Abstract
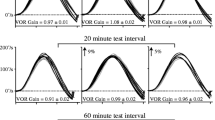
The vestibulo-ocular reflex (VOR) functions to maintain eye stability during head movement, and VOR gain can be dynamically increased or decreased by gain-up or gain-down adaptation. In this study, we investigated the impact of a differential training paradigm with varying frequencies and amplitudes on the level of VOR adaptation in mice. Training for gain-up (out of phase) or gain-down (in phase) VOR adaptation was applied for 60 min using two protocols: (1) oscillation of a drum and turntable with fixed frequency and differing amplitudes (0.5 Hz/2.5°, 0.5 Hz/5° and 0.5 Hz/10°). (2) Oscillation of a drum and turntable with fixed amplitude and a differing frequency (0.25 Hz/5°, 0.5 Hz/5° and 1 Hz/5°). VOR adaptation occurred distinctively in gain-up and gain-down learning. In gain-up VOR adaptation, the learned increase in VOR gain was greatest when trained with the same frequency and amplitude as the test stimulation, and VOR gain decreased after gain-up training with too high a frequency or amplitude. In gain-down VOR adaptation, the decrease in VOR gain increased as the training frequency or amplitude increased. These results suggest that different mechanisms are, at least in part, involved in gain-up and gain-down VOR adaptation.






Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
References
Anzai M, Nagao S (2014) Motor learning in common marmosets: vestibulo-ocular reflex adaptation and its sensitivity to inhibitors of Purkinje cell long-term depression. Neurosci Res 83:33–42. https://doi.org/10.1016/j.neures.2014.04.002
Bonnan A, Rowan MMJ, Baker CA, Bolton MM, Christie JM (2021) Autonomous Purkinje cell activation instructs bidirectional motor learning. Nat Commun 12:2153. https://doi.org/10.1038/s41467-021-22405-8
Boyden ES, Raymond JL (2003) Active reversal of motor memories reveals rules governing memory encoding. Neuron 39:1031–1042
Boyden ES, Katoh A, Pyle JL, Chatila TA, Tsien RW, Raymond JL (2006) Selective engagement of plasticity mechanisms for motor memory storage. Neuron 51:823–834. https://doi.org/10.1016/j.neuron.2006.08.026
Broussard DM, Titley HK, Antflick J, Hampson DR (2011) Motor learning in the VOR: the cerebellar component. Exp Brain Res 210:451–463. https://doi.org/10.1007/s00221-011-2589-z
De Zeeuw CI, Hansel C, Bian F, Koekkoek SK, van Alphen AM, Linden DJ, Oberdick J (1998) Expression of a protein kinase C inhibitor in Purkinje cells blocks cerebellar LTD and adaptation of the vestibulo-ocular reflex. Neuron 20:495–508
du Lac S, Raymond JL, Sejnowski TJ, Lisberger SG (1995) Learning and memory in the vestibulo-ocular reflex. Annu Rev Neurosci 18:409–441. https://doi.org/10.1146/annurev.ne.18.030195.002205
Fanning AS (2021) Population calcium responses of Purkinje cells in the oculomotor cerebellum. Nat Commun 126:1391–1402. https://doi.org/10.1038/s41467-021-22405-8
Faulstich BM, Onori KA, du Lac S (2004) Comparison of plasticity and development of mouse optokinetic and vestibulo-ocular reflexes suggests differential gain control mechanisms. Vision Res 44:3419–3427. https://doi.org/10.1016/j.visres.2004.09.006
Faulstich M, van Alphen AM, Luo C, du Lac S, De Zeeuw CI (2005) Defective control and adaptation of reflex eye movements in mutant mice deficient. J Neurophysiol 21:1315–1326. https://doi.org/10.1152/jn.00045.2006
França de Barros F, Schenberg L, Tagliabue M, Beraneck M (2020) Long term visuo-vestibular mismatch in freely behaving mice differentially affects gaze stabilizing reflexes. Sci Rep 10:20018. https://doi.org/10.1038/s41598-020-77026-w
Gao Z, van Beugen BJ, De Zeeuw CI (2012) Distributed synergistic plasticity and cerebellar learning. Nat Rev Neurosci 13:619–635. https://doi.org/10.1038/nrn3312
Godaux E, Halleux J, Gobert C (1983) Adaptive change of the vestibulo-ocular reflex in the cat: the effects of a long-term frequency-selective procedure. Exp Brain Res 49:28–34
Grasselli G (2020) SK2 channels in cerebellar Purkinje cells contribute to excitability modulation. J Neurosci 18:e3000596. https://doi.org/10.1523/jneurosci.1651-19.2020
