Abstract
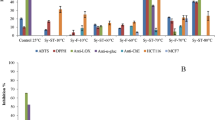
Winemaking industry produces large amounts of by-products (grape pomace or marc) every year, making necessary a sustainable management of such waste. Among red wines, Tannat variety possesses a unique polyphenolic profile. Consequently, the study of the bioactive properties of its by-product and the extraction optimization of its bioactive compounds is of great interest to state its health promoting potential. In this study, different extraction processes were tested: maceration with ethanol (95%), ultrasound-assisted ethanolic extraction optimization with surface methodology, ultrasound-assisted aqueous extraction, and hydro-alcoholic-acid extraction (EHAA). Using this last extraction procedure, higher polyphenolic content (11.459 ± 1.048 g GAE/100 g of dry extract), monomeric anthocyanins (2.030 ± 0.085 g Cyd/100 g of dry extract), antioxidant capacity (0.474 ± 0.036 mg/mL for ABTS and 0.715 ± 0.063 µmol TE/mg of dry extract for ORAC-FL), α-glucosidase (IC50 888.5 ± 79.3 µg/mL), and pancreatic lipase inhibition capacity (IC50 2431.0 ± 79.9 µg/mL) were found on Tannat grape skin. Considering as a whole, these results may imply a great antidiabetic and antiobesity potential. Moreover, EHAA showed to have anti-inflammatory capacity (IC50 587 µg/mL) on RAW 264.7 cells. In summary, EHAA possesses great potential as a functional ingredient for prevention and/or treatment of chronic diseases.






Similar content being viewed by others
References
Teixeira A, Baenas N, Dominguez-Perles R, Barros A, Rosa E, Moreno DA, Garcia-Viguera C (2014) Natural bioactive compounds from winery by-products as health promoters: a review. Int J Mol Sci 15(9):15638–15678. https://doi.org/10.3390/ijms150915638
Da Silva C, Zamperin G, Ferrarini A, Minio A, Dal Molin A, Venturini L, Buson G, Tononi P, Avanzato C, Zago E, Boido E, Dellacassa E, Gaggero C, Pezzotti M, Carrau F, Delledonne M (2013) The high polyphenol content of grapevine cultivar Tannat berries is conferred primarily by genes that are not shared with the reference genome. Plant Cell 25:4777–4788. https://doi.org/10.1105/tpc.113.118810
Puyares V, Ares G, Carrau F (2010) Searching a specific bottle for Tannat wine using a check-all-that apply question and conjoint analysis. Food Qual Prefer 21:684–691. https://doi.org/10.1016/j.foodqual.2010.05.008
Pazzini CEF, Colpo AC, Poetini MR, Pires CF, de Camargo VB, Mendez ASL, Azevedo ML, Mendes Soares JC, Folmer V (2015) Effects of red wine Tannat on oxidative stress induced by glucose and fructose in erythrocytes in vitro. Int J Med Sci 12:478–486. https://doi.org/10.7150/ijms.10529
Georgiev V, Ananga A, Tsolova V (2014) Recent advances and uses of grape flavonoids as nutraceuticals. Nutrients 6:391–415. https://doi.org/10.3390/nu6010391
Drosou C, Kyriakopoulou K, Bimpilas A, Tsimogiannis D, Krokida M (2015) A comparative study on different extraction techniques to recover red grape pomace polyphenols from vinification byproducts. Ind Crops Prod 75:141–149. https://doi.org/10.1016/j.indcrop.2015.05.063
Pedreschi R, Cisneros-Zevallos L (2006) Antimutagenic and antioxidant properties of phenolic fractions from Andean purple corn (Zea mays L.). J Agric Food Chem 54:4557–4567. https://doi.org/10.1021/jf0531050
Goula AM, Thymiatis K, Kaderides K (2016) Valorization of grape pomace: drying behavior and ultrasound extraction of phenolics. Food Bioprod Process 100:132–144. https://doi.org/10.1016/j.fbp.2016.06.016
Sasaki R, Nishimura N, Hoshino H, Isa Y, Kadowaki M, Ichi T, Tanaka A, Nishiumi S, Fukuda I, Ashida H, Horio F, Tsuda T (2007) Cyanidin 3-glucoside ameliorates hyperglycemia and insulin sensitivity due to downregulation of retinol binding protein 4 expression in diabetic mice. Biochem Pharmacol 74:1619–1627. https://doi.org/10.1016/j.bcp.2007.08.008
Hong SH, Heo JI, Kim JH, Kwon SO, Yeo KM, Bakowska-Barczak AM, Kolodziejczyk P, Ryu O-H, Choi M-K, Kang Y-H, Lim SS, Suh H-W, Huh S-O, Lee JY (2013) Antidiabetic and beta cell-protection activities of purple corn anthocyanins. Biomol Ther 21:284–289. https://doi.org/10.4062/biomolther.2013.016
