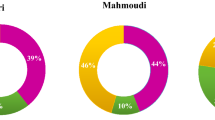
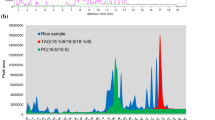
Abstract
Given the lack of information about the lipid profile of local durum wheat landraces, and their presumed high nutritional value, gas chromatography coupled to a time-of-flight mass analyzer was used to investigate the lipid profiles of three of the most important local durum wheat landraces. The obtained results indicated the nutritional adequacy and quality of Mahmoudi’s and Jnah khottifa’s whole grain wheat flour. Analysis of the biosynthetic pathway highlighted that the linoleic acid metabolism and the fatty acid biosynthesis pathway are impactful metabolic pathways in discriminating between landraces. β-Sitosterol and 2-linoleoylglycerol were identified as hub metabolites. Based on this, GC–TOF-MS™-based lipid profiling could be used as an authenticity tool to differentiate local whole grain flours or guide the selection of specific marker compounds to set proper quality standards for better restoration and in situ and ex situ conservation of the subsequent use of these valuable germ.





Similar content being viewed by others
References
Deghaïs M, Kouki M, Gharbi MS, El Felah M (2007) Les Variétés de Céréales Cultivées en Tunisie (blé dur, blé Tendre, orge et Triticale); INRAT: Tunis, Tunisia. (In French).
Conversa G, Lazzizera C, Bonasia A, Cifarelli S, Losavio F, Sonnante G, Elia A (2020) Exploring on-farm agro-biodiversity: a study case of vegetable landraces from Puglia region (Italy). Biodivers Conserv 29:747–770
FAO Roles and importance of biodiversity for food and agriculture (2019) In: The state of the world’s biodiversity for food and agriculture; Bélanger J, Pilling D (Eds) FAO commission on genetic resources for food and agriculture assessments: Rome, Italy, pp. 17–62
Spisni E, Imbesi V, Giovanardi E, Petrocelli G, Alvisi P, Valerii MC (2019) Differential physiological responses elicited by ancient and heritage wheat cultivars compared to modern ones. Nutrients 11:2879
Ouaja M, Bahri BA, Aouini L et al (2021) Morphological characterization and genetic diversity analysis of Tunisian durum wheat (Triticum turgidum var. durum) accessions. BMC Genom Data 22:3
Ayed S, Karmous C, Trifa Y, Slama AO, Amara HS (2010) Phenotypic diversity of Tunisian durum wheat landraces. Afr Crop Sci J 18:35–42
Boukid F, Dall’Asta M, Bresciani L et al (2019) Phenolic profile and antioxidant capacity of landraces, old and modern Tunisian durum wheat. Eur Food Res Technol 245:73–82
Chamekh Z, Karmous C, Ayadi S, Sahli A, Hammami Z, Fraj MB, Benaissa N, Trifa Y, Slim-Amara H (2015) Stability analysis of yield component traits in 25 durum wheat (Triticum durum Desf.) genotypes under contrasting irrigation water salinity. Agric Water Manag 152:1–6
Robbana C, Kehel Z, Ammar K, Guzmán C, Naceur MB, Amri A (2021) Unlocking the patterns of the Tunisian durum wheat landraces genetic structure based on phenotypic characterization in relation to farmer’s vernacular name. Agronomy 11:634
Saia S, Fragasso M, De Vita P, Beleggia R (2019) Metabolomics provides valuable insight for the study of durum wheat: a review. J Agric Food Chem 67:3069–3085
Han L, Lin Q, Liu G, Han D, Niu L, Su D (2019) Lipids promote glycated phospholipid formation by inducing hydroxyl radicals in a maillard reaction model system. J Agric Food Chem 67(28):7961–7967
Hammann S, Korf A, Bull ID, Hayen H, Cramp L (2019) Lipid profiling and analytical discrimination of seven cereals using high temperature gas chromatography coupled to high resolution quadrupole time-of-flight mass spectrometry. Food Chem 282:27–35
Boukid F, Folloni S, Ranieri R, Vittadini E (2018) A compendium of wheat germ: separation, stabilization and food applications. Trends Food Sci Technol 78:120–133
Ross AB, Shepherd MJ, Schüpphaus M, Sinclair V, Alfaro B, Kamal-Eldin A, Åman P (2003) Alkylresorcinols in cereals and cereal products. J Agric Food Chem 51(14):4111–4118
Prinsen P, Gutierrez A, Faulds CB, del Rio JC (2014) Comprehensive study of valuable lipophilic phytochemicals in wheat bran. J Agric Food Chem 62(7):1664–1673
Cert A, Moreda W, Pérez-Camino M (2000) Chromatographic analysis of minor constituents in vegetable oils. J Chromatogr A 881(1–2):131–148
Rupérez FJ, Martı́n D, Herrera E, Barbas C (2001) Chromatographic analysis of α-tocopherol and related compounds in various matrices. J Chromatogr A 935(1):45–69
Janssen H-G, de Koning S, Brinkman UA (2004) On-line LC-GC and comprehensive two-dimensional LCxGC-ToF MS for the analysis of complex samples. Anal Bioanal Chem 378(8):1944–1947
Park SY, Lim SH, Ha SH, Yeo Y, Park WT, Kwon DY, Park SU, Kim JK (2013) Metabolite profiling approach reveals the interface of primary and secondary metabolism in colored cauliflowers (Brassica oleracea L. ssp botrytis). J Agric Food Chem 61(28):6999–7007
