Abstract
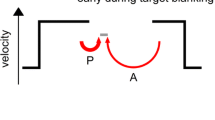
Background: Evidence supports an association between liability to schizophrenia and smooth-pursuit eye movement (SPEM) abnormalities. Knowledge of the biological mechanisms of SPEM abnormalities may provide critical insights into the etiology of schizophrenia. SPEM is elicited by sensory motor information from the movement of the object’s image on the retina (retinal motion signal) and subsequent extraretinal motion signals. Previous studies suggest that a deficit in predictive responses to extraretinal motion signals may underlie the SPEM phenotype in schizophrenia. Data suggest that at-risk individuals for schizophrenia depend less on extraretinal and more on retinal motion signals to maintain pursuit than healthy individuals. Methods: We designed a pursuit task that employs unexpected changes in target direction during smooth pursuit. The unpredictable task is unique in that performance is expected to be better if the subject’s response is biased towards retinal motion. Results: The study included 23 schizophrenia patients and 22 normal controls. Results showed that schizophrenia patients showed significantly superior performance (i.e. higher smooth pursuit gain) for a brief period after an unexpected change in target direction compared with healthy subjects. Conclusion: Findings of superior performance by schizophrenic patients are interesting because they circumvent confounds of generalized deficits. These results provide further evidence of specific deficit in the predictive pursuit mechanism and over-reliance on retinal error signals to maintain pursuit in schizophrenia.


Similar content being viewed by others
References
Avila MT, Adami HM, McMahon RP, Thaker GK (2003a) Using neurophysiological markers of genetic risk to define the boundaries of the schizophrenia spectrum phenotype. Schizophr Bull 29:299–309
Avila MT, Hong LE, Thaker GK (2003b) Evidence of predictive smooth-pursuit eye movement deficits in schizophrenia using a newly developed cued target paradigm. Schizophr Res 60 (Suppl):S265 (abstract)
Barnes GR, Asselman PT (1992) Pursuit of intermittently illuminated moving targets in the human. J Physiol 445:617–637
Barnes GR, Donelan SF (1999) The remembered pursuit task: evidence for segregation of timing and velocity storage in predictive oculomotor control. Exp Brain Res 129:57–67
Barnes GR, Schmid AM (2002) Sequence learning in human ocular smooth pursuit. Exp Brain Res 144:322–335
Barnes GR, Donnelly SF, Eason RD (1987) Predictive velocity estimation in the pursuit reflex response to pseudo-random and step displacement stimuli in man. J Physiol 389:111–136
Carpenter WT Jr, Gold JM, Lahti AC, Queern CA, Conley RR, Bartko JJ, Kovnick J, Appelbaum PS (2000) Decisional capacity for informed consent in schizophrenia research. Arch Gen Psychiatry 57:533–538
Chen Y, Nakayama K, Levy DL, Matthysse S, Holzman PS (1999) Psychophysical isolation of a motion-processing deficit in schizophrenics and their relatives and its association with impaired smooth pursuit. Proc Natl Acad Sci USA 96:4724–4729
Churchland MM, Lisberger SG (2001) Experimental and computational analysis of monkey smooth-pursuit eye movements. J Neurophysiol 86:741–759
Clementz BA (1996) Saccades to moving targets in schizophrenia: evidence for normal posterior cortex functioning. Psychophysiology 33:650–654
Clementz BA, McDowell JE (1994) Smooth pursuit in schizophrenia: abnormalities of open- and closed-loop responses. Psychophysiology 31(1):79–86
Clementz BA, Reid SA, McDowell JE, Cadenhead KS (1995) Abnormality of smooth-pursuit eye movement initiation: specificity to the schizophrenia spectrum? Psychophysiology 32:130–134
Ettinger U, Kumari V, Zachariah E, Galea A, Crawford TJ, Corr PJ, Taylor D, Das M, Sharma T (2003) Effects of procyclidine on eye movements in schizophrenia. Neuropsychopharmacology 28(12):2199–2208
