Abstract
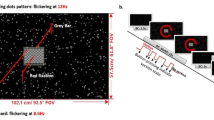
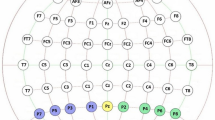
Visually induced motion sickness (VIMS) is a common phenomenon when using visual devices such as smartphones and virtual reality applications, with symptoms including nausea, fatigue, and headache. To date, the neuro-cognitive processes underlying VIMS are not fully understood. Previous studies using electroencephalography (EEG) delivered mixed findings, with some reporting an increase in delta and theta power, and others reporting increases in alpha and beta frequencies. The goal of the study was to gain further insight into EEG correlates for VIMS. Participants viewed a VIMS-inducing visual stimulus, composed of moving black-and-white vertical bars presented on an array of three adjacent monitors. The EEG was recorded during visual stimulation and VIMS ratings were recorded after each trial using the Fast Motion Sickness Scale. Time–frequency analyses were conducted comparing neural activity of participants reporting minimal VIMS (n = 21) and mild–moderate VIMS (n = 12). Results suggested a potential increase in delta power in the centro-parietal regions (CP2) and a decrease in alpha power in the central regions (Cz) for participants experiencing mild–moderate VIMS compared to those with minimal VIMS. Event-related spectral perturbations (ERSPs) suggested that group differences in EEG activity developed with increasing duration of a trial. These results support the hypothesis that the EEG might be sensitive to differences in information processing in VIMS and minimal VIMS contexts, and indicate that it may be possible to identify neurophysiological correlate of VIMS. Differences in EEG activity related to VIMS may reflect differential processing of conflicting visual and vestibular sensory information.




Similar content being viewed by others
Data availability
Anonymized data may be made available by the authors upon request.
Notes
No sex-related differences showed with respect to VIMS severity or vection intensity. Thus, biological sex will not be considered for further discussion.
Note that we deliberately chose a baseline that contained visual motion. If a baseline with a static visual scene was applied, differences in the EEG recordings would likely be dominated by the processing of visual motion in general (and not specific to VIMS). In other words, a baseline with visual motion may help to control at least partially the portion of the EEG associated with the processing of motion.
Note that no statistical tests are reported for the ERSP analysis as these analysis were purely exploratory and no hypotheses were generated a priori. The purpose of these analyses were to deliver insights into the dynamics of potential differences between the two VIMS groups.
References
Andrews PLR (2013) Nausea, vomiting, and the autonomic nervous system. In: Mathias CJ, Bannister SR (eds) Autonomic Failure: A Textbook of Clinical Disorders of the Autonomic Nervous System. Oxford University Press, p 0
Andrievskaia P, Spaniol J, Berti S, Keshavarz B (2023) Influence of stimulus speed and cognitive factors on cortical responses to vection-inducing stimuli. Poster presented at the 1st International Conference on Ageing, Innovation, and Rehabilitation (iCAIR). Toronto, ON.
Berti S, Keshavarz B (2020) Neuropsychological approaches to visually-induced vection: an overview and evaluation of neuroimaging and neurophysiological studies. Multisens Res 34(2):153–186. https://doi.org/10.1163/22134808-bja10035
Caserman P, Garcia-Agundez A, Gámez Zerban A, Göbel S (2021) Cybersickness in current-generation virtual reality head-mounted displays: systematic review and outlook. Virtual Reality. https://doi.org/10.1007/s10055-021-00513-6
Cha Y-H, Baloh RW, Cho C et al (2020) Mal de débarquement syndrome diagnostic criteria: Consensus document of the Classification Committee of the Bárány Society. J Vestib Res 30:285–293. https://doi.org/10.3233/VES-200714
Chang E, Billinghurst M, Yoo B (2023) Brain activity during cybersickness: a scoping review. Virtual Reality. https://doi.org/10.1007/s10055-023-00795-y
Chelen WE, Kabrisky M, Rogers SK (1993) Spectral analysis of the electroencephalographic response to motion sickness. Aviat Space Environ Med 64:24–29
