Abstract
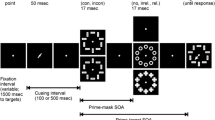
In a typical Simon task responses are faster when the task-irrelevant stimulus location corresponds to the response location than when it does not. In the case of noncorrespondence it is assumed that externally triggered and internally selected responses are in conflict. Crucially, such conflict appears to be subject to contextual modulations as induced by the immediately preceding event, i.e., the Simon effect was found to be absent when a conflict trial preceded the current event (Stürmer et al. 2002, JEP:HPP). Here, we examined two possible accounts of this context effect in terms of early suppression of externally triggered S-R coding at a premotoric level versus late suppression at a motoric level. Lateralized event-related brain potentials (L-ERPs) were recorded in a Simon task and analyzed as a function of the correspondence sequence. L-ERP activity started earliest over occipito-parietal brain areas and revealed location-based S-R priming irrespective of the prior correspondence context. By contrast, when a noncorresponding trial preceded, such location-based priming was absent in L-ERP activity over the motor cortex (MC). Thus, in support of the late suppression view L-ERPs suggest a clear dissociation in function between externally triggered visuomotor functions within the dorsal stream and the MC reflecting context-controlled response activation.




Similar content being viewed by others
Notes
It is worth mentioning that an overall reverse Simon effect had been observed with an incongruent mapping, suggesting that task-relevant S-R mapping rules might be applied to task-irrelevant S-R transformations as well (De Jong et al. 1994; Hedge and Marsh 1975; but see Simon et al. 1981). It is not yet completely clear, however, in which way changes in the S-R mapping condition exert modulatory effects on dual-route processing and whether there is a link to short-term contextual effects as induced by the correspondence sequence. The present study can therefore be considered a first attempt to examine this issue.
References
Andersen RA, Snyder LH, Bradley DC, Xing J (1997) Multimodal representation of space in the PPC and its use in planning movements. Annu Rev Neurosci 20:303–330
Bench CJ, Frith CD, Griaby PM, Friston KJ, Paulesu E, Frackowiak RSJ, Dolan RJ (1993) Investigations of the functional anatomy of attention using the Stroop test. Neuropsychologia 31:907–922
Berg P (1986) The residual after correcting event-related potentials for blink artefacts. Psychophysiology 23:354–364
Botvinick MM, Nystrom LE, Fissell K, Carter CS, Cohen JD (1999) Conflict monitoring versus selection-for-action in anterior cingulate cortex. Nature 402:179–181
Botvinick MM, Braver TS, Barch DM, Carter CS, Cohen JD (2001) Conflict monitoring and cognitive control. Psychol Rev 108 (3):624–652
Carter CS, Braver TS, Barch DM, Botvinick MM, Noll D, Cohen JD (1998) Anterior cingulate cortex, error detection, and the online monitoring of performance. Science 280:747–749
Carter CS, Macdonald AM, Botvinick MM, Ross LL, Stenger VA, Noll D, Cohen JD (2000) Parsing executive processes: strategic vs. evaluative functions of the anterior cingulate cortex. P Acad Nat Sci 97:1944–1948
Coles MGH (1989) Modern mind-brain reading: psychophysiology, physiology, and cognition. Psychophysiology 26:251–269
De Jong R, Liang CC, Lauber E (1994) Conditional and unconditional automaticity: a dual process model of effects of spatial stimulus-response correspondence. J Exp Psychol Hum Percept Performan 20:731–750
Dehaene S, Posner MI, Tucker DM (1994) Localization of a neural system for error detection and compensation. Psychol Sci 5:303–305
Di Russo F, Martinez A, Sereno MI, Pitzalis S, Hillyard SA (2002) Cortical sources of the early components of the visual evoked potential. Hum Brain Mapp 15(2):95–111
Duhamel JR, Colby C, Goldberg ME (1992) The updating of the representation of visual space in parietal cortex by intended eye movements. Science 255:90–92
Eimer M (1996) The N2pc component as an indicator of attentional selectivity. Electroen Clin Neuro 99:225–234
Foxe JJ, Simpson GV (2002) Flow of activation from V1 to frontal cortex in humans: a framework for defining "early" visual processing. Exp Brain Res 142:139–150
