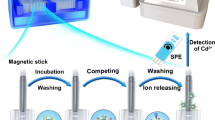
Abstract
Detection methods with high sensitivity and short assay time are urgently required for quantitative analysis of small-molecule hazardous substances in food monitoring. In this work, a new anti-aflatoxin B1 (AFB1) nanobody was screened from an immunized nanobody library, and an ultrafast one-step detection of AFB1 without immobilization and multi-step washing was developed based on magnetic separation technology and nanobody (Nb)-alkaline phosphatase (ALP) fusion protein. Compared to conventional one-step chemiluminescent enzyme-linked immunosorbent assay (CLEIA) based on Nb-ALP, it was surprising to find the sensitivity and lowest limit of detection (LOD) of this method was significantly improved about threefold and fivefold separately, and the total assay time could be reduced to 30 from 120 min. Under optimal conditions, the developed method achieved the sensitive detection of AFB1 with LOD with 0.743 pg mL−1, IC50 = 0.33 ng mL−1, the linear range was 7.23 pg mL−1 ~ 12.38 ng mL−1, and showed powerful tolerance and utility for complex matrix environments in sample detection. It is believed this method could provide a newly way for the quick and sensitive detection of AFB1 and could expand the application of Nbs.







Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings of this study are available within the article.
References
Caceres I, Snini SP, Puel O, Mathieu F (2018) Streptomyces roseolus, a promising biocontrol agent against Aspergillus flavus, the main aflatoxin B1 producer. Toxins 10:442
Battilani P, Toscano P, Van der Fels-Klerx HJ, Moretti A, Camardo Leggieri M, Brera C, Rortais A, Goumperis T, Robinson T (2016) Aflatoxin B1 contamination in maize in Europe increases due to climate change. Sci Rep 6:24328
Streit E, Schatzmayr G, Tassis P, Tzika E, Marin D, Taranu I, Tabuc C, Nicolau A, Aprodu I, Puel O, Oswald IP (2012) Current situation of mycotoxin contamination and co-occurrence in animal feed–focus on Europe. Toxins (Basel) 4:788–809
Cimbalo A, Alonso-Garrido M, Font G, Manyes L (2020) Toxicity of mycotoxins in vivo on vertebrate organisms: a review. Food Chem Toxicol 137:111161
Mahato DK, Lee KE, Kamle M, Devi S, Dewangan KN, Kumar P, Kang SG (2019) Aflatoxins in food and feed: an overview on prevalence detection and control strategies. Front Microbiol 10:2266
Abrar M, Anjum FM, Butt MS, Pasha I, Randhawa MA, Saeed F, Waqas K (2013) Aflatoxins: biosynthesis, occurrence, toxicity, and remedies. Crit Rev Food Sci Nutr 53:862–874
Khayoon WS, Saad B, Lee TP, Salleh B (2012) High performance liquid chromatographic determination of aflatoxins in chilli, peanut and rice using silica based monolithic column. Food Chem 133:489–496
Romera D, Mateo EM, Mateo-Castro R, Gómez JV, Gimeno-Adelantado JV, Jiménez M (2018) Determination of multiple mycotoxins in feedstuffs by combined use of UPLC-MS/MS and UPLC-QTOF-MS. Food Chem 267:140–148
Chen F, Luan C, Wang L, Wang S, Shao L (2017) Simultaneous determination of six mycotoxins in peanut by high-performance liquid chromatography with a fluorescence detector. J Sci Food Agric 97:1805–1810
Wang X, Niessner R, Tang D, Knopp D (2016) Nanoparticle-based immunosensors and immunoassays for aflatoxins. Anal Chim Acta 912:10–23
Zhang D, Li P, Zhang Q, Zhang W (2011) Ultrasensitive nanogold probe-based immunochromatographic assay for simultaneous detection of total aflatoxins in peanuts. Biosens Bioelectron 26:2877–2882
Gazzaz SS, Rasco BA, Dong FM (1992) Application of immunochemical assays to food analysis. Crit Rev Food Sci Nutr 32:197–229