Chen Y-C, Duann J-R, Chuang S-W et al (2010) Spatial and temporal EEG dynamics of motion sickness. Neuroimage 49:2862–2870. https://doi.org/10.1016/j.neuroimage.2009.10.005
Cheung B, Vaitkus P (1998) Perspectives of electrogastrography and motion sickness. Brain Res Bull 47:421–431
Chuang S-W, Chuang C-H, Yu Y-H et al (2016) EEG Alpha and Gamma Modulators Mediate Motion Sickness-Related Spectral Responses. Int J Neural Syst 26:1650007. https://doi.org/10.1142/S0129065716500076
Cobb SV (1999) Measurement of postural stability before and after immersion in a virtual environment. Appl Ergon 30:47–57
Cohen MX, Donner TH (2013) Midfrontal conflict-related theta-band power reflects neural oscillations that predict behavior. J Neurophysiol 110:2752–2763. https://doi.org/10.1152/jn.00479.2013
Cowings PS, Suter S, Toscano WB et al (1986) General Autonomic Components of Motion Sickness. Psychophysiology 23:542–551. https://doi.org/10.1111/j.1469-8986.1986.tb00671.x
D’Amour S, Bos JE, Keshavarz B (2017) The efficacy of airflow and seat vibration on reducing visually induced motion sickness. Exp Brain Res 235:2811–2820. https://doi.org/10.1007/s00221-017-5009-1
D’Amour S, Harris LR, Berti S, Keshavarz B (2021) The role of cognitive factors and personality traits in the perception of illusory self-motion (vection). Atten Percept Psychophys 83:1804–1817. https://doi.org/10.3758/s13414-020-02228-3
Delorme A, Makeig S (2004) EEGLAB: an open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J Neurosci Methods 134:9–21. https://doi.org/10.1016/j.jneumeth.2003.10.009
Dennison MS, Wisti AZ, D’Zmura M (2016) Use of physiological signals to predict cybersickness. Displays 44:42–52. https://doi.org/10.1016/j.displa.2016.07.002
Drummer C, Stromeyer H, Riepl RL et al (1990) Hormonal changes after parabolic flight: implications on the development of motion sickness. Aviat Space Environ Med 61:821–828
Ebenholtz SM, Cohen MM, Linder BJ (1994) The possible role of nystagmus in motion sickness: a hypothesis. Aviat Space Environ Med 65:1032–1035
Foxe JJ, Snyder AC (2011) The Role of Alpha-Band Brain Oscillations as a Sensory Suppression Mechanism during Selective Attention. Front Psychol. https://doi.org/10.3389/fpsyg.2011.00154
Gianaros PJ, Quigley KS, Muth ER et al (2003) Relationship between temporal changes in cardiac parasympathetic activity and motion sickness severity. Psychophysiology 40:39–44
Golding JF (1992) Phasic skin conductance activity and motion sickness. Aviat Space Environ Med 63:165–171
Haciahmet CC, Frings C, Beste C et al (2023) Posterior delta/theta EEG activity as an early signal of Stroop conflict detection. Psychophysiology. https://doi.org/10.1111/psyp.14195
Haile A, Watts M, Aichner S et al (2022) Central correlates of placebo effects in nausea differ between men and women. Brain Behav. https://doi.org/10.1002/brb3.2685
Henry EH, Bougard C, Bourdin C, Bringoux L (2022) Changes in Electroencephalography Activity of Sensory Areas Linked to Car Sickness in Real Driving Conditions. Front Hum Neurosci. https://doi.org/10.3389/fnhum.2021.809714
Hu S, Grant WF, Stern RM, Koch KL (1991) Motion sickness severity and physiological correlates during repeated exposures to a rotating optokinetic drum. Aviat Space Environ Med 62:308–314
Hu S, McChesney KA, Player KA et al (1999) Systematic investigation of physiological correlates of motion sickness induced by viewing an optokinetic rotating drum. Aviat Space Environ Med 70:759–765
Jacob C, Olliges E, Haile A et al (2023) Placebo effects on nausea and motion sickness are resistant to experimentally-induced stress. Sci Rep 13:9908. https://doi.org/10.1038/s41598-023-36296-w
Kennedy RS, Lane NE, Berbaum KS, Lilienthal MG (1993) Simulator Sickness Questionnaire: An Enhanced Method for Quantifying Simulator Sickness. Int J Aviat Psychol 3:203–220. https://doi.org/10.1207/s15327108ijap0303_3
Keshavarz B, Golding JF (2022) Motion sickness: current concepts and management. Curr Opin Neurol 35:107–112. https://doi.org/10.1097/WCO.0000000000001018
Keshavarz B, Hecht H (2011) Validating an Efficient Method to Quantify Motion Sickness. Human Factors: the Journal of the Human Factors and Ergonomics Society 53:415–426. https://doi.org/10.1177/0018720811403736