Garavan H, Ross TJ, Stein EA (1999) Right hemispheric dominance of inhibitory control: an event-related functional MRI study. Proc Natl Acad Sci 96:8301–8306
Gehring WJ, Knight RT (2002) Lateral prefrontal damage affects processing selection but not attention switching. Brain Res Cogn Brain Res 13:267–279
Hedge A, Marsh NWA (1975) The effect of irrelevant spatial correspondence on two-choice response-time. Acta Psychol 39:427–439
Hopf JM, Luck SJ, Girelli M, Hagner T, Mangun GR, Scheich H, Heinze HJ (2000) Neural sources of focused attention in visual search. Cereb Cortex 10:1233-1241
Huynh H, Feldt LS (1976) Estimation of the Box correction for degrees of freedom from sample data in the randomized block and split-plot designs. J Educational Statistics 1:69–82
Iacoboni M, Woods RP, Mazziotta JC (1996) Brain-behavior relationships: evidence from practice effects in spatial stimulus-response compatibility. J Neurophysiol 76:321–331
Iacoboni M, Woods RP, Mazziotta JC (1998) Bimodal (auditory and visual) left frontoparietal circuitry for sensorimotor integration and sensorimotor learning. Brain 121:2135–2143
Kleinsorge T, Heuer H (1999) Hierarchical switching in a multi-dimensional task space. Psychol Res 62 (4):300–312
Kleinsorge T, Heuer H, Schmidtke V (2002) Process of task-set reconfiguration: switching operations and implementation operations. Acta Psychol 111 (1):1–28
Konishi S, Nakajima K, Uchida I, Kikyo H, Kameyama M, Miyashita Y (1999) Common inhibitory mechanism in human inferior prefrontal cortex revealed by event-related functional MRI. Brain 122:981–999
Kornblum S, Hasbroucq T, Osman A (1990) Dimensional overlap: cognitive basis for stimulus-response compatibility. In: Stelmach GE, Requin J (eds) Tutorial in motor behavior 87. North-Holland, Amsterdam pp743–777
Leuthold H, Jentzsch I (2001) Neural correlates of advance movement preparation: a dipole source analysis approach. Cogn Brain Res 12, 207–224
Leuthold H, Jentzsch I (2002) Distinguishing neural sources of movement preparation and execution: an electrophysiological analysis. Biol Psychol 60:173–198
Lu CH, Proctor RW (1995) The influence of irrelevant location information on performance: a review of the Simon and spatial Stroop effect. Psychon B Rev 2:174–207
Luck SJ, Hillyard SA (1994a) Electrophysiological correlates of feature analysis during visual search. Psychophysiology 31:291–308
Luck SJ, Hillyard SA (1994b) Spatial filtering during visual search: evidence from human electrophysiology. J Exp Psychol Hum Percept Performan 20 (5):1000–1014
Mattingley JB, Husian M, Rorden C, Kennard C, Driver J (1998) Motor role of human inferior parietal lobe revealed in unilateral neglect patients. Nature 392:179–182
Milham MP, Banich MT, Webb A, Barad V, Cohen NJ, Wszalek T, Kramer AF (2001) The relative involvement of anterior cingulate and prefrontal cortex in attentional control depends on nature of conflict. Brain Res Cogn Brain Res 12:467–473
Miller J, Riehle A, Requin J (1992) Effects of preliminary perceptual output on neuronal activity of the primary motor cortex. J Exp Psychol Hum Percept Performan 18:1121–1138
Milner DA, Goodale MA (1995) The visual brain in action. Oxford University Press, Oxford, NY
Mordkoff T (1998) The gating of irrelevant information in selective-attention tasks. Abstracts of the Psychonomic Society 3:193
Oostenveld R, Praamstra P, Stegeman D F, van Oosterom A (2001) Overlap of attention and movement-related activity in lateralized event-related brain potentials. Clin Neurophys 112:477–484
Pardo JV, Pardo PJ, Janer KW, Raichle ME (1990) The anterior cingulate cortex mediates processing selection in the Stroop attentional conflict paradigm. P Acad Nat Sci 87:256–259
Petersen BS, Kane MJ, Alexander GM, Lacadie C, Skudlarski P, Leung HC, May J, Gore JC (2002) An event-related functional MRI study comparing interference effects in the Simon and Stroop tasks. Brain Res Cogn Brain Res 13:427–440
Pivik RT, Broughton RJ, Coppola R, Davidson RJ, Fox N, Nuwer NR (1993) Guidelines for the recording and quantitative analysis of electroencephalographic activity in research contexts. Psychophysiology 30:547–558
