Abstract
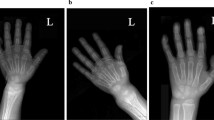
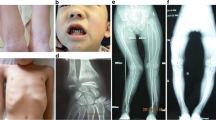
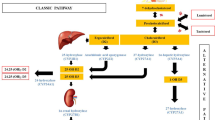
Vitamin D-dependent rickets type IA (VDDR-IA) is caused by biallelic mutations in CYP27B1. Data regarding genotype–phenotype correlation in VDDR-IA are scarce. Here, we aimed to investigate clinical/genotypic features and long-term follow-up of 13 new cases with VDDR-IA and genotype–phenotype correlation of reported cases in the literature. Thirteen patients with VDDR-IA were evaluated. Eight patients had reached their final height at the time of the study and, for whom, long-term outcome data were analyzed. Further, all VDDR-IA patients in the literature (n:183) were analyzed and clinical–genetic features were recorded. The median age of diagnosis was 2.55 ± 1.13 (1.0–12) years. Initial diagnoses before referral to our clinic were nutritional rickets (n:7), hypophosphatemic rickets (n:2), and pseudohypoparathyroidism (n:1). All had biochemical evidence suggestive of VDDR-IA; except one with elevated 1,25(OH)2D3 and another with hyperphosphatemia, in whom pseudohypoparathyroidism was excluded with molecular tests. Combined analyses of our cohort and other series in the literature demonstrated that three most common CYP27B1 mutations are p.F443Pfs*24, c.195 + 2T > G, and p.V88Wfs*71. In Turkish population, p.K192E mutation along with the former two is the most common mutations. Comparison of clinical features demonstrated that c.195 + 2T > G mutation causes the most severe and p.K192E mutation causes the least severe phenotype with respect to age and height at presentation and calcitriol requirement. We found a clear genotype–phenotype correlation in VDDR-IA, notably CYP27B1 intronic c.195 + 2T > G mutation causes a more severe phenotype with lower height SDS at presentation and, higher calcitriol requirement, while less severe phenotype occurs in p.K192E mutation.


Similar content being viewed by others
References
Chiellini G, DeLuca F (2011) The importance of stereochemistry on the actions of vitamin D. Curr Top Med Chem 11:840–859
Acar S, Demir K, Shi Y (2017) Genetic causes of rickets. J Clin Res Pediatr Endocrinol 9:88–105. https://doi.org/10.4274/jcrpe.2017.S008
Roizen JD, Li D, O’Lear L et al (2018) CYP3A4 mutation causes vitamin D-dependent rickets type 3. J Clin Invest 128:1913–1918
Fraser D, Kooh SW, Kind HP et al (1973) Pathogenesis of hereditary vitamin-D-dependent rickets: an inborn error of vitamin D metabolism involving defective conversion of 25-hydroxyvitamin D to 1α, 25-dihydroxyvitamin D. N Engl J Med 289:817–822
Edouard T, Alos N, Chabot G et al (2011) Short- and long-term outcome of patients with pseudo-vitamin D deficiency rickets treated with calcitriol. J Clin Endocrinol Metab 96:82–89. https://doi.org/10.1210/jc.2010-1340
Giannakopoulos A, Efthymiadou A, Chrysis D (2017) A case of vitamin-D-dependent rickets type 1A with normal 1,25-dihydroxyvitamin D caused by two novel mutations of the CYP27B1 gene. Horm Res Paediatr 87:58–63. https://doi.org/10.1159/000446774
Tahir S, Demirbilek H, Ozbek MN et al (2016) Genotype and phenotype characteristics in 22 patients with vitamin D-dependent rickets type I. Horm Res Paediatr 85:309–317. https://doi.org/10.1159/000444483
Demir K, Kattan WE, Zou M et al (2015) Novel CYP27B1 gene mutations in patients with vitamin D-dependent rickets type 1A. PLoS ONE 10:1–14. https://doi.org/10.1371/journal.pone.0131376
Baykan O, Yaman A, Arpa M et al (2014) The comparison of serum vitamin D3 measurement with HPLC, HPLC coupled tandem mass spectrometry using atmospheric pressure chemical ionization, and immunoassay methods. Clin Chem Lab Med 52:3. https://doi.org/10.1515/cclm-2014-4042
Untergasser A, Cutcutache I, Koressaar T et al (2012) Primer3—new capabilities and interfaces. Nucleic Acids Res 40:e115–e115
Stenson PD, Ball EV, Mort M et al (2003) Human gene mutation database (HGMD®): 2003 update. Hum Mutat 21:577–581
Wang JT, Lin CJ, Burridge SM et al (1998) Genetics of vitamin D 1α-hydroxylase deficiency in 17 families. Am J Hum Genet 63:1694–1702. https://doi.org/10.1086/302156
Chan JK, Kaplan LE, Perwad F et al (2007) Vitamin D 1α-hydroxylase gene mutations in patients with 1α-hydroxylase deficiency. J Clin Endocrinol Metab 92:3177–3182. https://doi.org/10.1210/jc.2006-2664
Ito N, Peña AS, Perano S et al (2014) First Australian report of vitamin D-dependent rickets type I. Med J Aust 201:420–421. https://doi.org/10.5694/mja13.00220
Durmaz E, Zou M, Al-Rijjal RA et al (2012) Clinical and genetic analysis of patients with vitamin D-dependent rickets type 1A. Clin Endocrinol (Oxf) 77:363–369. https://doi.org/10.1111/j.1365-2265.2012.04394.x
Fatma D, Gamze O, Heves K et al (2019) Genetic and clinical characteristics of patients with vitamin D dependent rickets type 1A. J Clin Res Pediatr Endocrinol 11:34–40. https://doi.org/10.4274/jcrpe.galenos.2018.2018.0121
