Abstract
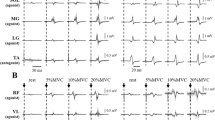
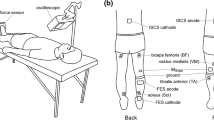
Transcutaneous spinal cord stimulation (tSCS) is a useful technique for the clinical assessment of neurological disorders. However, the characteristics of the spinal cord circuits activated by tSCS are not yet fully understood. In this study, we examined whether remote muscle contraction enhances the spinal reflexes evoked by tSCS in multiple lower-limb muscles. Eight healthy men participated in the current experiment, which required them to grip a dynamometer as fast as possible after the presentation of an auditory cue. Spinal reflexes were evoked in multiple lower-limb muscles with different time intervals (50–400 ms) after the auditory signals. The amplitudes of the spinal reflexes in all the recorded leg muscles significantly increased at 50–250 ms after remote muscle activation onset. This suggests that remote muscle contraction simultaneously facilitates the spinal reflexes in multiple lower-limb muscles. In addition, eight healthy men performed five different tasks (i.e., rest, hand grip, pinch grip, elbow flexion, and shoulder flexion). Compared to control values recorded just before each task, the spinal reflexes evoked at 250 ms after the auditory signals were significantly enhanced by the above tasks except for the rest task. This indicates that such facilitatory effects are also induced by remote muscle contractions in different upper-limb areas. The present results demonstrate the existence of a neural interaction between remote upper-limb muscles and spinal reflex circuits activated by tSCS in multiple lower-limb muscles. The combination of tSCS and remote muscle contraction may be useful for the neurological examination of spinal cord circuits.





Similar content being viewed by others
Abbreviations
- APB:
-
Abductor pollicis brevis
- AD:
-
Anterior deltoid
- BB:
-
Biceps brachii
- BF:
-
Biceps femoris
- EMG:
-
Electromyographic
- Exp:
-
Experiment
- ECR:
-
Extensor carpi radialis
- FDI:
-
First dorsal interosseous
- FCR:
-
Flexor carpi radialis
- H-reflex:
-
Hoffmann reflex
- JM:
-
Jendrássik maneuver
- MVC:
-
Maximum voluntary contraction
- MVF:
-
Maximum voluntary force
- MG:
-
Medial gastrocnemius
- RMS:
-
Root mean square
- SOL:
-
Soleus
- T-reflex:
-
Tendon-reflex
- TA:
-
Tibialis anterior
- tSCS:
-
Transcutaneous spinal cord stimulation
- VM:
-
Vastus medialis
References
Andriyanova EY (2010) Characteristics of multisegmental monosynaptic responses of leg muscles in subjects with lumber nerve compression. Hum Physiol 36:440–446
Boroojerdi B, Battaglia F, Muellbacher W, Cohen LG (2000) Voluntary teeth clenching facilitates human motor system excitability. Clin Neurophysiol 111:988–993
Burke JR, Schutten MC, Koceja DM, Kamen G (1996) Age-dependent effects of muscle vibration and the Jendrassik maneuver on the patellar tendon reflex response. Arch Phys Med Rehabil 77:600–604
Bussel B, Morin C, Pierrot-Deseilligny E (1978) Mechanism of monosynaptic reflex reinforcement during Jendrassik manoeuvre in man. J Neurol Neurosurg Psychiatry 41:40–44
Courtine G, Harkema SJ, Dy CJ, Gerasimenko YP, Dyhre-Poulsen P (2007) Modulation of multisegmental monosynaptic responses in a variety of leg muscles during walking and running in humans. J Physiol 582:1125–1139
Danner SM, Hofstoetter US, Ladenbauer J, Rattay F, Minassian K (2011) Can the human lumbar posterior columns be stimulated by transcutaneous spinal cord stimulation? A modeling study. Artif Organs 35:257–262
Danner SM, Krenn M, Hofstoetter US, Toth A, Mayr W, Minassian K (2016) Body position influences which neural structures are recruited by lumbar transcutaneous spinal cord stimulation. PLoS One 11:e0147479