Boswell KM, D’Elia M, Johnston MW, Mohan JA, Warren JD, Wells RJ et al (2020) Oceanographic structure and light levels drive patterns of sound scattering layers in a low-latitude oceanic system. Front Mar Sci 7:51
Rosso MC, Stilo F, Bicchi C, Charron M, Rosso G, Menta R, Reichenbach SE, Weinert CH, Mack CI, Kulling SE, Cordero C (2021) Combined untargeted and targeted fingerprinting by comprehensive two-dimensional gas chromatography to track compositional changes on hazelnut primary metabolome during roasting. Appl Sci 11(2):1–18
Chen J, Liu H (2020) Nutritional Indices for assessing fatty acids: a mini-reviewinternational. J Mol Sci 21:5695
Fiehn O, Wohlgemuth G, Scholz M, Kind T, Lee DY, Lu Y, Moon S, Nikolau B (2008) Quality control for plant metabolomics: reporting MSI-compliant studies. Plant J 53:691–704
Narducci V, Finotti E, Galli V, Carcea M (2019) Lipids and fatty acids in Italian durum wheat (Triticum durum Desf.) cultivars. Foods 8:223
Ackman RG, Chow CK (2008) Fatty acids in foods and their heath implications. CRC Press, London, pp 155–185
Fernandes CE, da Silva Vasconcelos MA, de Almeida Ribeiro M, Sarubbo LA, Andrade SAC, de Melo Filho AB (2014) Nutritional and lipid profiles in marine fish species from Brazil. Food Chem 160:67–71
Mora-Escobedo R, Hernández-Luna P, Joaquín-Torres IC, Ortiz-Moreno A, Robles-Ramírez MDC (2015) Physicochemical properties and fatty acid profile of eight peanut varieties grown in Mexico. CyTA–J Food 13(2):300–304
Wang X, Jin Q, Wang T, Huang J, Wang X (2013) An improved method for the synthesis of 1-monoolein. J Mol Catal B Enzym 97:130–136
Piironen V, Toivo J, Lampi AM (2002) Plant sterols in cereals and cereal products. Cereal Chem 79:148–154
Perkowski J, Buśko M, Stuper K et al (2008) Concentration of ergosterol in small-grained naturally contaminated and inoculated cereals. Biologia 63:542–547
Lachman J, Hejtmánková A, Orsák M, Popov M, Martinek P (2018) Tocotrienols and tocopherols in colored-grain wheat, tritordeum and barley. Food Chem 240:725–735
Munteanu A, Zingg J-M, Azzi A (2004) Anti-atherosclerotic effects of vitamin E? Myth or reality? J Cell Mol Med 8(1):59–76
Biskup I, Zaczynska E, Krauze-Baranowska M, Fecka I (2017) Evaluation of cytotoxicity of 5-n-alkylresorcinol homologs and fraction on mouse fibroblast cell line L929. Eur Food Res Technol 243:1137–1148
Marentes-Culma R, Orduz-Díaz LL, Coy-Barrera E (2019) Targeted metabolite profiling-based identification of antifungal 5-n alkylresorcinols occurring in different cereals against Fusarium oxysporum. Molecules 24(4):770
Righetti L, Rubert J, Galaverna G, Hurkova K, Dallasta C, Hajslova J, Stranska-Zachariasova MA (2018) Novel approach based on untargeted lipidomics reveals differences in the lipid pattern among durum and common wheat. Food Chem 240:775–783
Tavoletti S, Pasquini M, Mozzon M, Servadio D, Merletti A, Mannozzi C, Foligni R (2021) Discrimination among varieties of Triticum turgidum subspecies (dicoccon, turanicum and durum) based on the fatty acid profile. J Cereal Sci 9:103–213
Rascio A, Beleggia R, Platani C, Nigro F, Codianni P, Rinaldi M, Fragasso M (2016) Metabolomic diversity for biochemical traits of Triticum sub-species. J Cereal Sci 71:224
Mihálik D, Klčová L, Ondreičková K, Hudcovicová M, Gubišová M, Klempová T, Čertík M, Pauk J, Kraic J (2015) Biosynthesis of essential polyunsaturated fatty acids in wheat triggered by expression of artificial gene. Int J Mol Sci 16(12):30046–30060
Horvath S, Dong J (2008) Geometric interpretation of gene coexpression network analysis. PLoS Comput Biol 4(8):e1000117
Schaller H (2004) New aspects of sterol biosynthesis in growth and development of higher plants. Plant Physiol Biochem 42:465–476
Bach L, Faure JD (2010) Role of very-long-chain fatty acids in plant development, when chain length does matter. CR Biol 333:361–370
Zheng H, Rowland O, Kunst L (2005) Disruptions of the Arabidopsis enoyl-CoA reductase gene reveal an essential role for very-long-chain fatty acid synthesis in cell expansion during plant morphogenesis. Plant Cell 17(5):1467–1481
Acknowledgements
This work was supported by the Tunisian Ministry of Higher Education and Scientific Research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors have no conflicts of interests.
Compliance with ethics requirements
This study does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lamine, M., Gargouri, M., Rahali, F.Z. et al. Local Tunisian durum wheat landraces revisited and rediscovered through modern integrative GC–TOF-MS™-based lipidomic profiling and chemometric approaches. Eur Food Res Technol 248, 1239–1252 (2022). https://doi.org/10.1007/s00217-022-03958-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-022-03958-4