Du J, Chen X, Liu K, Zhao D, Bai Y (2022) Dual recognition and highly sensitive detection of Listeria monocytogenes in food by fluorescence enhancement effect based on Fe3O4@ZIF-8-aptamer. Sens Actuators B 360:131654
Zhang W, Serpe MJ (2017) Antigen detection using fluorophore-modified antibodies and magnetic microparticles. Sens Actuators B 238:441–446
Yue Q, Li X, Fang J, Li M, Zhang J, Zhao G, Cao W, Wei Q (2022) Oxygen Free Radical Scavenger PtPd@PDA as a dual-mode quencher of electrochemiluminescence immunosensor for the detection of AFB1. Anal Chem 94:11476–11482
Gonzalez-Sapienza G, Rossotti MA, Tabares-da Rosa S (2017) Single-domain antibodies as versatile affinity reagents for analytical and diagnostic applications. Front Immunol. https://doi.org/10.3389/fimmu.2017.00977
Shu M, Xu Y, Wang D, Liu X, Li Y, He Q, Tu Z, Qiu Y, Ji Y, Wang X (2015) Anti-idiotypic nanobody: a strategy for development of sensitive and green immunoassay for Fumonisin B1. Talanta 143:388–393
Tang Z, Wang X, Lv J, Hu X, Liu X (2018) One-step detection of ochratoxin A in cereal by dot immunoassay using a nanobody-alkaline phosphatase fusion protein. Food Control 92:430–436
Ren W, Li Z, Xu Y, Wan D, Barnych B, Li Y, Tu Z, He Q, Fu J, Hammock BD (2019) One-step ultrasensitive bioluminescent enzyme immunoassay based on nanobody/nanoluciferase fusion for detection of aflatoxin B1 in cereal. J Agric Food Chem 67:5221–5229
Dunlop EH, Feiler WA, Mattione MJ (1984) Magnetic separation in biotechnology. Biotechnol Adv 2:63–74
Wang F, Li ZF, Yang YY, Wan DB, Vasylieva N, Zhang YQ, Cai J, Wang H, Shen YD, Xu ZL, Hammock BD (2020) Chemiluminescent enzyme immunoassay and bioluminescent enzyme immunoassay for tenuazonic acid mycotoxin by exploitation of nanobody and nanobody-nanoluciferase fusion. Anal Chem 92:11935–11942
Di S, Ning T, Yu J, Chen P, Yu H, Wang J, Yang H, Zhu S (2020) Recent advances and applications of magnetic nanomaterials in environmental sample analysis. TrAC Trends Anal Chem 126:115864
Zhao F, Shi R, Liu R, Tian Y, Yang Z (2021) Application of phage-display developed antibody and antigen substitutes in immunoassays for small molecule contaminants analysis: a mini-review. Food Chem 339:128084
Sun T, Zhao Z, Liu W, Xu Z, He H, Ning B, Jiang Y, Gao Z (2020) Development of sandwich chemiluminescent immunoassay based on an anti-staphylococcal enterotoxin B nanobody-alkaline phosphatase fusion protein for detection of staphylococcal enterotoxin B. Anal Chim Acta 1108:28–36
Zhang YY, Li LH, Wang Y, Wang H, Xu ZL, Tian YX, Sun YM, Yang JY, Shen YD (2022) Ultrasensitive and rapid colorimetric detection of paraquat via a high specific VHH nanobody. Biosens Bioelectron 205:114089
Güttler T, Aksu M, Dickmanns A, Stegmann KM, Gregor K, Rees R, Taxer W, Rymarenko O, Schünemann J, Dienemann C, Gunkel P, Mussil B, Krull J, Teichmann U, Groß U, Cordes VC, Dobbelstein M, Görlich D (2021) Neutralization of SARS-CoV-2 by highly potent, hyperthermostable, and mutation-tolerant nanobodies. Embo j 40:e107985
Muyldermans S (2013) Nanobodies: natural single-domain antibodies. Annu Rev Biochem 82:775–797
Mateo C, Grazu V, Palomo JM, Lopez-Gallego F, Fernandez-Lafuente R, Guisan JM (2007) Immobilization of enzymes on heterofunctional epoxy supports. Nat Protoc 2:1022–1033
Hehir MJ, Murphy JE, Kantrowitz ER (2000) Characterization of heterodimeric alkaline phosphatases from Escherichia coli: an investigation of intragenic complementation. J Mol Biol 304:645–656
Sowadski JM, Handschumacher MD, Murthy HM, Foster BA, Wyckoff HW (1985) Refined structure of alkaline phosphatase from Escherichia coli at 2.8 A resolution. J Mol Biol 186:417–433
