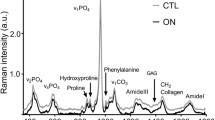
Abstract
Ischemic osteonecrosis of the femoral head (IOFH) can lead to excessive resorption of the trabecular bone and collapse of the femoral head as a structure. A well-known mineral component to trabecular bone is hydroxyapatite, which can be present in many forms due to ionic substitution, thus altering chemical composition. Unfortunately, very little is known about the chemical changes to bone apatite following IOFH. We hypothesized that the apatite composition changes in necrotic bone possibly contribute to increased osteoclast resorption and structural collapse of the femoral head. The purpose of this study was to assess the macroscopic and local phosphate composition of actively resorbed necrotic trabecular bone to isolate differences between areas of increased osteoclast resorption and normal bone formation. A piglet model of IOFH was used. Scanning electron microscopy (SEM), histology, X-ray absorbance near edge structure (XANES), and Raman spectroscopy were performed on femoral heads to characterize normal and necrotic trabecular bone. Backscattered SEM, micro-computed tomography and histology showed deformity and active resorption of necrotic bone compared to normal. XANES and Raman spectroscopy obtained from actively resorbed necrotic bone and normal bone showed increased carbonate-to-phosphate content in the necrotic bone. The changes in the apatite composition due to carbonate substitution may play a role in the increased resorption of necrotic bone due to its increase in solubility. Indeed, a better understanding of the apatite composition of necrotic bone could shed light on osteoclast activity and potentially improve therapeutic treatments that target excessive resorption of bone.






Similar content being viewed by others
References
Ha YC, Jung WH, Kim JR, Seong NH, Kim SY, Koo KH (2006) Prediction of collapse in femoral head osteonecrosis: a modified Kerboul method with use of magnetic resonance images. J Bone Joint Surg Am 88(Suppl 3):35–40
Conzemius MG, Brown TD, Zhang Y, Robinson RA (2002) A new animal model of femoral head osteonecrosis: one that progresses to human-like mechanical failure. J Orthop Res 20:303–309
Kim HK, Su PH (2002) Development of flattening and apparent fragmentation following ischemic necrosis of the capital femoral epiphysis in a piglet model. J Bone Joint Surg Am 84(A):1329–1334
Hofstaetter JG, Roschger P, Klaushofer K, Kim HK (2010) Increased matrix mineralization in the immature femoral head following ischemic osteonecrosis. Bone 46:379–385
Aruwajoye OO, Patel MK, Allen MR, Burr DB, Aswath PB, Kim HKW (2013) Microcrack density and nanomechanical properties in the subchondral region of the immature piglet femoral head following ischemic osteonecrosis. Bone 52:632–639
Norman D, Reis D, Zinman C, Misselevich I, Boss JH (1998) Vascular deprivation-induced necrosis of the femoral head of the rat. An experimental model of avascular osteonecrosis in the skeletally immature individual or Legg-Perthes disease. Int J Exp Pathol 79:173
Rowe SM, Lee JJ, Chung JY, Moon ES, Song EK, Seo HY (2006) Deformity of the femoral head following vascular infarct in piglets. Acta Orthop 77:33–38
Kim HKW, Aruwajoye O, Stetler J, Stall A (2012) Effects of non-weight-bearing on the immature femoral head following ischemic osteonecrosisan experimental investigation in immature pigs. J Bone Joint Surg 94:2228–2237
Kim HK, Randall TS, Bian H, Jenkins J, Garces A, Bauss F (2005) Ibandronate for prevention of femoral head deformity after ischemic necrosis of the capital femoral epiphysis in immature pigs. J Bone Joint Surg Am 87:550–557
Capuccini C, Torricelli P, Sima F, Boanini E, Ristoscu C, Bracci B, Socol G, Fini M, Mihailescu I, Bigi A (2008) Strontium-substituted hydroxyapatite coatings synthesized by pulsed-laser deposition: in vitro osteoblast and osteoclast response. Acta Biomater 4:1885–1893
Spence G, Patel N, Brooks R, Bonfield W, Rushton N (2010) Osteoclastogenesis on hydroxyapatite ceramics: The effect of carbonate substitution. J Biomed Mater Res, Part A 92:1292–1300
Shepherd DV, Kauppinen K, Brooks RA, Best SM (2014) An in vitro study into the effect of zinc substituted hydroxyapatite on osteoclast number and activity. J Biomed Mater Res A 102(11):4136–4141
Yerramshetty JS, Akkus O (2008) The associations between mineral crystallinity and the mechanical properties of human cortical bone. Bone 42:476–482
Awonusi A, Morris MD, Tecklenburg MM (2007) Carbonate assignment and calibration in the Raman spectrum of apatite. Calcif Tissue Int 81:46–52
Morris MD, Mandair GS (2011) Raman assessment of bone quality. Clin Orthop Relat Res® 469:2160–2169
Bazin D, Daudon M, Chappard C, Rehr J, Thiaudiere D, Reguer S (2011) The status of strontium in biological apatites: an XANES investigation. J Synchrotron Radiat 18:912–918
Meirer F, Pemmer B, Pepponi G, Zoeger N, Wobrauschek P, Sprio S, Tampieri A, Goettlicher J, Steininger R, Mangold S (2011) Assessment of chemical species of lead accumulated in tidemarks of human articular cartilage by X-ray absorption near-edge structure analysis. J Synchrotron Radiat 18:238–244
