Abstract
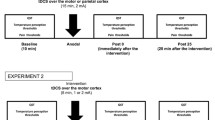
Recently (Ansari et al., PeerJ 6:e6163, 2018a; Somatosens Mot Res 35:69–79, 2018b), we showed using transcranial magnetic stimulation (TMS) that focal application of innocuous thermal stimuli to the distal hand produced variable responses in terms of motor-evoked potential (MEP) suppression or enhancement. Here, we sought to investigate possible causes of this variability by examining circuits mediating sensorimotor integration and intra-cortical inhibition. Participants (n = 21) first underwent TMS to assess baseline corticomotor excitability by measuring MEPs at rest with the index finger wrapped in a gel pack at room temperature (24 °C). Then, conditioned protocols were applied to assess short-latency afferent inhibition (SAI), short-latency afferent facilitation (SAF) and short-interval intra-cortical inhibition (SICI). Following baseline measures, MEP modulation in response to distal cooling was recorded with the index finger wrapped in a gel pack at ~ 10 °C. At baseline, participants exhibited variable levels of SAI, SAF and SICI. Participant also exhibited variable responses to cooling with about half of them (11/21) showing suppressed excitability and one-third showing enhanced excitability (7/21). A linear regression analysis revealed that SAI and SAF proved to be good predictors of cooling-induced variations in corticomotor excitability but not SICI. These results provide novel evidence linking variations in SAI and SAF with those in corticomotor excitability elicited in response to focal thermal stimulation, suggesting that these markers could be used to predict responses to sensory stimulation protocols.





Similar content being viewed by others
References
Ansari Y, Remaud A, Tremblay F (2018a) Modulation of corticomotor excitability in response to distal focal cooling. PeerJ 6:e6163. https://doi.org/10.7717/peerj.6163
Ansari Y, Remaud A, Tremblay F (2018b) Variations in corticomotor excitability in response to distal focal thermal stimulation. Somatosens Mot Res 35:69–79. https://doi.org/10.1080/08990220.2018.1460263
Bailey AZ, Asmussen MJ, Nelson AJ (2016) Short-latency afferent inhibition determined by the sensory afferent volley. J Neurophysiol 116:637–644. https://doi.org/10.1152/jn.00276.2016
Bokiniec P, Zampieri N, Lewin GR, Poulet JF (2018) The neural circuits of thermal perception. Curr Opin Neurobiol 52:98–106. https://doi.org/10.1016/j.conb.2018.04.006
Boroojerdi B, Kopylev L, Battaglia F, Facchini S, Ziemann U, Muellbacher W, Cohen LG (2000) Reproducibility of intracortical inhibition and facilitation using the paired-pulse paradigm. Muscle Nerve 23:1594–1597
Brown KE, Lohse KR, Mayer IMS et al (2017) The reliability of commonly used electrophysiology measures. Brain Stimul 10:1102–1111. https://doi.org/10.1016/j.brs.2017.07.011
Chen JC, Liang CC, Shaw FZ (2005) Facilitation of sensory and motor recovery by thermal intervention for the hemiplegic upper limb in acute stroke patients: a single-blind randomized clinical trial. Stroke 36:2665–2669. https://doi.org/10.1161/01.STR.0000189992.06654.ab
Chen JC, Lin CH, Wei YC, Hsiao J, Liang CC (2011) Facilitation of motor and balance recovery by thermal intervention for the paretic lower limb of acute stroke: a single-blind randomized clinical trial. Clin Rehabil 25:823–832. https://doi.org/10.1177/0269215511399591
Chipchase LS, Schabrun SM, Hodges PW (2011) Peripheral electrical stimulation to induce cortical plasticity: a systematic review of stimulus parameters. Clin Neurophysiol 122:456–463. https://doi.org/10.1016/j.clinph.2010.07.025