Kunz P, Zinner K, Mücke N, Bartoschik T, Muyldermans S, Hoheisel JD (2018) The structural basis of nanobody unfolding reversibility and thermoresistance. Sci Rep 8:7934
Le Basle Y, Chennell P, Tokhadze N, Astier A, Sautou V (2020) Physicochemical stability of monoclonal antibodies: a review. J Pharm Sci 109:169–190
Goldman ER, Liu JL, Zabetakis D, Anderson GP (2017) Enhancing stability of camelid and shark single domain antibodies: an overview. Front Immunol 8:865
Tang X, Li P, Zhang Q, Zhang Z, Zhang W, Jiang J (2017) Time-resolved fluorescence immunochromatographic assay developed using two idiotypic nanobodies for rapid, quantitative, and simultaneous detection of aflatoxin and zearalenone in maize and its products. Anal Chem 89:11520–11528
Yan T, Zhu J, Li Y, He T, Yang Y, Liu M (2022) Development of a biotinylated nanobody for sensitive detection of aflatoxin B(1) in cereal via ELISA. Talanta 239:123125
He T, Wang Y, Li P, Zhang Q, Lei J, Zhang Z, Ding X, Zhou H, Zhang W (2014) Nanobody-based enzyme immunoassay for aflatoxin in agro-products with high tolerance to cosolvent methanol. Anal Chem 86:8873–8880
Zhang X, Liao X, Wu Y, Xiong W, Du J, Tu Z, Yang W, Wang D (2022) A sensitive electrochemical immunosensing interface for label-free detection of aflatoxin B(1) by attachment of nanobody to MWCNTs-COOH@black phosphorene. Anal Bioanal Chem 414:1129–1139
Liu X, Wen Y, Wang W, Zhao Z, Han Y, Tang K, Wang D (2020) Nanobody-based electrochemical competitive immunosensor for the detection of AFB(1) through AFB(1)-HCR as signal amplifier. Mikrochim Acta 187:352
Xu W, Xiong Y, Lai W, Xu Y, Li C, Xie M (2014) A homogeneous immunosensor for AFB1 detection based on FRET between different-sized quantum dots. Biosens Bioelectron 56:144–150
Li J, Zhao X, Chen LJ, Qian HL, Wang WL, Yang C, Yan XP (2019) p-Bromophenol-enhanced bienzymatic chemiluminescence competitive immunoassay for ultrasensitive determination of aflatoxin B(1). Anal Chem 91:13191–13197
Acknowledgements
This research has received funding support from (20SWAQK16, 2019GGRC03, 2019CXTD03), Heilongjiang Province Key Laboratory of Microecology-Immune Regulation Network and Related Diseases (2021-SZD-JC-005), 2021 Heilongjiang Provincial Health and Wellness Committee Project (20210202040058)
Author information
Authors and Affiliations
Contributions
XW: conceptualization, writing—original draft, methodology, and writing—review and editing. WL: methodology, investigation, and software. HZ: investigation and methodology. WS: investigation and methodology. YZ: methodology and software. RL: methodology and validation. LG: investigation and validation. WW: investigation and validation. CS: writing—review and editing, supervision, and methodology. TS: investigation, writing—review and editing, validation, and funding acquisition.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
The research does not involve human participants.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
217_2023_4202_MOESM1_ESM.docx
Supplementary file1 Supplementary data associated with this article can be found in the Appendix A. Supporting information (DOCX 2165 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, X., Liu, W., Zuo, H. et al. Development of a magnetic separation immunoassay with high sensitivity and time-saving for detecting aflatoxin B1 in agricultural crops using nanobody. Eur Food Res Technol 249, 1125–1136 (2023). https://doi.org/10.1007/s00217-023-04202-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-023-04202-3