Abstract
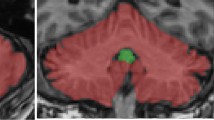
Parkinsonian-like motor impairments are common in the elderly. The etiology of these symptoms in the absence of clinically diagnosable Parkinson’s disease (PD) is unknown. The aim of this study was to evaluate associations between striatal dopaminergic neuron losses that occur with aging and gait in healthy adults. Forty healthy subjects aged 21–85 years old underwent [11C]-β-CFT dopamine transporter (DAT) positron emission tomography (PET). Subjects were also asked to walk in a gait laboratory at their own pace. Gait variables of interest included average general spatiotemporal characteristics of walking patterns and their standard deviation reflecting gait variability. Segmented nonlinear models were used to investigate the relationship between striatal DAT activity and gait while controlling for age. Gait speed, cadence, and single and double support durations were significantly slower than age-based predictions in adults with lower striatal DAT activity (P < 0.05). After controlling for age, striatal DAT activity was not significantly associated with average step length and step width and with gait variability. We conclude that dopaminergic physiology influences certain aspects of gait independent of age-related changes. The findings of this study may augur novel therapeutic approaches to treating gait disorders in the elderly.





Similar content being viewed by others
References
Antonini A, Schwarz J, Oertel WH, Pogarell O, Leenders KL (1997) Long-term changes of striatal dopamine D2 receptors in patients with Parkinson’s disease: a study with positron emission tomography and [11C]raclopride. Mov Disord 12:33–38
Azulay JP, Van Den Brand C, Mestre D, Blin O, Sangla I, Pouget J, Serratrice G (1996) Automatic motion analysis of gait in patients with Parkinson disease: effects of levodopa and visual stimulations. Rev Neurol 152:128–134
Bennett DA, Beckett LA, Murray AM, Shannon KM, Goetz CG, Pilgrim DM, Evans DA (1996) Prevalence of parkinsonian signs and associated mortality in a community population of older people. N Engl J Med 334:71–76
Blin O, Ferrandez AM, Pailhous J, Serratrice G (1991) Dopa-sensitive and dopa-resistant gait parameters in Parkinson’s disease. J Neurol Sci 103:51–454
Bloem BR, van Vugt JP, Beckley DJ (2001) Postural instability and falls in Parkinson’s disease. Adv Neurol 87:209–223
Bohnen NI, Albin RL, Koeppe RA, Wernette KA, Kilbourn MR, Minoshima S, Frey KA (2006) Positron emission tomography of monoaminergic vesicular binding in aging and Parkinson disease. J Cereb Blood Flow Metab 26:1198–1212
Bowes SG, Clark PK, Leeman AL, O’Neill CJ, Weller C, Nicholson PW, Deshmukh AA, Dobbs SM, Dobbs RJ (1990) Determinants of gait in the elderly parkinsonian on maintenance levodopa/carbidopa therapy. Br J Clin Pharm 30:13–24
Cham R, Perera S, Studenski SA, Bohnen NI (2007) Striatal dopamine denervation and sensory integration for balance in middle-aged and older adults. Gait Posture 26(4):516–525
Fahn S, Elton R (1987) Unified Parkinson’s disease rating scale. In: Fahn S, Marsden C, Calne D, Goldstein M (eds) Recent developments in Parkinson’s disease. Macmillan Healthcare Information, Florham Park, pp 153–164
Fearnley JM, Lees AJ (1991) Ageing and Parkinson’s disease: substantia nigra regional selectivity. Brain 114:2283–2301
Ferrandez AM, Blin O (1991) A comparison between the effect of intentional modulations and the action of l-dopa on gait in Parkinson’s disease. Behav Brain Res 45:177–183
Fleischman DA, Wilson RS, Schneider JA, Bienias JL, Bennett DA (2007) Parkinsonian signs and functional disability in old age. Exp Aging Res 33:59–76
Hausdorff JM, Schaafsma JD, Balash Y, Bartels AL, Gurevich T, Giladi N (2003) Impaired regulation of stride variability in Parkinson’s disease subjects with freezing of gait. Exp Brain Res 149:187–194
Hornykiewicz O (1966) Dopamine (3-hydroxytyramine) and brain function. Pharmacol Rev 18:925–964
Ichise M, Liow JS, Lu JQ, Takano A, Model K, Toyama H, Suhara T, Suzuki K, Innis RB, Carson RE (2003) Linearized reference tissue parametric imaging methods: application to [11C]DASB positron emission tomography studies of the serotonin transporter in human brain. J Cereb Blood Flow Metab 23:1096–1112