Posner MI, DiGirolamo GJ (1998) Executive attention: conflict, target detection, and cognitive control. In: Parasuraman R (ed) The attentive brain. The Mit Press, Cambridge, MA pp 401–423
Praamstra P, Oostenveld R (2003) Attention and movement-related motor cortex activation: a high-density EEG study of spatial stimulus-response compatibility. Cogn Brain Res 16:309–322
Praamstra P, Stegemann DF, Horstink MWIM, Cools AR (1996) Dipole source analysis suggests selective modulation of the supplementary motor area contribution to the readiness potential. Electroenceph Clin Neurophysiol 98:468–477
Praamstra P, Kleine BU, Schnitzler A (1999a) Magnetic stimulation of the dorsal premotor cortex modulates the Simon effect. Neuro Report 10:3671–3674
Praamstra P, Schmitz F, Freund HJ, Schitzler A (1999b) Magneto-encephalographic correlates of the lateralized readiness potential. Cogn Brain Res 8:77–85
Ro T, Cohen A, Ivry RB, Rafal RD (1998) Response channel activation and the temporoparietal junction. Brain Cognition 37:461–476
Schubert T, Crammon DY, Niendorf T, Pollmann S, Bublak P (1998) Cortical areas and the control of self-determined finger movements: an fMRI study. NeuroReport 9:3171–3176
Simon JR, Sly PE, Vilapakkam S (1981) Effect of compatibility of S-R mapping on reactions toward the stimulus source. Acta Psychol 47(1):63–81
Sirigu A, Duhamel JR, Cohen L, Pillon B, Dubois B, Agid Y (1996) The mental representation of hand movements after parietal cortex damage. Science 273:1564–1568
Snyder LH, Batista AP, Andersen RA (2000) Intention-related activity in the posterior parietal cortex: a review. Vision Res 40:1433–1441
Stoffels EJ (1996a) On stage robustness and response selection: further evidence. Acta Psychol 91:67–88
Stoffels EJ (1996b) Uncertainty and processing routes in the selection of a response: an S-R compatibility study. Acta Psychol 94:227–252
Stürmer B, Siggelkow S, Dengler R, Leuthold H (2000) Response priming in the Simon paradigm: a transcranial magnetic stimulation study. Exp Brain Res 135:353–359
Stürmer B, Leuthold H, Soetens E, Schröter H, Sommer W (2002) Control over location-based priming in the Simon task: behavioral and electrophysiological evidence. J Exp Psychol Hum Percept Performan 28 (6):1345–1363
Taylor SF, Kornblum S, Minoshima S, Oliver LM, Koeppe RA (1994) Changes in medial cortical blood flow with a stimulus-response compatibility task. Neuropsychologia 32:249–255
Ungerleider LG, Courtney SM, Haxby JV (1998) A neural system for human visual working memory. P Acad Nat Sci 95:883–890
Valle-Inclán F, Hackley S, de Labra C (2002) Attention and response activation in the Simon task In: Prinz W, Hommel B (eds) Attention & Performance XIX. Oxford University Press, pp 474–493
Wascher E, Wauschkuhn B (1996) The interaction of stimulus- and response-related processes measured by event-related lateralizations of the EEG. Electroen Clin Neuro 99:149–162
Wascher E, Schatz U, Kuder T, Verleger R (2001) Validity and boundary conditions of automatic response activation in the Simon Task. J Exp Psychol Hum Percept Performan 27(3):731–751
Woldorff MG, Liotti M, Seabolt M, Busse L, Lancaster JL, Fox PT (2002) The temporal dynamics of the effects in occipital cortex of visual-spatial selective attention. Cog Brain Res 15:1–15
Acknowledgements
This research was supported by a grant of the Deutsche Forschungsgemeinschaft to Hartmut Leuthold and Werner Sommer (LE 1035/1–2). We appreciate the help of Irina Langenbruch and Ellen Seiß in data acquisition. We thank Thomas Goschke, Werner Sommer, and Rolf Ulrich for helpful comments on an earlier version of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Source models were determined by using Brain Electromagnetic Source Analysis (BESA2000). Symmetry constraints with respect to location and orientation were applied to lateral dipole-pairs in order to limit the number of parameters to be estimated. Source models for L-ERP activity were derived as described in more detail in a study of Praamstra et al. (1996; see also Leuthold and Jentzsch 2001). L-ERPs were projected to one hemisphere and copied to the other hemisphere with polarity inverted. This procedure resulted in an anti-symmetric scalp distribution of L-ERP activity (cf. Praamstra et al. 1996).
