Abstract
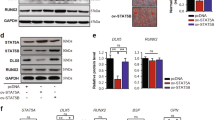
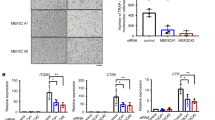
Sphingosine-1-phosphate (S1P) is an anabolic clastokine. Colony Stimulating Factor 1 (CSF1) induces expression of the rate limiting enzyme required for S1P synthesis, sphingosine kinase 1 (SPHK1) in bone in vivo, and in osteoclasts in vitro. To study the mechanism of CSF1-induced SPHK1 gene expression, a 2608 bp fragment of the murine SPHK1 gene (− 2497 to + 111 bp relative to the transcription start site) was cloned and transfected into pZen cells (murine fibroblasts engineered to express c-fms). SPHK1 promoter activity was assessed using a dual-luciferase reporter assay system. By analyzing a series of 5′-deletions, a CSF1-responsive region was identified in the region − 1250 to − 1016 bp. To define putative DNA binding site(s) in this fragment, two biotin-labeled fragments that completely overlapped this region were generated, one 163 bp in length (− 1301 to − 1139) and one 169 bp in length (− 1157 to − 989). EMSAs revealed the 163 bp fragment as the target for protein binding. Using EMSAs, the nuclear protein binding region was further narrowed to an 85 bp fragment, (− 1223 to − 1139). Using a series of unlabeled DNA sequences as “cold competitors” in EMSAs, a 22 bp sequence is identified as the smallest fragment that could successfully compete away protein binding. The same 22 bp sequence also competed DNA binding in EMSAs using nuclear protein isolated from primary murine osteoclasts. A full-length wild-type SPHK1 promoter and an SPHK1 promoter in which the ATGGGGG motif was mutated were subsequently expressed in pZen cells. Mutating this ATGGGGG motif nearly completely abrogated the ability of CSF1 to activate the promoter. Although two transcription factors, KLF6 and Sp1 have been reported to bind to this sequence, supershift EMSAs failed to detect either among the proteins bound to the 85 bp DNA fragment.




Similar content being viewed by others
References
Abboud SL, Ghosh-Choudhury N, Liu LC, Shen V, Woodruff K (2003) Osteoblast-specific targeting of soluble colony-stimulating factor-1 increases cortical bone thickness in mice. J Bone Miner Res 18:1386–1394
Lloyd SA, Yuan YY, Simske SJ, Riffle SE, Ferguson VL, Bateman TA (2009) Administration of high-dose macrophage colony-stimulating factor increases bone turnover and trabecular volume fraction. J Bone Miner Metab 27:546–554
Lotinun S, Kiviranta R, Matsubara T, Alzate JA, Neff L, Lüth A, Koskivirta I, Kleuser B, Vacher J, Vuorio E, Horne WC, Baron R (2013) Osteoclast-specific cathepsin K deletion stimulates S1P-dependent bone formation. J Clin Investig 123:666–681
Keller J, Catala-Lehnen P, Huebner AK, Jeschke A, Heckt T, Lueth A, Krause M, Koehne T, Albers J, Schulze J, Schilling S, Haberland M, Denninger H, Neven M, Hermans-Borgmeyer I, Streichert T, Breer S, Barvencik F, Levkau B, Rathkolb B, Wolf E, Calzada-Wack J, Neff F, Gailus-Durner V, Fuchs H, de Angelis MH, Klutmann S, Tsourdi E, Hofbauer LC, Kleuser B, Chun J, Schinke T, Amling M (2014) Calcitonin controls bone formation by inhibiting the release of sphingosine 1-phosphate from osteoclasts. Nat Commun 5:5215
Gandy KA, Obeid LM (2013) Regulation of the sphingosine kinase/sphingosine 1-phosphate pathway. In: Gulbins E, Petrache I (eds) Sphingolipids in disease (handbook of experimental pharmacology). Springer, Wien
Pitson SM, Moretti PA, Zebol JR, Lynn HE, Xia P, Vadas MA, Wattenberg BW (2003) Activation of sphingosine kinase 1 by ERK1/2-mediated phosphorylation. EMBO J 22:5491–5500
Sato C, Iwasaki T, Kitano S, Tsunemi S, Sano H (2012) Sphingosine 1-phosphate receptor activation enhances BMP-2-induced osteoblast differentiation. Biochem Biophys Res Commun 423:200–205
Alvarez SE, Milstien S, Spiegel S (2007) Autocrine and paracrine roles of sphingosine-1-phosphate. Trends Endocrinol Metab 18:300–307