Koeppe RA, Holthoff VA, Frey KA, Kilbourn MR, Kuhl DE (1991) Compartmental analysis of [11C]flumazenil kinetics for the estimation of ligand transport rate and receptor distribution using positron emission tomography. J Cereb Blood Flow Metab 11:735–744
Lammertsma AA, Hume SP (1996) Simplified reference tissue model for PET receptor studies. Neuroimage 4:153–158
Lee MS, Rinne JO, Marsden CD (2000) The pedunculopontine nucleus: its role in the genesis of movement disorders. Yonsei Med J 41:167–184
Matsumoto N, Hanakawa T, Maki S, Graybiel AM, Kimura M (1999) Role of nigrostriatal dopamine system in learning to perform sequential motor tasks in a predictive manner. J Neurophysiol 82:978–998
Nagren K, Halldin C, Muller L, Swahn CG, Lehikoinen P (1995a) Comparison of [11C]methyl triflate and [11C]methyl iodide in the synthesis of PET radioligands such as [11C]beta-CIT and [11C]beta-CFT. Nucl Med Biol 22:965–979
Nagren K, Muller L, Halldin C, Swahn CG, Lehikoinen P (1995b) Improved synthesis of some commonly used PET radioligands by the use of [11C]methyl triflate. Nucl Med Biol 22:235–239
O’Sullivan JD, Said CM, Dillon LC, Hoffman M, Hughes AJ (1998) Gait analysis in patients with Parkinson’s disease and motor fluctuations: influence of levodopa and comparison with other measures of motor function. Movement Disorders 13:900–906
Ouchi Y, Kanno T, Okada H, Yoshikawa E, Futatsubashi M, Nobezawa S, Torizuka T, Tanaka K (2001) Changes in dopamine availability in the nigrostriatal and mesocortical dopaminergic systems by gait in Parkinson’s disease. Brain 124:784–792
Pahapill PA, Lozano AM (2000) The pedunculopontine nucleus and Parkinson’s disease. Brain 123(Pt 9):1767–1783
Schaafsma JD, Giladi N, Balash Y, Bartels AL, Gurevich T, Hausdorff JM (2003) Gait dynamics in Parkinson’s disease: relationship to Parkinsonian features, falls and response to levodopa. J Neurol Sci 212:47–53
Shan DE, Lee SJ, Chao LY, Yeh SI (2001) Gait analysis in advanced Parkinson’s disease-effect of levodopa and tolcapone. Can J Neurol Sci 28:70–75
Visser JE, Bloem BR (2005) Role of the basal ganglia in balance control. Neural Plast 12:161–174
Volkow ND, Gur RC, Wang GJ, Fowler JS, Moberg PJ, Ding YS, Hitzemann R, Smith G, Logan J (1998a) Association between decline in brain dopamine activity with age and cognitive and motor impairment in healthy individuals. Am J Psychiatry 155:344–349
Volkow ND, Wang GJ, Fowler JS, Ding YS, Gur RC, Gatley J, Logan J, Moberg PJ, Hitzemann R, Smith G, Pappas N (1998b) Parallel loss of presynaptic and postsynaptic dopamine markers in normal aging. Ann Neurol 44:143–147
Volkow ND, Logan J, Fowler JS, Wang GJ, Gur RC, Wong C, Felder C, Gatley SJ, Ding YS, Hitzemann R, Pappas N (2000) Association between age-related decline in brain dopamine activity and impairment in frontal and cingulate metabolism. Am J Psychiatry 157:75–80
Wilson RS, Schneider JA, Beckett LA, Evans DA, Bennett DA (2002) Progression of gait disorder and rigidity and risk of death in older persons. Neurology 58:1815–1819
Wiseman MB, Nichols TE, Woods RP, Sweeney JA, Mintun MA (1995) Stereotaxic techniques comparing foci intensity and location of activation areas in the brain as obtained using positron emission tomography (PET). J Nucl Med 36 (suppl):93P
Woods RP, Mazziotta JC, Cherry SR (1993) MRI-PET registration with automated algorithm. J Comput Assis Tomogr 17:536–546
Wu Y, Carson RE (2002) Noise reduction in the simplified reference tissue model for neuroreceptor functional imaging. J Cereb Blood Flow Metab 22:1440–1452
Acknowledgments
This study was supported by the Department of Veterans Affairs, Rehabilitation Research & Development Service, Grant E3270R. Also, partial support was provided by the Pittsburgh Claude D. Pepper Older Americans Independence Center (NIH/NIA P30 AG-024827).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cham, R., Studenski, S.A., Perera, S. et al. Striatal dopaminergic denervation and gait in healthy adults. Exp Brain Res 185, 391–398 (2008). https://doi.org/10.1007/s00221-007-1161-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-007-1161-3