Keshavarz B, Riecke BE, Hettinger LJ, Campos JL (2015) Vection and visually induced motion sickness: how are they related? Front Psychol. https://doi.org/10.3389/fpsyg.2015.00472
Keshavarz B, Speck M, Haycock B, Berti S (2017) Effect of Different Display Types on Vection and Its Interaction With Motion Direction and Field Dependence. Iperception. https://doi.org/10.1177/2041669517707768
Keshavarz B, Murovec B, Mohanathas N, Golding JF (2021) The Visually Induced Motion Sickness Susceptibility Questionnaire (VIMSSQ): Estimating Individual Susceptibility to Motion Sickness-Like Symptoms When Using Visual Devices. Hum Factors. https://doi.org/10.1177/00187208211008687
Keshavarz B, Peck K, Rezaei S, Taati B (2022) Detecting and predicting visually induced motion sickness with physiological measures in combination with machine learning techniques. Int J Psychophysiol 176:14–26. https://doi.org/10.1016/j.ijpsycho.2022.03.006
Kim YY, Kim HJ, Kim EN et al (2005) Characteristic changes in the physiological components of cybersickness. Psychophysiology 42:616–625. https://doi.org/10.1111/j.1469-8986.2005.00349.x
Klimesch W (2012) Alpha-band oscillations, attention, and controlled access to stored information. Trends Cogn Sci 16:606–617. https://doi.org/10.1016/j.tics.2012.10.007
Kooijman L, Berti S, Asadi H, et al (2022) Measuring vection: A review and critical evaluation of different methods for quantifying illusory self-motion. PsyArXiv
Krokos E, Varshney A (2022) Quantifying VR cybersickness using EEG. Virtual Reality 26:77–89. https://doi.org/10.1007/s10055-021-00517-2
Lawson BD, Riecke BE (2014) The Perception of Body Motion. In: Hale KS, Stanney KM (eds) Handbook of Virtual Environments: Design, Implementation, and Applications, 2nd edn. CRC Press, pp 163–195
Lin C-T, Tsai S-F, Ko L-W (2013) EEG-based learning system for online motion sickness level estimation in a dynamic vehicle environment. IEEE Trans Neural Netw Learn Syst 24:1689–1700. https://doi.org/10.1109/TNNLS.2013.2275003
Min B-C, Chung S-C, Min Y-K, Sakamoto K (2004) Psychophysiological evaluation of simulator sickness evoked by a graphic simulator. Appl Ergon 35:549–556. https://doi.org/10.1016/j.apergo.2004.06.002
Miyazaki J, Yamamoto H, Ichimura Y et al (2015) Inter-hemispheric desynchronization of the human MT+ during visually induced motion sickness. Exp Brain Res 233:2421–2431. https://doi.org/10.1007/s00221-015-4312-y
Muth ER (2006) Motion and space sickness: intestinal and autonomic correlates. Auton Neurosci 129:58–66. https://doi.org/10.1016/j.autneu.2006.07.020
Muth ER, Stern RM, Koch KL (1996) Effects of vection-induced motion sickness on gastric myoelectric activity and oral-cecal transit time. Dig Dis Sci 41:330–334
Napadow V, Sheehan JD, Kim J et al (2013) The brain circuitry underlying the temporal evolution of nausea in humans. Cereb Cortex 23:806–813. https://doi.org/10.1093/cercor/bhs073
Naqvi SAA, Badruddin N, Jatoi MA et al (2015) EEG based time and frequency dynamics analysis of visually induced motion sickness (VIMS). Australas Phys Eng Sci Med 38:721–729. https://doi.org/10.1007/s13246-015-0379-9
Neuper C, Scherer R, Reiner M, Pfurtscheller G (2005) Imagery of motor actions: Differential effects of kinesthetic and visual–motor mode of imagery in single-trial EEG. Cogn Brain Res 25:668–677. https://doi.org/10.1016/j.cogbrainres.2005.08.014
Nürnberger M, Klingner C, Witte OW, Brodoehl S (2021) Mismatch of visual-vestibular information in virtual reality: is motion sickness part of the brains attempt to reduce the prediction error? Front Hum Neurosci 15. https://doi.org/10.3389/fnhum.2021.757735
Oman CM (1990) Motion sickness: a synthesis and evaluation of the sensory conflict theory. Can J Physiol Pharmacol 68:294–303
Otto B, Riepl RL, Klosterhalfen S, Enck P (2006) Endocrine correlates of acute nausea and vomiting. Auton Neurosci 129:17–21. https://doi.org/10.1016/j.autneu.2006.07.010
Pan F, Zhang L, Ou Y, Zhang X (2019) The audio-visual integration effect on music emotion: Behavioral and physiological evidence. PLoS ONE. https://doi.org/10.1371/journal.pone.0217040