First MB, Spitzer RL, Gibbon M, Williams JBW (1997) Structured clinical interview for DSM-IV axis I disorders: clinician version: administration booklet. American Psychiatric Press, Washington DC USA
Hedlund JL, Vieweg BW (1980) The brief psychiatric rating scale (BPRS) a comprehensive review. J Operational Psychiatry 11:48–64
Hong LE, Avila MT, Adami H, Elliott A, Thaker GK (2003) Components of the smooth pursuit function in deficit and nondeficit schizophrenia. Schizophr Res 63:39–48
Hong LE, Tagamets M, Avila MT, Wonodi I, Holcomb HH, Thaker GK (2005) Specific motion processing pathway deficit during eyetracking in schizophrenia: a performance-matched fMRI study. Biol Psychiatry 57:726–732
Hutton SB, Crawford TJ, Gibbins H, Cuthbert I, Barnes TR, Kennard C, Joyce EM (2001) Short and long term effects of antipsychotic medication on smooth pursuit eye tracking in schizophrenia. Psychopharmacology (Berl) 157:284–291
Kirkpatrick B, Buchanan RW, McKenney PD, Alphs LD, Carpenter WT (1989) The schedule for the deficit syndrome: an instrument for research in schizophrenia. Psychiatry Res 30:119–123
Lencer R, Nagel M, Sprenger A, Zapf S, Erdmann C, Heide W, Binkofski F (2004) Cortical mechanisms of smooth pursuit eye movements with target blanking. An fMRI study. Eur J Neurosci 195:1430–1436
Levy DL, Holzman PS, Matthysse S, Mendell NR (1993) Eye tracking dysfunction and schizophrenia: a critical perspective. Schizophr Bull 19:461–536
Levy DL, Lajonchere CM, Dorogusker B, Min D, Lee S, Tartaglini A, Lieberman JA, Mendell NR (2000) Quantitative characterization of eye tracking dysfunction in schizophrenia. Schizophr Res 42:171–185
Lisberger SG, Westbrook LE (1985) Properties of visual inputs that initiate horizontal smooth-pursuit eye movements in monkeys. J Neurosci 5:1662–1673
MacAvoy MG, Bruce CJ (1995) Comparison of the smooth eye tracking disorder of schizophrenics with that of nonhuman primates with specific brain lesions. Int J Neurosci 80:117–151
Stuve TA, Friedman L, Jesberger JA, Gilmore GC, Strauss ME, Meltzer HY (1997) The relationship between smooth pursuit performance, motion perception and sustained visual attention in patients with schizophrenia and normal controls. Psychol Med 27:143–152
Sweeney JA, Luna B, Srinivasagam NM, Keshavan MS, Schooler NR, Haas GL, Carl JR (1998) Eye tracking abnormalities in schizophrenia: evidence for dysfunction in the frontal eye fields. Biol Psychiatry 44:698–708
Thaker GK, Ross DE, Buchanan RW, Moran MJ, Lahti A, Kim C, Medoff D (1996) Does pursuit abnormality in schizophrenia represent a deficit in the predictive mechanism? Psychiatry Res 59:221–237
Thaker GK, Ross DE, Cassady SL, Adami HM, LaPorte D, Medoff DR, Lahti A (1998) smooth-pursuit eye movements to extraretinal motion signals: deficits in relatives of patients with schizophrenia. Arch Gen Psychiatry 55:830–836
Thaker GK, Ross DE, Buchanan RW, Adami HM, Medoff DR (1999) smooth-pursuit eye movements to extraretinal motion signals: deficits in patients with schizophrenia. Psychiatry Res 88:209–219
Thaker GK, Avila MT, Hong LE, Medoff DR, Ross DE, Adami HM (2003) A model of smooth-pursuit eye movement deficit associated with the schizophrenia phenotype. Psychophysiology 40:277–284
Trillenberg P, Lencer R, Heide W (2004) Eye movements and psychiatric disease. Curr Opin Neurol 17(1):43–47
Turano KA, Massof RW (2001) Nonlinear contribution of eye velocity to motion perception. Vis Res 41:385–395
Acknowledgements
Support was received from NIMH Grants MH67014, 68282, 68580, 49826, and 40279.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hong, L.E., Avila, M.T. & Thaker, G.K. Response to unexpected target changes during sustained visual tracking in schizophrenic patients. Exp Brain Res 165, 125–131 (2005). https://doi.org/10.1007/s00221-005-2276-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-005-2276-z