First, we fitted separate source models for the initial parieto-temporal L-ERP activity of grand-average (across subjects) stimulus-locked waveforms of corresponding and noncorresponding conditions. That is, one dipole pair was fitted in an interval between 150–200 ms relative to stimulus onset, when initial L-ERP activity was fully developed. For both conditions the resulting dipole model was located in occipito-parietal regions and accounted for more than 97% of the variance in this 50-ms time interval. Next, we derived the dipole model for the movement-related lateralization in response-locked grand-averages. To this end, an additional dipole was added to the earlier model, which was fitted in a time interval from −200 to −50 ms prior to response onset (cf. Leuthold and Jentzsch 2001). The resulting two-dipole model accounted for 98.8% of the variance in this 150-ms time interval with Dipole 2 taken to reflect MC activity (cf. Leuthold and Jentzsch 2002). We applied this model to original stimulus-locked L-ERP data from noncorresponding conditions preceded by either a corresponding or noncorresponding trialN-1; the model accounted for more than 95% of the data variance between 150–350 ms after stimulus onset. It was clearly apparent that activity within the occipito-parietal source (Dipole 1) resembled initial L-ERP activity starting about 125 ms after stimulus onset. Importantly, the MC source perfectly reflected later location-based MC activation, as indicated by the dip into the incorrect direction peaking at around 240 ms, as well as later activity related to the execution of the correct response (see Fig. 4).
We also conducted forward simulations where location of Dipole 1 was systematically varied and the resulting L-ERP topographies were compared with the original L-ERP distributions. For example, we positioned dipoles in the occipito-temporal region like Oostenveld et al. (2001) in their forward simulation of N2pc sources. Critically, these additional forward solutions revealed simulated L-ERP topographies that did produce as good an explanation (in terms of residual variance) of original L-ERP data as the above source model. Dipoles located in the occipito-temporal lobe, i.e., the ventral processing stream (cf. Milner and Goodale 1995), produced asymmetric sensory activity over lateral motor cortices of about 20–30% of the maximal amplitude measured over occipito-parietal sites. This particular finding agrees with earlier reports in the simulation study of Oostenveld et al. (2001). Most importantly, however, the present L-ERP data revealed activation recorded over occipito-parietal sites that did not propagate to MC electrodes.
Of course, conclusions from present source localization results about the neural origin of the initial L-ERP waveform over occipito-parietal recording sites should be taken tentatively until high-density L-ERP studies are available. Such studies will help to provide a clearer picture about the topography of occipito-parietal L-ERP waveforms, thereby also allowing an improved estimation of the underlying neural sources.
Rights and permissions
About this article
Cite this article
Stürmer, B., Leuthold, H. Control over response priming in visuomotor processing: a lateralized event-related potential study. Exp Brain Res 153, 35–44 (2003). https://doi.org/10.1007/s00221-003-1579-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-003-1579-1