Abstract
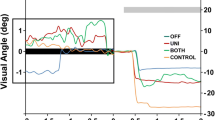
Deep brain stimulation of the subthalamic nucleus (STN DBS) significantly improves clinical motor symptoms, as well as intensive aspects of movement like velocity and amplitude in patients with Parkinson’s disease (PD). However, the effects of bilateral STN DBS on integrative and coordinative aspects of motor control are equivocal. The aim of this study was to investigate the effects of bilateral STN DBS on integrative and coordinative aspects of movement using a memory-guided sequential reaching task. The primary outcomes were eye and finger velocity and end-point error. We expected that bilateral STN DBS would increase reaching velocity. More importantly, we hypothesized that bilateral STN DBS would increase eye and finger end-point error and this would not simply be the result of a speed accuracy trade-off. Ten patients with PD and bilaterally implanted subthalamic stimulators performed a memory-guided sequential reaching task under four stimulator conditions (DBS-OFF, DBS-LEFT, DBS-RIGHT, and DBS-BILATERAL) over 4 days. DBS-BILATERAL significantly increased eye velocity compared to DBS-OFF, DBS-LEFT, and DBS-RIGHT. It also increased finger velocity compared to DBS-OFF and DBS-RIGHT. DBS-BILATERAL did not change eye end-point error. The novel finding was that DBS-BILATERAL increased finger end-point error compared to DBS-OFF, DBS-LEFT, and DBS-RIGHT even after adjusting for differences in velocity. We conclude that bilateral STN DBS may facilitate basal ganglia–cortical networks that underlie intensive aspects of movement like velocity, but it may disrupt selective basal ganglia–cortical networks that underlie certain integrative and coordinative aspects of movement such as spatial accuracy.





Similar content being viewed by others
References
Alberts JL, Voelcker-Rehage C, Hallahan K, Vitek M, Bamzai R, Vitek JL (2008) Bilateral subthalamic stimulation impairs cognitive-motor performance in Parkinson’s disease patients. Brain 131:3348–3360. https://doi.org/10.1093/brain/awn238
Alhourani A et al (2015) Network effects of deep brain stimulation. J Neurophysiol 114:2105–2117. https://doi.org/10.1152/jn.00275.2015
Ballanger B et al (2009) Stimulation of the subthalamic nucleus and impulsivity: release your horses. Ann Neurol 66:817–824. https://doi.org/10.1002/ana.21795
Barbey AK, Koenigs M, Grafman J (2013) Dorsolateral prefrontal contributions to human. working memory. Cortex 49:1195–1205. https://doi.org/10.1016/j.cortex.2012.05.022
Bastian AJ, Kelly VE, Revilla FJ, Perlmutter JS, Mink JW (2003) Different effects of unilateral versus bilateral subthalamic nucleus stimulation on walking and reaching in Parkinson’s disease. Mov Disord 18:1000–1007. https://doi.org/10.1002/mds.10493
Batista AP, Buneo CA, Snyder LH, Andersen RA (1999) Reach plans in eye-centered. coordinates. Science 285:257–260
Boecker H, Jankowski J, Ditter P, Scheef L (2008) A role of the basal ganglia and midbrain nuclei for initiation of motor sequences. Neuroimage 39:1356–1369. https://doi.org/10.1016/j.neuroimage.2007.09.069
Buneo CA, Jarvis MR, Batista AP, Andersen RA (2002) Direct visuomotor transformations for reaching. Nature 416:632–636. https://doi.org/10.1038/416632a
CANTABeclipse® [Cognitive assessment software] Cambridge Cognition (2012) All rights reserved. http://www.cantab.com. Accessed 2014
Cohen J (1988) Statistical power analysis for the behavioral sciences, 2nd edn