Delwaide PJ, Toulouse P (1980) Jendrassik maneuver vs controlled contractions conditioning the excitability of soleus monosynaptic reflexes. Arch Phys Med Rehabil 61:505–510
Delwaide PJ, Toulouse P (1981) Facilitation of monosynaptic reflexes by voluntary contraction of muscle in remote parts of the body. Mechanisms involved in the Jendrassik Manoeuvre. Brain 104:701–709
Dietz V, Muller R, Colombo G (2002) Locomotor activity in spinal man: significance of afferent input from joint and load receptors. Brain 125:2626–2634
Dowman R, Wolpaw JR (1988) Jendrassik maneuver facilitates soleus H-reflex without change in average soleus motoneuron pool membrane potential. Exp Neurol 101:288–302
Dy CJ, Gerasimenko YP, Edgerton VR, Dyhre-Poulsen P, Courtine G, Harkema SJ (2010) Phase-dependent modulation of percutaneously elicited multisegmental muscle responses after spinal cord injury. J Neurophysiol 103:2808–2820
Furubayashi T, Sugawara K, Kasai T, Hayashi A, Hanajima R, Shiio Y, Iwata NK, Ugawa Y (2003) Remote effects of self-paced teeth clenching on the excitability of hand motor area. Exp Brain Res 148:261–265
Gerasimenko YP, Lu DC, Modaber M, Zdunowski S, Gad P, Sayenko DG, Morikawa E, Haakana P, Ferguson AR, Roy RR, Edgerton VR (2015) Noninvasive reactivation of motor descending control after paralysis. J Neurotrauma 32:1968–1980
Hagbarth KE, Wallin G, Burke D, Löfstedt L (1975) Effects of the Jendrassik manoeuvre on muscle spindle activity in man. J Neurol Neurosurg Psychiatry 38:1143–1153
Hubli M, Dietz V (2013) The physiological basis of neurorehabilitation-locomotor training after spinal cord injury. J Neuroeng Rehabil 10:5
Kawamura T, Watanabe S (1975) Timing as a prominent factor of the Jendrassik manoeuvre on the H reflex. J Neurol Neurosurg Psychiatry 38:508–516
Kitano K, Koceja DM (2009) Spinal reflex in human lower leg muscles evoked by transcutaneous spinal cord stimulation. J Neurosci Methods 180:111–115
Kojima N, Nakazawa K, Yamamoto S-I, Yano H (1998) Phase-dependent electromyographic activity of the lower-limb muscles of a patient with clinically complete spinal cord injury during orthotic gait. Exp Brain Res 120:139–142
Kojima N, Nakazawa K, Yano H (1999) Effects of limb loading on the lower-limb electromyographic activity during orthotic locomotion in a paraplegic patient. Neurosci Lett 274:211–213
Krenn M, Hofstoetter US, Danner SM, Minassian K, Mayr W (2015) Multi-electrode array for transcutaneous lumbar posterior root stimulation. Artif Organs 39:834–840
Landau WM, Clare MH (1964) Fusimotor function. VI. H reflex, tendon jerk, and reinforcement in hemiplegia. Arch Neurol 10:128–134
Masugi Y, Obata H, Nakazawa K (2017) Effects of anode position on the responses elicited by transcutaneous spinal cord stimulation. Conf Proc IEEE Eng Med Biol Soc 2017:1114–1117
Minassian K, Persy I, Rattay F, Dimitrijevic MR, Hofer C, Kern H (2007) Posterior root-muscle reflexes elicited by transcutaneous stimulation of the human lumbosacral cord. Muscle Nerve 35:327–336
Miyahara T, Hagiya N, Ohyama T, Nakamura Y (1996) Modulation of human soleus H reflex in association with voluntary clenching of the teeth. J Neurophysiol 76:2033–2041
Roy FD, Gibson G, Stein RB (2012) Effect of percutaneous stimulation at different spinal levels on the activation of sensory and motor roots. Exp Brain Res 223:281–289
Roy FD, Bosgra D, Stein RB (2014) Interaction of transcutaneous spinal stimulation and transcranial magnetic stimulation in human leg muscles. Exp Brain Res 232:1717–1728
Sayenko DG, Atkinson DA, Dy CJ, Gurley KM, Smith VL, Angeli C, Harkema SJ, Edgerton VR, Gerasimenko YP (2015) Spinal segment-specific transcutaneous stimulation differentially shapes activation pattern among motor pools in humans. J Appl Physiol (1985) 118:1364–1374