Hansel C, Linden DJ, D’Angelo E (2001) Beyond parallel fiber LTD: the diversity of synaptic and non-synaptic plasticity in the cerebellum. Nat Neurosci 4:467–475. https://doi.org/10.1038/87419
Hubner PP, Khan SI, Migliaccio AA (2014) Velocity-selective adaptation of the horizontal and cross-axis vestibulo-ocular reflex in the mouse. Exp Brain Res 232:3035–3046. https://doi.org/10.1007/s00221-014-3988-8
Jang DC, Shim HG, Kim SJ (2020) Intrinsic plasticity of cerebellar purkinje cells contributes to motor memory. Neuroscience 40:4145–4157. https://doi.org/10.1016/j.neuroscience.2020.07.021
Kim YG, Kim SJ (2020) Decreased intrinsic excitability of cerebellar Purkinje cells following optokinetic learning in mice. Mol Brain 13:136. https://doi.org/10.1186/s13041-020-00678-2
Kimpo RR, Boyden ES, Katoh A, Ke MC, Raymond JL (2005) Distinct patterns of stimulus generalization of increases and decreases in VOR gain. J Neurophysiol 94:3092–3100. https://doi.org/10.1152/jn.00048.2005
Kuki Y, Hirata Y, Blazquez PM, Heiney SA, Highstein SM (2004) Memory retention of vestibuloocular reflex motor learning in squirrel monkeys. NeuroReport 15:1007–1011
Nagao S (2021) Ocular reflex adaptation as an experimental model of cerebellar learning. Proc Natl Acad Sci U S A 462:191–204. https://doi.org/10.1073/pnas.2102635118
Paige GD, Sargent EW (1991) Visually-induced adaptive plasticity in the human vestibulo-ocular reflex. Exp Brain Res 84:25–34
Peter S, Urbanus BHA, Klaassen RV (2020) AMPAR auxiliary protein SHISA6 facilitates Purkinje cell synaptic excitability. J Neurosci 31:107515. https://doi.org/10.1523/jneurosci.1651-19.2020
Pham NC, Kim YG, Kim SJ, Kim CH (2019) Differential effects of inferior olive lesion on vestibulo-ocular and optokinetic motor learning. NeuroReport. https://doi.org/10.1097/wnr.0000000000001333
Pham NC, Kim YG, Kim SJ, Kim CH (2020) Differential effects of inferior olive lesion on vestibulo-ocular and optokinetic motor learning. NeuroReport 31:9–16. https://doi.org/10.1097/wnr.0000000000001333
Pham NC, Kim YG, Kim SJ, Kim C-H (2021) Efficacy of spaced learning in adaptation of optokinetic response. Brain Behav 11:e01944. https://doi.org/10.1002/brb3.1944
Pham NC, Kim YG, Kim SJ, Kim C-H (2022a) Adaptation of optokinetic reflex by training with different frequency and amplitude. JIN. https://doi.org/10.31083/j.jin2105131
Pham NC, Kim YG, Kim SJ, Kim CH (2022b) Adaptation of vestibulo-ocular and optokinetic reflexes after massed and spaced vestibulo-ocular motor learning. Behav Brain Res 426:113837. https://doi.org/10.1016/j.bbr.2022.113837
Pompeiano O, Van Neerven J, Collewijn H, Van der Steen J (1991) Changes in VOR adaptation after local injection of beta-noradrenergic agents in the flocculus of rabbits. Acta Otolaryngol 111:176–181
Raymond JL, Lisberger SG (1996) Behavioral analysis of signals that guide learned changes in the amplitude and dynamics of the vestibulo-ocular reflex. J Neurosci 16:7791–7802
Raymond JL, Soga J (2021) Cerebellar roles in frequency competitive motor learning of the vestibulo-ocular. J Neurophysiol 462:205–219. https://doi.org/10.1152/jn.00715.2020
Rinaudo CN, Schubert MC, Figtree WVC, Todd CJ, Migliaccio AA (2019) Human vestibulo-ocular reflex adaptation is frequency selective. J Neurophysiol 122:984–993. https://doi.org/10.1152/jn.00162.2019
Robinson DA (2022a) Basic framework of the vestibulo-ocular reflex. Prog Brain Res 267:131–153. https://doi.org/10.1016/bs.pbr.2021.10.006
Robinson DA (2022b) The functional operation of the vestibulo-ocular reflex. Prog Brain Res 267:95–130. https://doi.org/10.1016/bs.pbr.2021.10.005
Ryu C, Jang DC, Jung D et al (2017) STIM1 regulates somatic Ca(2+) signals and intrinsic firing properties of cerebellar Purkinje neurons. J Neurosci 37:8876–8894. https://doi.org/10.1523/jneurosci.3973-16.2017
Schairer JO, Bennett MV (1986) Changes in gain of the vestibulo-ocular reflex induced by combined visual and vestibular stimulation in goldfish. Brain Res 373:164–176
Schonewille M (2021) NMDARs in granule cells contribute to parallel fiber-Purkinje cell synaptic. Proc Natl Acad Sci USA. https://doi.org/10.1073/pnas.2102635118