Goelz LC, David FJ, Sweeney JA, Vaillancourt DE, Poizner H, Metman LV, Corcos DM (2017) The effects of unilateral versus bilateral subthalamic nucleus deep brain stimulation on prosaccades and antisaccades in Parkinson’s disease. Exp Brain Res 235:615–626. https://doi.org/10.1007/s00221-016-4830-2
Goetz CG et al (2008) Movement Disorder Society—sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): scale presentation and clinimetric testing results. Mov Disord 23:2129–2170. https://doi.org/10.1002/mds.22340
Graziano MS (2006) Progress in understanding spatial coordinate systems in the primate brain. Neuron 51:7–9. https://doi.org/10.1016/j.neuron.2006.06.011
Hamlett ARL, Serrano-Trespalacios P, Wolfinger R (2003) Mixed models for assessing correlation in the presence of replication. J Air Waste Manag Assoc 53:442–450
Hening W, Harrington DL, Poizner H (2009) Basal ganglia: motor functions of. In: Binder MD, Hirokawa N, Windhorst U (eds) Encyclopedia of Neuroscience. Springer, Berlin, Heidelberg
Hershey T, Wu J, Weaver PM, Perantie DC, Karimi M, Tabbal SD, Perlmutter JS (2008) Unilateral vs. bilateral STN DBS effects on working memory and motor function in Parkinson disease. Exp Neurol 210:402–408. https://doi.org/10.1016/j.expneurol.2007.11.011
Hilker R et al (2004) Subthalamic nucleus stimulation restores glucose metabolism in associative and limbic cortices and in cerebellum: evidence from a FDG-PET study in advanced Parkinson’s disease. J Cereb Blood Flow Metab 24:7–16. https://doi.org/10.1097/01.WCB.0000092831.44769.09
Hoehn MM, Yahr MD (1967) Parkinsonism: onset progression mortality. Neurology 17:427–442
Hughes AJ, Daniel SE, Kilford L, Lees AJ (1992) Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: a clinico-pathological study of 100 cases. J Neurol Neurosurg Psychiatry 55:181–184
Innovative Sports Training (2010) The motion monitor, 8.99 edn. Innovative Sports Training, Inc., Chicago
Kumar R, Lozano AM, Sime E, Halket E, Lang AE (1999) Comparative effects of unilateral and bilateral subthalamic nucleus deep brain stimulation. Neurology 53:561–566
Lee D, Henriques DY, Snider J, Song D, Poizner H (2013) Reaching to proprioceptively defined targets in Parkinson’s disease: effects of deep brain stimulation therapy. Neuroscience 244:99–112. https://doi.org/10.1016/j.neuroscience.2013.04.009
Lohnes CA, Earhart GM (2012) Effect of subthalamic deep brain stimulation on turning kinematics and related saccadic eye movements in Parkinson disease. Exp Neurol 236:389–394. https://doi.org/10.1016/j.expneurol.2012.05.001
Mattia M et al (2012) Stop-event-related potentials from intracranial electrodes reveal a key role of premotor and motor cortices in stopping ongoing movements. Front Neuroeng 5:12. https://doi.org/10.3389/fneng.2012.00012
Mattia M, Pani P, Mirabella G, Costa S, Del Giudice P, Ferraina S (2013) Heterogeneous attractor cell assemblies for motor planning in premotor cortex. J Neurosci 33:11155–11168. https://doi.org/10.1523/JNEUROSCI.4664-12.2013
Medina J, Jax SA, Coslett HB (2009) Two-component models of reaching: evidence from deafferentation in a Fitts’ law task. Neurosci Lett 451:222–226. https://doi.org/10.1016/j.neulet.2009.01.002
Mirabella G, Pani P, Ferraina S (2011) Neural correlates of cognitive control of reaching movements in the dorsal premotor cortex of rhesus monkeys. J Neurophysiol 106:1454–1466. https://doi.org/10.1152/jn.00995.2010
Mirabella G, Iaconelli S, Romanelli P, Modugno N, Lena F, Manfredi M, Cantore G (2012) Deep brain stimulation of subthalamic nuclei affects arm response inhibition in Parkinson’s patients. Cereb Cortex 22:1124–1132. https://doi.org/10.1093/cercor/bhr187
