, Huang J, Wang J, Zhao D, Xiao G, Wang Y, Chen D (2020) Inhibition of Axin1 in osteoblast precursor cells leads to defects in postnatal bone growth through suppressing osteoclast formation. Bone Res 8:31
Wolter M, de Vink P, Neves JF, Srdanovic S, Higuchi Y, Kato N, Wilson A, Landrieu I, Brunsveld L, Ottmann C (2020) Selectivity via cooperativity: preferential stabilization of the p65/14-3-3 interaction with semisynthetic natural products. J Am Chem Soc 142:11772–11783
Lu C, Miclau T, Hu D, Hansen E, Tsui K, Puttlitz C, Marcucio RS (2005) Cellular basis for age-related changes in fracture repair. J Orthop Res 23:1300–1307
Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR (1999) Multilineage potential of adult human mesenchymal stem cells. Science 284:143–147
Shao J, Zhang W, Yang T (2015) Using mesenchymal stem cells as a therapy for bone regeneration and repairing. Biol Res 48:62
Cenci S, Weitzmann MN, Roggia C, Namba N, Novack D, Woodring J, Pacifici R (2000) Estrogen deficiency induces bone loss by enhancing T-cell production of TNF-alpha. J Clin Investig 106:1229–1237
Zheng SX, Vrindts Y, Lopez M, De Groote D, Zangerle PF, Collette J, Franchimont N, Geenen V, Albert A, Reginster JY (1997) Increase in cytokine production (IL-1 beta, IL-6, TNF-alpha but not IFN-gamma, GM-CSF or LIF) by stimulated whole blood cells in postmenopausal osteoporosis. Maturitas 26:63–71
Chen L, Cheng S, Sun K, Wang J, Liu X, Zhao Y, Yang J, Zhao D, Xue C, Tao Y, Zhao S, Zhang H, Lu S, Shi Q, Wang Y, Shu B (2021) Changes in macrophage and inflammatory cytokine expressions during fracture healing in an ovariectomized mice model. BMC Musculoskelet Disord 22:494
Huang H, Zhao N, Xu X, Xu Y, Li S, Zhang J, Yang P (2011) Dose-specific effects of tumor necrosis factor alpha on osteogenic differentiation of mesenchymal stem cells. Cell Prolif 44:420–427
Wang F, Wu L, Li L, Chen S (2014) Monotropein exerts protective effects against IL-1beta-induced apoptosis and catabolic responses on osteoarthritis chondrocytes. Int Immunopharmacol 23:575–580
Wang C, Mao C, Lou Y, Xu J, Wang Q, Zhang Z, Tang Q, Zhang X, Xu H, Feng Y (2018) Monotropein promotes angiogenesis and inhibits oxidative stress-induced autophagy in endothelial progenitor cells to accelerate wound healing. J Cell Mol Med 22:1583–1600
Chen Y, Lu Y, Pei C, Liang J, Ding P, Chen S, Hou SZ (2020) Monotropein alleviates secondary liver injury in chronic colitis by regulating TLR4/NF-kappaB signaling and NLRP3 inflammasome. Eur J Pharmacol 883:173358
Jiang F, Xu XR, Li WM, Xia K, Wang LF, Yang XC (2020) Monotropein alleviates H2O2induced inflammation, oxidative stress and apoptosis via NFkappaB/AP1 signaling. Mol Med Rep 22:4828–4836
Ingles-Esteve J, Morales M, Dalmases A, Garcia-Carbonell R, Jene-Sanz A, Lopez-Bigas N, Iglesias M, Ruiz-Herguido C, Rovira A, Rojo F, Albanell J, Gomis RR, Bigas A, Espinosa L (2012) Inhibition of specific NF-kappa B activity contributes to the tumor suppressor function of 14-3-3 sigma in breast cancer. Plos One 7:e38347
Zhou XY, Hu DX, Chen RQ, Chen XQ, Dong WL, Yi CL (2017) 14-3-3 Isoforms differentially regulate NF kappa B signaling in the brain after ischemia-reperfusion. Neurochem Res 42:2354–2362
Nguyen DT, Burg KJ (2015) Bone tissue engineering and regenerative medicine: targeting pathological fractures. J Biomed Mater Res, Part A 103:420–429
Vas WJ, Shah M, Al Hosni R, Owen HC, Roberts SJ (2017) Biomimetic strategies for fracture repair: engineering the cell microenvironment for directed tissue formation. J Tissue Eng 8:2041731417704791
Irastorza-Lorenzo A, Sanchez-Porras D, Ortiz-Arrabal O, de Frutos MJ, Esteban E, Fernandez J, Janer A, Campos A, Campos F, Alaminos M (2021) Evaluation of marine agarose biomaterials for tissue engineering applications. Int J Mol Sci 22:1923
Gibbs DM, Black CR, Dawson JI, Oreffo RO (2016) A review of hydrogel use in fracture healing and bone regeneration. J Tissue Eng Regen Med 10:187–198