Davidson T, Tremblay F (2013) Hemispheric differences in corticospinal excitability and in transcallosal inhibition in relation to degree of handedness. PLoS One 8:e70286. https://doi.org/10.1371/journal.pone.0070286
Degardin A, Devos D, Cassim F, Bourriez J-L, Defebvre L, Derambure P, Devanne H (2011) Deficit of sensorimotor integration in normal aging. Neurosci Lett 498:208–212. https://doi.org/10.1016/j.neulet.2011.05.010
Devanne H, Degardin A, Tyvaert L et al (2009) Afferent-induced facilitation of primary motor cortex excitability in the region controlling hand muscles in humans. Eur J Neurosci 30:439–448. https://doi.org/10.1111/j.1460-9568.2009.06815.x
Di Lazzaro V, Oliviero A, Tonali PA et al (2002) Noninvasive in vivo assessment of cholinergic cortical circuits in AD using transcranial magnetic stimulation. Neurology 59:392–397
Di Lazzaro V, Profice P, Pilato F et al (2012) The level of cortical afferent inhibition in acute stroke correlates with long-term functional recovery in humans. Stroke 43:250–252. https://doi.org/10.1161/strokeaha.111.631085
Doeltgen SH, Ridding MC (2010) Behavioural exposure and sleep do not modify corticospinal and intracortical excitability in the human motor system. Clin Neurophysiol 121:448–452. https://doi.org/10.1016/j.clinph.2009.11.085
Dyke K, Kim S, Jackson GM, Jackson SR (2018) Reliability of single and paired pulse transcranial magnetic stimulation parameters across eight testing sessions. Brain Stimul 11:1393–1394. https://doi.org/10.1016/j.brs.2018.08.008
Garry M, Thomson R (2009) The effect of test TMS intensity on short-interval intracortical inhibition in different excitability states. Exp Brain Res 193:267–274
Geurts CL, Sleivert GG, Cheung SS (2005) Effect of cold-induced vasodilatation in the index finger on temperature and contractile characteristics of the first dorsal interosseus muscle during cold-water immersion. Eur J Appl Physiol 93:524–529. https://doi.org/10.1007/s00421-004-1254-7
Guerra A, Lopez-Alonso V, Cheeran B, Suppa A (2017) Variability in non-invasive brain stimulation studies: reasons and results. Neurosci Lett. https://doi.org/10.1016/j.neulet.2017.12.058
Hinder MR, Goss EL, Fujiyama H, Canty AJ, Garry MI, Rodger J, Summers JJ (2014) Inter- and Intra-individual variability following intermittent theta burst stimulation: implications for rehabilitation and recovery. Brain Stimul 7:365–371. https://doi.org/10.1016/j.brs.2014.01.004
Hooks BM (2016) Sensorimotor convergence in circuitry of the motor cortex. Neuroscientist 23:251–263. https://doi.org/10.1177/1073858416645088
Hsu HW, Lee CL, Hsu MJ, Wu HC, Lin R, Hsieh CL, Lin JH (2013) Effects of noxious versus innocuous thermal stimulation on lower extremity motor recovery 3 months after stroke. Arch Phys Med Rehabil 94:633–641. https://doi.org/10.1016/j.apmr.2012.11.021
Keel JC, Smith MJ, Wassermann EM (2001) A safety screening questionnaire for transcranial magnetic stimulation. Clin Neurophysiol 112:720. https://doi.org/10.1016/S1388-2457(00)00518-6
Koizume Y, Hirano M, Kubota S, Tanaka S, Funase K (2017) Relationship between the changes in M1 excitability after motor learning and arousal state as assessed by short-latency afferent inhibition. Behav Brain Res 330:56–62. https://doi.org/10.1016/j.bbr.2017.05.037
Lazzaro VD, Restuccia D, Oliviero A et al (1998) Magnetic transcranial stimulation at intensities below active motor threshold activates intracortical inhibitory circuits. Exp Brain Res 119:265–268. https://doi.org/10.1007/s002210050341
López-Alonso V, Cheeran B, Río-Rodríguez D, Fernández-del-Olmo M (2014) Inter-individual variability in response to non-invasive brain stimulation paradigms. Brain Stimulation 7:372–380. https://doi.org/10.1016/j.brs.2014.02.004