Mirabella G, Iaconelli S, Modugno N, Giannini G, Lena F, Cantore G (2013) Stimulation of subthalamic nuclei restores a near normal planning strategy in Parkinson’s patients. PLoS One 8:e62793. https://doi.org/10.1371/journal.pone.0062793
Mosier K, Lau C, Wang Y, Venkadesan M, Valero-Cuevas FJ (2011) Controlling instabilities in manipulation requires specific cortical–striatal–cerebellar networks. J Neurophysiol 105:1295–1305. https://doi.org/10.1152/jn.00757.2010
Nambu A, Tokuno H, Inase M, Takada M (1997) Corticosubthalamic input zones from forelimb representations of the dorsal and ventral divisions of the premotor cortex in the macaque monkey: comparison with the input zones from the primary motor cortex and the supplementary motor area. Neurosci Lett 239:13–16
Nasreddine ZS et al (2005) The Montreal cognitive assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc 53:695–699. https://doi.org/10.1111/j.1532-5415.2005.53221.x
Northern Digital (2018) Northern Digital Optotrak 3020 Active-marker 3D optical tracking system. Northern Digital Inc., Waterloo
Oldfield RC (1971) The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 9:97–113
Owen AM, McMillan KM, Laird AR, Bullmore E (2005) N-back working memory paradigm: a meta-analysis of normative functional neuroimaging studies. Hum Brain Mapp 25:46–59. https://doi.org/10.1002/hbm.20131
Pani P, Di Bello F, Brunamonti E, D’Andrea V, Papazachariadis O, Ferraina S (2014) Alpha- and beta-band oscillations subserve different processes in reactive control of limb movements. Front Behav Neurosci 8:383. https://doi.org/10.3389/fnbeh.2014.00383
Pesaran B, Nelson MJ, Andersen RA (2006) Dorsal premotor neurons encode the relative position of the hand, eye, and goal during reach planning. Neuron 51:125–134. https://doi.org/10.1016/j.neuron.2006.05.025
Prodoehl J, Corcos DM, Vaillancourt DE (2009) Basal ganglia mechanisms underlying precision grip force control. Neurosci Biobehav Rev 33:900–908. https://doi.org/10.1016/j.neubiorev.2009.03.004
Sarlegna FR, Sainburg RL (2009) The roles of vision and proprioception in the planning of reaching movements Adv. Exp Med Biol 629:317–335. https://doi.org/10.1007/978-0-387-77064-2_16
Schettino LF, Adamovich SV, Hening W, Tunik E, Sage J, Poizner H (2006) Hand preshaping in Parkinson’s disease: effects of visual feedback and medication state. Exp Brain Res 168:186–202. https://doi.org/10.1007/s00221-005-0080-4
Snider J, Lee D, Harrington DL, Poizner H (2014) Scaling and coordination deficits during dynamic object manipulation in Parkinson’s disease. J Neurophysiol 112:300–315. https://doi.org/10.1152/jn.00041.2014
Spraker MB, Yu H, Corcos DM, Vaillancourt DE (2007) Role of individual basal ganglia nuclei in force amplitude generation. J Neurophysiol 98:821–834. https://doi.org/10.1152/jn.00239.2007
SR Research Eyelink II (2018) SR Research Ltd., Ottawa
Swann N et al (2011) Deep brain stimulation of the subthalamic nucleus alters the cortical profile of response inhibition in the beta frequency band: a scalp EEG study in Parkinson’s disease. J Neurosci 31:5721–5729. https://doi.org/10.1523/jneurosci.6135-10.2011
Temperli P, Ghika J, Villemure JG, Burkhard PR, Bogousslavsky J, Vingerhoets FJ (2003) How do parkinsonian signs return after discontinuation of subthalamic DBS? Neurology 60:78–81
The MathWorks (2014) Matlab, R2014b edn. The MathWorks, Inc., Natick
Thermo CRS Catalyst 5 Robot Arm (2018) Thermo CRS Ltd., Burlington
Tomlinson CL, Stowe R, Patel S, Rick C, Gray R, Clarke CE (2010) Systematic review of levodopa dose equivalency reporting in Parkinson’s disease. Mov Disord 25:2649–2653. https://doi.org/10.1002/mds.23429