Sugawara K, Kasai T (2002) Facilitation of motor evoked potentials and H-reflexes of flexor carpi radialis muscle induced by voluntary teeth clenching. Hum Mov Sci 21:203–212
Takada Y, Miyahara T, Tanaka T, Ohyama T, Nakamura Y (2000) Modulation of H reflex of pretibial muscles and reciprocal Ia inhibition of soleus muscle during voluntary teeth clenching in humans. J Neurophysiol 83:2063–2070
Takeoka A, Vollenweider I, Courtine G, Arber S (2014) Muscle spindle feedback directs locomotor recovery and circuit reorganization after spinal cord injury. Cell 159:1626–1639
Tazoe T, Komiyama T (2014) Interlimb neural interactions in the corticospinal pathways. J Sports Med Phys Fit 3:181–190
Tazoe T, Kida T, Wasaka T, Sakamoto M, Nakajima T, Nishihira Y, Komiyama T (2005) Attenuation of the effect of remote muscle contraction on the soleus H-reflex during plantar flexion. Clin Neurophysiol 116:1362–1369
Troni W, Di Sapio A, Berra E, Duca S, Merola A, Sperli F, Bertolotto A (2011) A methodological reappraisal of non invasive high voltage electrical stimulation of lumbosacral nerve roots. Clin Neurophysiol 122:2071–2080
Zehr EP (2002) Considerations for use of the Hoffmann reflex in exercise studies. Eur J Appl Physiol 86:455–468
Zehr EP, Stein RB (1999) Interaction of the Jendrassik maneuver with segmental presynaptic inhibition. Exp Brain Res 124:474–480
Acknowledgements
This work was supported by JSPS KAKENHI Grant number 18K17760.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that no conflict of interests exist.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
221_2019_5536_MOESM1_ESM.tif
Supplementary Fig.1: Results of the double-pulse stimulation test: A) Typical example of the averaged waveform of the responses elicited by double-pulse stimulation (n=1) in multiple lower-limb muscles. tSCS was delivered at 0 ms and 50 ms. B) Medians (n=8) of the peak to-peak amplitude of both the first and the second response. The lines in the box plots indicate the medians. The ends of the boxes represent the 25th and 75th percentiles. The whiskers on the boxplot illustrate the 10th and 90th percentiles. Legend: * = p<0.05 (TIFF 7569 kb)
221_2019_5536_MOESM2_ESM.tif
Supplementary Fig.2: Results of the double-pulse stimulation test: A) Typical example of the averaged waveform of the responses elicited by double-pulse stimulation (n=1) in multiple lower-limb muscles. tSCS was delivered at 0 ms and 50 ms. B) Medians (n=8) of the peak to-peak amplitude of both the first and the second response. The lines in the box plots indicate the medians. The ends of the boxes represent the 25th and 75th percentiles. The whiskers on the boxplot illustrate the 10th and 90th percentiles. Legend: * = p<0.05 (TIFF 7047 kb)
221_2019_5536_MOESM3_ESM.tif
Supplementary Fig. 3: Background EMG activity as observed in experiment 1. Filled circles represent the mean (n=8). Unfilled circles represent the individual data points from each participant (TIFF 3890 kb)
221_2019_5536_MOESM4_ESM.tif
Supplementary Fig.4: Background EMG activity as observed in experiment 2. The lines in the box plots indicate median values. The ends of the boxes represent the 25th and 75th percentiles. The whiskers on the boxplot illustrate the 10th and 90th percentiles. Legend: * = p<0.05 (TIFF 2118 kb)
Rights and permissions
About this article
Cite this article
Masugi, Y., Sasaki, A., Kaneko, N. et al. Remote muscle contraction enhances spinal reflexes in multiple lower-limb muscles elicited by transcutaneous spinal cord stimulation. Exp Brain Res 237, 1793–1803 (2019). https://doi.org/10.1007/s00221-019-05536-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-019-05536-9