Dessombz A, Nguyen C, Ea HK, Rouzière S, Foy E, Hannouche D, Réguer S, Picca FE, Thiaudière D, Lioté F, Daudon M, Bazin D (2013) Combining μX-ray fluorescence, μXANES and μXRD to shed light on Zn2+ cations in cartilage and meniscus calcifications. J Trace Elem Med Biol 27(4):326–333
Bazin D, Carpentier X, Brocheriou I, Dorfmuller P, Aubert S, Chappard C, Thiaudière D, Reguer S, Waychunas G, Jungers P (2009) Revisiting the localisation of Zn2 cations sorbed on pathological apatite calcifications made through X-ray absorption spectroscopy. Biochimie 91:1294–1300
Bazin D, Daudon M, Combes C, Rey C (2012) Characterization and some physicochemical aspects of pathological microcalcifications. Chem Rev 112:5092–5120
Rajendran J, Gialanella S, Aswath PB (2013) XANES analysis of dried and calcined bones. Mater Sci Eng C 33:3968–3979
Demirkiran H, Hu Y, Zuin L, Appathurai N, Aswath PB (2011) XANES analysis of calcium and sodium phosphates and silicates and hydroxyapatite–Bioglass®45S5 co-sintered bioceramics. Mater Sci Eng C 31:134–143
Kim HK, Su PH, Qiu YS (2001) Histopathologic changes in growth-plate cartilage following ischemic necrosis of the capital femoral epiphysis. An experimental investigation in immature pigs. J Bone Joint Surg Am 83(A):688–697
Crane NJ, Popescu V, Morris MD, Steenhuis P, Ignelzi MA (2005) Raman spectroscopic evidence for octacalcium phosphate and other transient mineral species deposited during intramembranous mineralization. Bone 39:434–442
Draper ER, Morris MD, Camacho NP, Matousek P, Towrie M, Parker AW, Goodship AE (2005) Novel assessment of bone using time-resolved transcutaneous raman spectroscopy. J Bone Miner Res 20:1968–1972
Pasteris JD, Wopenka B, Freeman JJ, Rogers K, Valsami-Jones E, van der Houwen, Jacqueline AM, Silva MJ (2004) Lack of OH in nanocrystalline apatite as a function of degree of atomic order: implications for bone and biomaterials. Biomaterials 25:229–238
Wopenka B, Pasteris JD (2005) A mineralogical perspective on the apatite in bone. Mater Sci Eng C 25:131–143
Bigi A, Cojazzi G, Panzavolta S, Ripamonti A, Roveri N, Romanello M, Noris Suarez K, Moro L (1997) Chemical and structural characterization of the mineral phase from cortical and trabecular bone. J Inorg Biochem 68:45–51
Driessens F (1980) The mineral in bone, dentin and tooth enamel. Bulletin des sociétés chimiques belges 89:663–689
Rey C, Collins B, Goehl T, Dickson I, Glimcher M (1989) The carbonate environment in bone mineral: a resolution-enhanced Fourier transform infrared spectroscopy study. Calcif Tissue Int 45:157–164
Paschalis E, DiCarlo E, Betts F, Sherman P, Mendelsohn R, Boskey A (1996) FTIR microspectroscopic analysis of human osteonal bone. Calcif Tissue Int 59:480–487
Yeni Y, Yerramshetty J, Akkus O, Pechey C, Les C (2006) Effect of fixation and embedding on Raman spectroscopic analysis of bone tissue. Calcif Tissue Int 78:363–371
Akkus O, Adar F, Schaffler MB (2004) Age-related changes in physicochemical properties of mineral crystals are related to impaired mechanical function of cortical bone. Bone 34:443–453
Penel G, Leroy G, Rey C, Bres E (1998) MicroRaman spectral study of the PO4 and CO3 vibrational modes in synthetic and biological apatites. Calcif Tissue Int 63:475–481
Ito A, Maekawa K, Tsutsumi S, Ikazaki F, Tateishi T (1997) Solubility product of OH-carbonated hydroxyapatite. J Biomed Mater Res 36:522–528
Rey C, Renugopalakrishman V, Collins B, Glimcher M (1991) Fourier transform infrared spectroscopic study of the carbonate ions in bone mineral during aging. Calcif Tissue Int 49:251–258
Sydney-Zax M, Mayer I, Deutsch D (1991) Carbonate content in developing human and bovine enamel. J Dent Res 70:913–916
Sa Y, Liang S, Ma X, Lu S, Wang Z, Jiang T, Wang Y (2014) Compositional, structural and mechanical comparisons of normal enamel and hypomaturation enamel. Acta Biomater 10(12):5169–5177
Acknowledgments
This study was funded by Texas Scottish Rite Hospital for Children. The XANES experiments were performed at the Canadian Light Source, which is supported by NSERC, NRC, CIHR, and the University of Saskatchewan. Raman experiments were conducted at Center for Characterizations for Materials and Biology at The University of Texas at Arlington.
Conflict of interest
None of the authors have any conflict of interest with any entity with regard to this study.
Human and Animal Rights and Informed Consent
This study was performed in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. This article does not contain any studies with human subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aruwajoye, O.O., Kim, H.K.W. & Aswath, P.B. Bone Apatite Composition of Necrotic Trabecular Bone in the Femoral Head of Immature Piglets. Calcif Tissue Int 96, 324–334 (2015). https://doi.org/10.1007/s00223-015-9959-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-015-9959-7