Trost M et al (2006) Network modulation by the subthalamic nucleus in the treatment of Parkinson’s disease. Neuroimage 31:301–307. https://doi.org/10.1016/j.neuroimage.2005.12.024
Vafaee MS, K OS, Sunde N, Gjedde A, Dupont E, Cumming P (2004) Focal changes of oxygen consumption in cerebral cortex of patients with Parkinson’s disease during subthalamic stimulation. Neuroimage 22:966–974. https://doi.org/10.1016/j.neuroimage.2004.02.019
Vaillancourt DE, Prodoehl J, Verhagen Metman L, Bakay RA, Corcos DM (2004) Effects of deep brain stimulation and medication on bradykinesia and muscle activation in Parkinson‘s disease. Brain 127:491–504. https://doi.org/10.1093/brain/awh057
Vaillancourt DE, Prodoehl J, Sturman MM, Bakay RA, Metman LV, Corcos DM (2006) Effects of deep brain stimulation and medication on strength, bradykinesia, and electromyographic patterns of the ankle joint in Parkinson’s disease. Mov Disord 21:50–58. https://doi.org/10.1002/mds.20672
van den Wildenberg WP, van Boxtel GJ, van der Molen MW, Bosch DA, Speelman JD, Brunia CH (2006) Stimulation of the subthalamic region facilitates the selection and inhibition of motor responses in Parkinson’s disease. J Cogn Neurosci 18:626–636. https://doi.org/10.1162/jocn.2006.18.4.626
Wechsler D (2010) Wechsler Memory Scale—fourth edition (WMS-IV). Flexible approach manual. NCS Pearson Inc, San Antonio
Acknowledgements
The authors thank the participants and our professional colleagues, Maya Cottongim, and Christiane Alford for their important contributions to the successful implementation of this project.
Funding
This study was supported by National Institutes of Health (R56NS040902 and R01NS09295001). The sponsors were not involved in the design, conduct, collection, management, analysis, and/or interpretation of the study results and preparation, review, or approval of the manuscript. The views expressed in this article are those of the authors and do not necessarily reflect the position or policy of NIH. Statistical analysis: Conducted by FJD.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
FJD, RZT, and LCG have nothing to report. DMC is a Full Professor at Northwestern University and receives a salary, has additional NIH funding (5R01NS074343, 5R01HD075777, 1R01DK110699, 5T15HD074546), and receives honoraria and/or consults for the following: University of Florida, Ohio University Athens, Temple University, Iowa State University, University of Alabama, Birmingham, Oregon Health Sciences Institute, University of Westminster, University of Waterloo, University of Colorado, Denver, Several NIH Study Sections, ACRM, ASNR, University of New Hampshire, University of Minnesota, Movement Disorders Society. LVM has foundation research support from Michael J. Fox Foundation; commercial research support from Medtronic, Inc., US WorldMeds LLC, Pfizer Inc, Boston Scientific, Avanir Pharmaceuticals, Inc., and Adamas Pharmaceuticals, Inc.; is on the scientific advisory board of St. Jude Medical, AbbVie, Inc., and Britannia Pharmaceuticals Ltd.; and consults for St. Jude Medical, AbbVie, Inc., Medtronic, Inc., and Boston Scientific.
Additional information
Lisa C. Goelz is equally credited as first author.
Rights and permissions
About this article
Cite this article
David, F.J., Goelz, L.C., Tangonan, R.Z. et al. Bilateral deep brain stimulation of the subthalamic nucleus increases pointing error during memory-guided sequential reaching. Exp Brain Res 236, 1053–1065 (2018). https://doi.org/10.1007/s00221-018-5197-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-018-5197-3