Soga J (2021) Cerebellar roles in frequency competitive motor learning of the vestibulo-ocular. J Nerosci 462:205–219. https://doi.org/10.1016/j.neuroscience.2020.09.016
Stahl JS, van Alphen AM, De Zeeuw CI (2000) A comparison of video and magnetic search coil recordings of mouse eye movements. J Neurosci Methods 99:101–110
Tanaka S, Kawaguchi SY, Shioi G, Hirano T (2013) Long-term potentiation of inhibitory synaptic transmission onto cerebellar Purkinje neurons contributes to adaptation of vestibulo-ocular reflex. J Neurosci 33:17209–17220. https://doi.org/10.1523/jneurosci.0793-13.2013
Titley HK, Hansel C (2016) Asymmetries in cerebellar plasticity and motor learning. Cerebellum 15:87–92. https://doi.org/10.1007/s12311-014-0635-7
Titley HK, Heskin-Sweezie R, Broussard DM (2009) Consolidation and disruption of motor memory generalize across stimulus conditions in the vestibulo-ocular reflex. Brain Res 1267:37–43. https://doi.org/10.1016/j.brainres.2009.02.041
Funding
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIP) (2021R1F1A1062019).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by Bill J Yates.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
221_2023_6601_MOESM1_ESM.pptx
Supplemental data 1. Gain-up and gain-down VOR adaptation protocols. The drum and turntable were simultaneously rotated in and out of phase for the gain-down and gain-up VOR adaptation, respectively. (PPTX 7279 KB)
221_2023_6601_MOESM2_ESM.pptx
Supplemental data 2. Raw eye movements before (A) and after (B) 60 min gain-up VOR adaptation with 0.25 Hz and 5°. And raw eye movements after 24 hr in dark rearing are shown (C). (PPTX 1053 KB)
221_2023_6601_MOESM3_ESM.pptx
Supplemental data 3. Raw eye movements before (A) and after (B) 60 min gain-up VOR adaptation with 0.5 Hz and 5°. And raw eye movements after 24 hr in dark rearing are shown (C). (PPTX 1202 KB)
221_2023_6601_MOESM4_ESM.pptx
Supplemental data 4. Raw eye movements before (A) and after (B) 60 min gain-up VOR adaptation with 1 Hz and 5°. And raw eye movements after 24 hr in dark rearing are shown (C). (PPTX 1233 KB)
221_2023_6601_MOESM5_ESM.pptx
Supplemental data 5. Raw eye movements before (A) and after (B) 60 min gain-down VOR adaptation with 0.25 Hz and 5°. And raw eye movements after 24 hr in dark rearing are shown (C). (PPTX 1200 KB)
221_2023_6601_MOESM6_ESM.pptx
Supplemental data 6. Raw eye movements before (A) and after (B) 60 min gain-down VOR adaptation with 0.5 Hz and 5°. And raw eye movements after 24 hr in dark rearing are shown (C). (PPTX 1203 KB)
221_2023_6601_MOESM7_ESM.pptx
Supplemental data 7. Raw eye movements before (A) and after (B) 60 min gain-down VOR adaptation with 1 Hz and 5°. And raw eye movements after 24 hr in dark rearing are shown (C). (PPTX 1218 KB)
221_2023_6601_MOESM8_ESM.pptx
Supplemental data 8. Raw eye movements before (A) and after (B) 60 min gain-up VOR adaptation with 0.5 Hz and 2.5°. And raw eye movements after 24 hr in dark rearing are shown (C). (PPTX 1172 KB)
221_2023_6601_MOESM9_ESM.pptx
Supplemental data 9. Raw eye movements before (A) and after (B) 60 min gain-up VOR adaptation with 0.5 Hz and 10°. And raw eye movements after 24 hr in dark rearing are shown (C) (PPTX 1200 KB)
221_2023_6601_MOESM10_ESM.pptx
Supplemental data 10. Raw eye movements before (A) and after (B) 60 min gain-down VOR adaptation with 0.5 Hz and 2.5°. And raw eye movements after 24 hr in dark rearing are shown (C). (PPTX 1215 KB)
221_2023_6601_MOESM11_ESM.pptx
Supplemental data 11. Raw eye movements before (A) and after (B) 60 min gain-down VOR adaptation with 0.5 Hz and 10°. And raw eye movements after 24 hr in dark rearing are shown (C). (PPTX 1174 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pham, N.C., Kim, Y.G., Kim, S.J. et al. Effect of a differential training paradigm with varying frequencies and amplitudes on adaptation of vestibulo-ocular reflex in mice. Exp Brain Res 241, 1299–1308 (2023). https://doi.org/10.1007/s00221-023-06601-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-023-06601-0