Mohamed S (2014) Functional foods against metabolic syndrome (obesity, diabetes, hypertension and dyslipidemia) and cardiovasular disease. Trends Food Sci Technol 35:114–128. https://doi.org/10.1016/j.tifs.2013.11.001
Basalan M, Gungor T, Owens FN, Yalcinkaya I (2011) Nutrient content and in vitro digestibility of Turkish grape pomaces. Anim Feed Sci Technol 169:194–198. https://doi.org/10.1016/j.anifeedsci.2011.07.005
Elleuch M, Bedigian D, Roiseux O, Besbes S, Blecker C, Attia H (2011) Dietary fibre and fibre-rich by-products of food processing: characterisation, technological functionality and commercial applications: a review. Food Chem 124:411–421. https://doi.org/10.1016/j.foodchem.2010.06.077
Pozuelo MJ, Agis-Torres A, Hervert-Hernández D, Elvira López-Oliva M, Muñoz-Martínez E, Rotger R, Goñi I (2012) Grape antioxidant dietary fiber stimulates lactobacillus growth in rat cecum. J Food Sci 77:H59–H62. https://doi.org/10.1111/j.1750-3841.2011.02520.x
Saura-Calixto F (1998) Antioxidant dietary fiber product, a new concept and a potential food ingredient. J Agric Food Chem 46:4303–4306. https://doi.org/10.1021/jf9803841
Balasundram N, Sundram K, Samman S (2006) Phenolic compounds in plants and agri-industrial by-products: antioxidant activity, occurrence, and potential uses. Food Chem 99:191–203. https://doi.org/10.1016/j.foodchem.2005.07.042
Both S, Strube J, Cravatto G (2015) In: Chemat F, Strube J (eds) Mass transfer enhancement for solid–liquid extractions., green extraction of natural products. Wiley, Weinheim
González-Centeno MR, Comas-Serra F, Femenia A, Rosselló C, Simal S (2015) Effect of power ultrasound application on aqueous extraction of phenolic compounds and antioxidant capacity from grape pomace (Vitis vinifera L.): experimental kinetics and modeling. Ultrason Sonochem 22:506–514. https://doi.org/10.1016/j.ultsonch.2014.05.027
Da Porto C, Porretto E, Decorti D (2013) Comparison of ultrasound-assisted extraction with conventional extraction methods of oil and polyphenols from grape (Vitis vinifera L.) seeds. Ultrason Sonochem 20:1076–1080. https://doi.org/10.1016/j.ultsonch.2012.12.002
Roselló-Soto E, Galanakis CM, Brnčić M, Orlien V, Trujillo FJ, Mawson R, Knoerzerf K, Tiwari BK, Barba FJ (2015) Clean recovery of antioxidant compounds from plant foods, by-products and algae assisted by ultrasounds processing. Modeling approaches to optimize processing conditions. Trends Food Sci Technol 42:134–149. https://doi.org/10.1016/j.tifs.2015.01.002
Da Porto C, Natolino A, Decorti D (2015) The combined extraction of polyphenols from grape marc: ultrasound assisted extraction followed by supercritical CO2 extraction of ultrasound-raffinate. LWT Food Sci Technol 61:98–104. https://doi.org/10.1016/j.lwt.2014.11.027
Box GEP, Hunter JS, Hunter WG (1978) Statistics for experimenters. An introduction to design, data analysis, and model building. Wiley, New York
AOAC (1999) Official methods of analysis, 16th edn. Association of Official Analytical Chemists, Washington
Masuko T, Minami A, Iwasaki N, Majima T, Nishimura SI, Lee YC (2005) Carbohydrate analysis by a phenol-sulfuric acid method in microplate format. Anal Biochem 339:69–72. https://doi.org/10.1016/j.ab.2004.12.001
Giusti M, Wrolstad R (2001) In: Wrolstad RE (ed) Current protocols in food analytical chemistry. Characterization and measurement of anthocyanins by UV-Visible spectroscopy. Wiley, New York
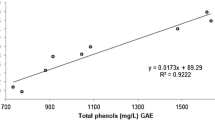
Slinkard K, Singleton VL (1977) Total phenol analysis: automation and comparison with manual methods. Am J Ecol Viticult 28:49–55
Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Rice-Evans C (1999) Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic Biol Med 26:1231–1237
Ou B, Hampsch-Woodill M, Prior R (2001) Development and validation of an improved absorbance capacity assay using fluorescein as the fluorescent. J Agric Food Chem 49:4619–4626. https://doi.org/10.1021/jf010586o
Dávalos A, Bartolomé B, Gómez-Cordovés C (2005) Antioxidant properties of commercial grape juices and vinegars. Food Chem 93:325–330. https://doi.org/10.1016/j.foodchem.2004.09.030
Martinez-Saez N, Hochkogler CM, Somoza V, del Castillo MD (2017) Biscuits with no added sugar containing stevia, coffee fibre and fructooligosaccharides modifies α-glucosidase activity and the release of GLP-1 from HuTu-80 cells and serotonin from Caco-2 cells after in vitro digestion. Nutrients 9:1–15. https://doi.org/10.3390/nu9070694