Chi Y, Sun J, Pang L et al (2018) Mutation update and long-term outcome after treatment with active vitamin D3 in Chinese patients with pseudovitamin D-deficiency rickets (PDDR). Osteoporos Int 30:481–489. https://doi.org/10.1007/s00198-018-4607-5
Cui N, Xia W, Su H et al (2012) Novel mutations of CYP27B1 gene lead to reduced activity of 1α-hydroxylase in Chinese patients. Bone 51:563–569. https://doi.org/10.1016/j.bone.2012.05.006
Smith SJ, Rucka AK, Berry JL et al (1999) Three families with pseudovitamin D-deficiency rickets resulting in loss of functional enzyme activity. J bone Miner Res 14:730–739
Yoshida T, Monkawa T, Tenenhouse HS et al (1998) Two novel 1α-hydroxylase mutations in French-Canadians with vitamin D dependency rickets type I. Kidney Int 54:1437–1443. https://doi.org/10.1046/j.1523-1755.1998.00133.x
Beck-Nielsen SS, Hertel NT, Brock-Jacobsen B (2006) Vitamin D 1 alpha-hydroxylase deficiency as the cause of severe rickets in a 1-year-old-old boy. Ugeskr Laeger 168:700–702
Porcu L, Meloni A, Casula L et al (2002) A novel splicing defect (IVS6+ 1G> T) in a patient with pseudovitamin D deficiency rickets. J Endocrinol Invest 25:557–560
Orbak Z (2017) A novel mutation of CYP27B1 in two siblings with vitamin D-dependent rickets type 1A. In: 8th International Conference on Children
Kitanaka S, Murayama A, MToshioyuki S et al (1999) No enzyme activity of 25-hydroxyvitamin D3 1-a hydroxylase gene product in pseudovitamin D deficiency rickets, including that with mild clinical manifestation. J Clin Endocrinol Metab 84:4111–4117
Wang X, Zhang MYH, Miller WL, Portale AA (2002) Novel gene mutations in patients with 1α-hydroxylase deficiency that confer partial enzyme activity in vitro. J Clin Endocrinol Metab 87:2424–2430. https://doi.org/10.1210/jc.87.6.2424
Alzahrani AS, Zou M, Baitei EY et al (2010) Alzahrani2010 a novel G102E mutation of CYP27B1 in a large family with vitamin D-dependent rickets type 1.pdf. J Clin Endocrinol Metab 95:4176–4183
Füchtbauer L, Brusgaard K, Ledaal P et al (2015) Case report: vitamin D-dependent rickets type 1 caused by a novel CYP27B1 mutation. Clin Case Reports 3:1012–1016. https://doi.org/10.1002/ccr3.406
Haffner D, Emma F, Eastwood DM et al (2019) Clinical practice recommendations for the diagnosis and management of X-linked hypophosphataemia. Nat Rev Nephrol 15:435–455. https://doi.org/10.1038/s41581-019-0152-5
Hu WW, Ke YH, He JW et al (2014) A novel compound mutation of CYP27B1 in a Chinese family with vitamin D-dependent rickets type 1A. J Pediatr Endocrinol Metab 27:335–341. https://doi.org/10.1515/jpem-2013-0183
Kim CJ (2011) Vitamin D dependent rickets type I. Korean J Pediatr 54:51–54. https://doi.org/10.3345/kjp.2011.54.2.51
Kitanaka S, Takeyama K, Murayama A et al (1998) Inactivating mutations in the 25-hydroxyvitamin D3 1α-hydroxylase gene in patients with pseudovitamin D-deficiency rickets. N Engl J Med 338:653–662
Larner AJ (1995) Pseudohyperphosphatemia. Clin Biochem 28:391–393
Koek WNH, Zillikens MC, van der Eerden BCJ, van Leeuwen JPTM (2016) Novel compound heterozygous mutations in the CYP27B1 gene lead to pseudovitamin D-deficient rickets. Calcif Tissue Int 99:326–331. https://doi.org/10.1007/s00223-016-0165-z
Nishikawa M, Yasuda K, Takamatsu M et al (2019) Generation of 1, 25-dihydroxyvitamin D3 in Cyp27b1 knockout mice by treatment with 25-hydroxyvitamin D3 rescued their rachitic phenotypes. J Steroid Biochem Mol Biol 185:71–79
Slominski AT, Manna PR, Tuckey RC (2015) On the role of skin in the regulation of local and systemic steroidogenic activities. Steroids. https://doi.org/10.1016/j.steroids.2015.04.006.On
Wang J, Slominski A, Tuckey RC, Janjetovic Z (2012) 20-Hydroxyvitamin D3 inhibits proliferation of cancer cells with high efficacy while being non-toxic. Anticancer Res 32:739–746
Acknowledgements
We are deeply grateful to the patients and their families without whom this study could not be performed.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Sare Betul Kaygusuz, Ceren Alavanda, Tarik Kirkgoz, Mehmet Eltan, Zehra Yavas Abali, Didem Helvacioglu, Tulay Guran, Pinar Ata, Abdullah Bereket, and Serap Turan declare that they have no conflicts of interest.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Marmara University School of Medicine Ethical Committee and with the 1964 Helsinki declaration and its later amendments.
Consent to Participate
Informed consent was obtained from parents of all patients for genetic testing.
Consent for Publication
Informed consent was obtained from parents of all patients for publication of data.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kaygusuz, S.B., Alavanda, C., Kirkgoz, T. et al. Does Genotype–Phenotype Correlation Exist in Vitamin D-Dependent Rickets Type IA: Report of 13 New Cases and Review of the Literature. Calcif Tissue Int 108, 576–586 (2021). https://doi.org/10.1007/s00223-020-00784-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-020-00784-2