Abstract
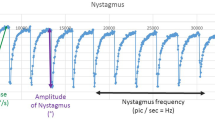
To investigate whether the primary planes of eye and body responses to galvanic vestibular stimulation (GVS) are congruent, we have measured the binocular, three-dimensional eye movements (scleral coil technique) to bilateral bipolar GVS in six normal human subjects. Stimulation intensities were kept deliberately low in order to characterize the response to near-threshold intensities of stimulation (0.1–0.9 mA) that had been used previously to characterise body postural responses. Stimuli were applied for 4 s, but only the early responses that occurred within the initial 300 ms of turning the current on or off were measured. At intensities of 0.1–0.7 mA the 'on' response consisted almost exclusively of a torsional slow phase eye movement in which the top of the eyes rotated towards the anode. The latency of the torsional response was ca. 46 ms. A weak polarity-dependent disconjugate response was also observed in which the intorting eye elevated and the extorting eye depressed ('skew eye deviation'). When the current was turned off similar responses occurred in the reverse direction. Removal of the visual fixation light-emitting diode (LED) had no consistent effect on the short-latency ocular responses. The direction of the ocular response was similar to that of the postural response and is compatible with GVS inducing an apparent dynamic roll-tilt of the head towards the cathode. However, weak horizontal eye movements, which became more prominent as the stimulus intensity was increased to 0.9 mA, were also observed. This suggests that an additional weak rotational component about the yaw axis, or a component of lateral translation in the frontal plane, is contained in the GVS-evoked signal. The overall pattern of eye movement suggests that semicircular canal afferents contribute to these low-intensity GVS responses.



Similar content being viewed by others
References
Breson K, Elberling C, Fangel J (1971) Galvanic nystagmography. Acta Otolaryngol 71:449–455
Britton TC, Day BL, Brown P, Rothwell JC, Thompson PD, Marsden CD (1993) Postural electromyographic responses in the arm and leg following galvanic vestibular stimulation in man. Exp Brain Res 94:143–151
Cohen B, Suzuki J–I, Bender MB (1964) Eye movements from semicircular canal nerve stimulation in the cat. Ann Otol Rhinol Laryngol 73:153–169
Courjon JH, Precht W, Sirkin DW (1987) Vestibular nerve and nuclei unit responses and eye movement responses to repetitive galvanic stimulation of the labyrinth in the rat. Exp Brain Res 66:41–48
Day BL, Cole J (2002) Vestibular-evoked postural responses in the absence of somatosensory information. Brain 125:2081–2088
Day BL, Severac Cauquil A, Bartolomei L, Pastor MA, Lyon IN (1997) Human body-segment tilts induced by galvanic stimulation: a vestibularly driven balance protection mechanism. J Physiol 500:661–672
Goldberg JM, Smith CE, Fernandez C (1984) Relation between discharge regularity and responses to externally applied galvanic currents in vestibular nerve afferents of the squirrel monkey. J Neurophysiol 51:1236–1256
Jáuregui-Renaud K, Faldon M, Clarke A, Bronstein AM, Gresty MA (1996) Skew deviation of the eyes in normal human subjects induced by semicircular canal stimulation. Neurosci Lett 205:135–137
Jáuregui-Renaud K, Bronstein A, Faldon M, Gresty M (1997) Binocular response to roll-motion in man. J Physiol 505.P:78P–79P
Jáuregui-Renaud K, Faldon ME, Gresty MA, Bronstein AM (2001) Horizontal ocular vergence and the three-dimensional response to whole-body roll motion. Exp Brain Res 136:79–92
Karlberg M, McGarvie L, Magnusson M, Aw ST, Halmagyi GM (2000) The effects of galvanic stimulation on the human vestibulo-ocular reflex. Neuroreport 11:3897–3901
Kleine JF, Guldin WO, Clarke AH (1999) Variable otolith contribution to the galvanically induced vestibulo-ocular reflex. Neuroreport 10:1143–1148
Lowenstein O (1955) The effect of galvanic polarization on the impulse discharge from sense endings in the isolated labyrinth of the thornback ray (Raja clavata). J Physiol 127:104–117
Lund S, Broberg C (1983) Effects of different head positions on postural sway in man induced by a reproducible vestibular error signal. Acta Physiol Scand 117:307–309
Pfaltz CR (1970) The diagnostic importance of nystagmography in the galvanic test. In: Stahle J (ed) Vestibular function on earth and in space. Pergamon, Oxford, pp 187–199
Quarck G, Etard O, Normand H, Pottier M, Denise P (1998) Low intensity galvanic vestibulo-ocular reflex in normal subjects. Clin Neurophysiol 28:413–422
Schneider E, Glasauer S, Dieterich M (2000) Central processing of human ocular torsion analyzed by galvanic vestibular stimulation. Neuroreport 11:1559–1563
Schneider E, Glasauer S, Dieterich M (2002) Comparison of human ocular torsion patterns during natural and galvanic vestibular stimulation. J Neurophysiol 87:2064–2073
Severac Cauquil A, Faldon M, Popov K, Bronstein A, Day BL (1998) Torsional eye movements induced by galvanic vestibular stimulation in man. J Physiol 506.P:110P
Suzuki J–I, Tokumasu K, Goto K (1969) Eye movements from single utricular nerve stimulation in the cat. Acta Otolaryngol 68:350–362
Watson SRD, Brizuela AE, Curthoys IS, Curthoys IS, Colebatch JG, Macdougall HG, Halmagyi GM (1998) Maintained ocular torsion produced by bilateral and unilateral galvanic (DC) vestibular stimulation in humans. Exp Brain Res 122:453–458
Zink R, Steddin S, Weiss A, Brandt T, Dieterich M (1997) Galvanic vestibular stimulation in humans: effects on otolith function in roll. Neurosci Lett 232:171–174
Zink R, Bucher SF, Weiss A, Brandt Th, Dieterich M (1998) Effects of galvanic vestibular stimulation on otolithic and semicircular canal eye movements and perceived vertical. Electroencephalogr Clin Neurophysiol 107:200–205
Acknowledgements.
Support from the Dix Foundation and the CEC grant 'Transnational Access to Research Infrastructures' is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Séverac Cauquil, A., Faldon, M., Popov, K. et al. Short-latency eye movements evoked by near-threshold galvanic vestibular stimulation. Exp Brain Res 148, 414–418 (2003). https://doi.org/10.1007/s00221-002-1326-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-002-1326-z