Quint P, Ruan M, Pederson L, Kassem M, Westendorf JJ, Khosla S, Oursler MJ (2013) Sphingosine 1-phosphate (S1P) receptors 1 and 2 coordinately induce mesenchymal cell migration through S1P activation of complementary kinase pathways. J Biol Chem 288:5398–5406
Yao G-Q, Zhu M, Sun B-H, Walker J, Insogna KL (2016) The anabolic actions of PTH are mediated in part through a colony stimulating factor 1-sphingosine-1-phosphate paracrine loop. J Bone Min Res 31(Supp 1). https://www.asbmr.org/education/AbstractDetail?aid=e30fc568-568b568d-544fd-abb564-567acba00701df. Accessed 10 July 2019
Yao C, Yao GQ, Sun BH, Zhang C, Tommasini SM, Insogna K (2014) The transcription factor T-box 3 regulates colony-stimulating factor 1-dependent Jun dimerization protein 2 expression and plays an important role in osteoclastogenesis. J Biol Chem 289:6775–6790
Reedijk M, Liu XQ, Pawson T (1990) Interactions of phosphatidylinositol kinase, GTPase-activating protein (GAP), and GAP-associated proteins with the colony-stimulating factor 1 receptor. Mol Cell Biol 10:5601–5608
Weir EC, Horowitz MC, Baron R, Centrella M, Kacinski BM, Insogna KL (1993) Macrophage colony-stimulating factor release and receptor expression in bone cells. J Bone Miner Res 8:1507–1518
Cartharius K, Frech K, Grote K, Klocke B, Haltmeier M, Klingenhoff A, Frisch M, Bayerlein M, Werner T (2005) MatInspector and beyond: promoter analysis based on transcription factor binding sites. Bioinformatics 21:2933–2942
Zhao JL, Austen KF, Lam BK (2000) Cell-specific transcription of leukotriene C(4) synthase involves a Kruppel-like transcription factor and Sp1. J Biol Chem 275:8903–8910
Lassen NE, Andersen TL, Ploen GG, Soe K, Hauge EM, Harving S, Eschen GET, Delaisse JM (2017) Coupling of bone resorption and formation in real time: new knowledge gained from human haversian BMUs. J Bone Miner Res 32:1395–1405
Simms N, Martin TJ (2014) Coupling the activities of bone formation and resorption: a multitude of signals within the basic multicellular unit. Bone Key Rep 3:481
Drissi H, Sanjay A (2016) The multifaceted osteoclast; far and beyond bone resorption. J Cell Biochem 117:1753–1756
Crane JL, Cao X (2014) Function of matrix IGF-1 in coupling bone resorption and formation. J Mol Med (Berl) 92:107–115
Zhao C, Irie N, Takada Y, Shimoda K, Miyamoto T, Nishiwaki T, Suda T, Matsuo K (2006) Bidirectional ephrinB2-EphB4 signaling controls bone homeostasis. Cell Metab 4:111–121
Kim JH, Kim N (2016) Signaling pathways in osteoclast differentiation. Chonnam Med J 52:12–17
Hofstetter W, Wetterwald A, Cecchini MC, Felix R, Fleisch H, Mueller C (1992) Detection of transcripts for the receptor for macrophage colony-stimulating factor, c-fms, in murine osteoclasts. Proc Natl Acad Sci USA 89:9637–9641
Grey A, Chen Y, Paliwal I, Carlberg K, Insogna K (2000) Evidence for a functional association between phosphatidylinositol 3-kinase and c-src in the spreading response of osteoclasts to colony-stimulating factor-1. Endocrinology 141:2129–2138
Bryan L, Kordula T, Spiegel S, Milstien S (2008) Regulation and functions of sphingosine kinases in the brain. Biochim Biophys Acta 1781:459–466
Acknowledgements
This work was supported by a grant from NIH (NIAMS, AR069088) to KLI
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Gang Qing Yao, Meiling Zhu, Joanne Walker and Karl Insogna declare no conflict of interest.
Human and Animal Rights and Informed Consent
The Yale Animal Care and Use Committee approved the use of animals in this study. All applicable institutional and/or national guidelines for the care and use of animals were followed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yao, G.Q., Zhu, M., Walker, J. et al. Identification of a 22 bp DNA cis Element that Plays a Critical Role in Colony Stimulating Factor 1-Dependent Transcriptional Activation of the SPHK1 Gene. Calcif Tissue Int 107, 52–59 (2020). https://doi.org/10.1007/s00223-020-00685-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-020-00685-4