Zhang W, Shi Y, Li H, Yu M, Zhao J, Chen H, Kong M (2022) In situ injectable nano-complexed hydrogel based on chitosan/dextran for combining tumor therapy via hypoxia alleviation and TAMs polarity regulation. Carbohydr Polym 288:119418
Du X, Liu Y, Wang X, Yan H, Wang L, Qu L, Kong D, Qiao M, Wang L (2019) Injectable hydrogel composed of hydrophobically modified chitosan/oxidized-dextran for wound healing. Mater Sci Eng C Mater Biol Appl 104:109930
Nelson VJ, Dinnunhan MFK, Turner PR, Faed JM, Cabral JD (2017) A chitosan/dextran-based hydrogel as a delivery vehicle of human bone-marrow derived mesenchymal stem cells. Biomed Mater 12:035012
Ding XX, Li X, Li CY, Qi ML, Zhang Z, Sun XL, Wang L, Zhou YM (2019) Chitosan/dextran hydrogel constructs containing strontium-doped hydroxyapatite with enhanced osteogenic potential in rat cranium. Acs Biomater Sci Eng 5:4574–4586
Tong XM, Yang F (2018) Recent progress in developing injectable matrices for enhancing cell delivery and tissue regeneration. Adv Healthc Mater 7:1701065
Aziz MA, Cabral JD, Brooks HJL, McConnell MA, Fitzpatrick C, Hanton LR, Moratti SC (2015) In vitro biocompatibility and cellular interactions of a chitosan/dextran-based hydrogel for postsurgical adhesion prevention. J Biomed Mater Res B 103:332–341
Xiao H, Wang L, Zhang T, Chen C, Chen H, Li S, Hu J, Lu H (2020) Periosteum progenitors could stimulate bone regeneration in aged murine bone defect model. J Cell Mol Med 24:12199–12210
Li D, Yang Z, Luo Y, Zhao X, Tian M, Kang P (2022) Delivery of MiR335-5p-pendant tetrahedron DNA nanostructures using an injectable heparin lithium hydrogel for challenging bone defects in steroid-associated osteonecrosis. Adv Healthcare Mater 11:e2101412
Ni P, Ding Q, Fan M, Liao J, Qian Z, Luo J, Li X, Luo F, Yang Z, Wei Y (2014) Injectable thermosensitive PEG-PCL-PEG hydrogel/acellular bone matrix composite for bone regeneration in cranial defects. Biomaterials 35:236–248
Ji X, Yuan X, Ma L, Bi B, Zhu H, Lei Z, Liu W, Pu H, Jiang J, Jiang X, Zhang Y, Xiao J (2020) Mesenchymal stem cell-loaded thermosensitive hydroxypropyl chitin hydrogel combined with a three-dimensional-printed poly(epsilon-caprolactone) /nano-hydroxyapatite scaffold to repair bone defects via osteogenesis, angiogenesis and immunomodulation. Theranostics 10:725–740
Funding
This work was partially supported by the National Natural Science Foundation of China (Grant Numbers 81929004, 81973876 and 82274555).
Author information
Authors and Affiliations
Contributions
BS, WZ and XL designed the study. Experimental work and data collection were performed by SZ, LG, WC, YZ, YS, KS, HZ, DZ, XH and ZH. JW and SL were responsible for statistical analysis of the data. The first draft of the manuscript was written by BS, SZ and LG. WZ, XL and YW revised the manuscript critically. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
Shitian Zhao, Liqiang Guo, Wei Cui, Yongjian Zhao, Jing Wang, Kanghui Sun, Hong Zhang, Yueli Sun, Dongfeng Zhao, Xiaohui Hu, Ziyu Huang, Sheng Lu, Yongjun Wang, Xinhua Liu, Weian Zhang, and Bing Shu declares that there is no conflict of interest regarding the publication of this article.
Human and Animal Rights and Informed Consent
The experimental protocols were performed with the approval of the Laboratory Animal Management and Use (Animal Welfare) Committee of Longhua Hospital Affiliated to Shanghai University of Traditional Chinese Medicine (LHERAW-19051), and all the international, national and institutional guidelines for the care and use of animals were followed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, S., Guo, L., Cui, W. et al. Monotropein Protects Mesenchymal Stem Cells from Lipopolysaccharide-Induced Impairments and Promotes Fracture Healing in an Ovariectomized Mouse Model. Calcif Tissue Int 113, 558–570 (2023). https://doi.org/10.1007/s00223-023-01130-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-023-01130-y