Mang CS, Bergquist AJ, Roshko SM, Collins DF (2012) Loss of short-latency afferent inhibition and emergence of afferent facilitation following neuromuscular electrical stimulation. Neurosci Lett 529:80–85. https://doi.org/10.1016/j.neulet.2012.08.072
Miyaguchi S, Kojima S, Sasaki R, Tamaki H, Onishi H (2017) Modulation of short-latency afferent inhibition and short-interval intracortical inhibition by test stimulus intensity and motor-evoked potential amplitude. Neuroreport 28:1202–1207. https://doi.org/10.1097/WNR.0000000000000896
Samusyte G, Bostock H, Rothwell J, Koltzenburg M (2018) Short-interval intracortical inhibition: comparison between conventional and threshold-tracking techniques. Brain Stimul. https://doi.org/10.1016/j.brs.2018.03.002
Tai I, Lai CL, Hsu MJ et al (2014) Effect of thermal stimulation on corticomotor excitability in patients with stroke. Am J Phys Med Rehabil 93:801–808. https://doi.org/10.1097/PHM.0000000000000105
Tokimura H, Di Lazzaro V, Tokimura Y et al (2000) Short latency inhibition of human hand motor cortex by somatosensory input from the hand. J Physiol 523(Pt 2):503–513
Turco CV, El-Sayes J, Savoie MJ, Fassett HJ, Locke MB, Nelson AJ (2018) Short- and long-latency afferent inhibition; uses, mechanisms and influencing factors. Brain Stimul 11:59–74. https://doi.org/10.1016/j.brs.2017.09.009
Udupa K, Ni Z, Gunraj C, Chen R (2009) Interactions between short latency afferent inhibition and long interval intracortical inhibition. Exp Brain Res 199:177–183
Wang F, Belanger E, Cote SL, Desrosiers P, Prescott SA, Cote DC, De Koninck Y (2018) Sensory afferents use different coding strategies for heat and cold. Cell Rep 23:2001–2013. https://doi.org/10.1016/j.celrep.2018.04.065
Wassermann EM (2002) Variation in the response to transcranial magnetic brain stimulation in the general population. Clin Neurophysiol 113:1165–1171
Wassermann EM, Greenberg BD, Nguyen MB, Murphy DL (2001) Motor cortex excitability correlates with an anxiety-related personality trait. Biol Psychiatry 50:377–382
Yarnall AJ, Ho BSW, Eshun E, David R, Rochester L, Burn DJ, Baker MR (2016) Short latency afferent inhibition: effects of ageing. Clin Neurophysiol 127:2410–2413. https://doi.org/10.1016/j.clinph.2016.03.014
Young-Bernier M, Davidson PS, Tremblay F (2012) Paired-pulse afferent modulation of TMS responses reveals a selective decrease in short latency afferent inhibition with age. Neurobiol Aging 33:835 e831–e811. https://doi.org/10.1016/j.neurobiolaging.2011.08.012
Young-Bernier M, Tanguay AFN, Davidson PSR, Tremblay F (2014) Short-latency afferent inhibition is a poor predictor of individual susceptibility to rTMS-induced plasticity in the motor cortex of young and older adults. Front Aging Neurosci. https://doi.org/10.3389/fnagi.2014.00182
Ziemann U, Reis J, Schwenkreis P, Rosanova M, Strafella A, Badawy R, Muller-Dahlhaus F (2015) TMS and drugs revisited 2014. Clin Neurophysiol 126:1847–1868. https://doi.org/10.1016/j.clinph.2014.08.028
Acknowledgements
The authors wish to thank all participants for their time and patience during testing. Part of this work was performed in the context of a Ph.D. in Rehabilitation Sciences by Y. Ansari. Y. Ansari received financial support from the Faculty of Health Sciences, University of Ottawa, in the form of a graduate scholarship. This research was also supported by the Graduate Studentship Program at the Bruyère Research Institute.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ansari, Y., Tremblay, F. Short-latency afferent-induced facilitation and inhibition as predictors of thermally induced variations in corticomotor excitability. Exp Brain Res 237, 1445–1455 (2019). https://doi.org/10.1007/s00221-019-05522-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-019-05522-1