Peck K, Russo F, Campos JL, Keshavarz B (2020) Examining potential effects of arousal, valence, and likability of music on visually induced motion sickness. Exp Brain Res 238:2347–2358. https://doi.org/10.1007/s00221-020-05871-2
Poonian SK, McFadyen J, Ogden J, Cunnington R (2015) Implicit Agency in Observed Actions: Evidence for N1 Suppression of Tones Caused by Self-made and Observed Actions. J Cogn Neurosci 27:752–764. https://doi.org/10.1162/jocn_a_00745
Reason JT (1978) Motion sickness adaptation: a neural mismatch model. J R Soc Med 71:819–829
Reason JT, Brand JJ (1975) Motion sickness. Academic Press, London, New York
Rebenitsch L, Owen C (2016) Review on Cybersickness in Applications and Visual Displays. Virtual Real 20:101–125. https://doi.org/10.1007/s10055-016-0285-9
Riccio GE, Stoffregen TA (1991) An ecological Theory of Motion Sickness and Postural Instability. Ecol Psychol 3:195–240. https://doi.org/10.1207/s15326969eco0303_2
Tauscher J-P, Witt A, Bosse S et al (2020) Exploring neural and peripheral physiological correlates of simulator sickness. Comput Animat Virtual Worlds. https://doi.org/10.1002/cav.1953
van Driel J, Swart JC, Egner T et al (2015) (No) time for control: Frontal theta dynamics reveal the cost of temporally guided conflict anticipation. Cogn Affect Behav Neurosci 15:787–807. https://doi.org/10.3758/s13415-015-0367-2
Warwick-Evans LA, Church RE, Hancock C et al (1987) Electrodermal activity as an index of motion sickness. Aviat Space Environ Med 58:417–423
Wei Y, Okazaki YO, So RHY et al (2019) Motion sickness-susceptible participants exposed to coherent rotating dot patterns show excessive N2 amplitudes and impaired theta-band phase synchronization. Neuroimage. https://doi.org/10.1016/j.neuroimage.2019.116028
Wei Y, Wang Y, Okazaki YO et al (2023) Motion sickness resistant people showed suppressed steady-state visually evoked potential (SSVEP) under vection-inducing stimulation. Cogn Neurodyn. https://doi.org/10.1007/s11571-023-09991-7
Wood CD, Stewart JJ, Wood MJ et al (1990) Therapeutic effects of antimotion sickness medications on the secondary symptoms of motion sickness. Aviat Space Environ Med 61:157–161
Wu JP (1992) EEG changes in man during motion sickness induced by parallel swing. Space Med Med Eng 5(3):200–205
Yates BJ, Catanzaro MF, Miller DJ, McCall AA (2014) Integration of vestibular and emetic gastrointestinal signals that produce nausea and vomiting: potential contributions to motion sickness. Exp Brain Res 232:2455–2469. https://doi.org/10.1007/s00221-014-3937-6
Zhang C, Li S, Li Y et al (2020) Analysis of Motion Sickness Associated Brain Activity Using fNIRS: A Driving Simulator Study. IEEE Access 8:207415–207425. https://doi.org/10.1109/ACCESS.2020.3038039
Zumer JM, Scheeringa R, Schoffelen J-M et al (2014) Occipital Alpha Activity during Stimulus Processing Gates the Information Flow to Object-Selective Cortex. PLoS Biol. https://doi.org/10.1371/journal.pbio.1001965
Acknowledgements
We thank Bruce Haycock and Robert Shewaga for technical assistance and programming of the visual stimulus.
Funding
Funding for this study was awarded by the Natural Sciences and Engineering Research Council of Canada (NSERC RGPIN-2017-04387) to Behrang Keshavarz.
Author information
Authors and Affiliations
Contributions
All authors contributed to study conceptualization and design. Material preparation, data collection and analysis were performed by PA, BK, and SB. The first draft of the manuscript was written by PA and BK, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethics approval
The study complied with the American Psychological Association Code of Ethics and was approved by the Institutional Review Boards at the University Health Network and Toronto Metropolitan University.
Consent to participate and publish
Informed consent was obtained from all individuals included in the study. Only anonymized and de-identifiable data are presented in the manuscript.
Additional information
Communicated by Bill J Yates.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Andrievskaia, P., Berti, S., Spaniol, J. et al. Exploring neurophysiological correlates of visually induced motion sickness using electroencephalography (EEG). Exp Brain Res 241, 2463–2473 (2023). https://doi.org/10.1007/s00221-023-06690-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-023-06690-x