Adisakwattana S, Intrawangso J, Hemrid A, Chanathong B, Mäkynen K (2012) Extracts of edible plants inhibit pancreatic lipase, cholesterol esterase and cholesterol micellization, and bind bile acids. Food Technol Biotechnol 50:11–16
Benayad Z, Martinez-Villaluenga C, Frias J, Gomez-Cordoves C, Es-Safi NE (2014) Phenolic composition, antioxidant and anti-inflammatory activities of extracts from Moroccan Opuntia ficus-indica flowers obtained by different extraction methods. Ind Crops Prod 62:412–420. https://doi.org/10.1016/j.indcrop.2014.08.046
Deng Q, Penner MH, Zhao Y (2011) Chemical composition of dietary fiber and polyphenols of five different varieties of wine grape pomace skins. Food Res Int 44:2712–2720. https://doi.org/10.1016/j.foodres.2011.05.026
Heinonen J, Farahmandazad H, Vuorinen A, Kallio H, Yang B, Sainio T (2016) Extraction and purification of anthocyanins from purple-fleshed potato. Food Bioprod Process 99:136–146. https://doi.org/10.1016/j.fbp.2016.05.004
Pantelic MM, Dabic Zagorac D, Davidovic SM, Todic SR, Beslic ZS, Gasic UM, Tesic ZN, Natic MM (2016) Identification and quantification of phenolic compounds in berry skin, pulp, and seeds in 13 grapevine varieties grown in Serbia. Food Chem 211:243–252. https://doi.org/10.1016/j.foodchem.2016.05.051
Lapornik B, Prošek M, Wondra AG (2005) Comparison of extracts prepared from plant by-products using different solvents and extraction time. J Food Eng 71:214–222. https://doi.org/10.1016/j.jfoodeng.2004.10.036
Peralbo-Molina Á, Priego-Capote F, Dolores Luque De Castro M (2012) Comparison of extraction methods for exploitation of grape skin residues from ethanol distillation. Talanta 101:292–298. https://doi.org/10.1016/j.talanta.2012.09.028
Zu G, Zhang R, Yang L, Ma C, Zu Y, Wang W, Zhao C (2012) Ultrasound-assisted extraction of carnosic acid and rosmarinic acid using ionic liquid solution from Rosmarinus officinalis. Int J Mol Sci 13:11027–11043. https://doi.org/10.3390/ijms130911027
González-Centeno MR, Knoerzer K, Sabarez H, Simal S, Rosselló C, Femenia A (2014) Effect of acoustic frequency and power density on the aqueous ultrasonic-assisted extraction of grape pomace (Vitis vinifera L.)—a response surface approach. Ultrason Sonochem 21:2176–2184. https://doi.org/10.1016/j.ultsonch.2014.01.021
Ara KM, Karami M, Raofie F (2014) Application of response surface methodology for the optimization of supercritical carbon dioxide extraction and ultrasound-assisted extraction of Capparis spinosa seed oil. J Supercrit Fluids 85:173–182. https://doi.org/10.1016/j.supflu.2013.10.016
Gironés-Vilaplana A, Villaño D, Moreno D, García-Viguera C (2013) New isotonic drinks with antioxidant and biological capacities from berries (maqui, açaí and blackthorn) and lemon juice. Int J Food Sci Nutr 64:897–906. https://doi.org/10.3109/09637486.2013.809406
Lordan S, Smyth TJ, Soler-Vila A, Stanton C, Paul Ross R (2013) The α-amylase and α-glucosidase inhibitory effects of Irish seaweed extracts. Food Chem 141:2170–2176. https://doi.org/10.1016/j.foodchem.2013.04.123
Siqueira Melo P, Prado Massarioli A, Denny C, Ferracini Dos Santos L, Franchin M, Pereira GE, Ferreira de Souza Vieira TM, Luiz Rosalen P, de Alencar SM (2015) Winery by-products: extraction optimization, phenolic composition and cytotoxic evaluation to act as a new source of scavenging of reactive oxygen species. Food Chem 181:160–169. https://doi.org/10.1016/j.foodchem.2015.02.087
Rafi MM, Yadav PN, Rossi AO (2007) Glucosamine inhibits LPS-induced COX-2 and iNOS expression in mouse macrophage cells (RAW 264.7) by inhibition of p38-MAP kinase and transcription factor NF-κB. Mol Nutr Food Res 51:587–593. https://doi.org/10.1002/mnfr.200600226
de Lacerda Bezerra I, Rute Cordeiro Caillot A, Gusmão Ferreira Palhares LC, Paixão Santana-Filho A, Ferreira Chavante S, Lanzi Sassaki G (2018) Structural characterization of polysaccharides from Cabernet Franc, Cabernet Sauvignon and Sauvignon Blanc wines: anti-inflammatory activity in LPS stimulated RAW 264.7 cells. Carbohyd Polym 186:91–99. https://doi.org/10.1016/j.carbpol.2017.12.082
Chen YC, Shen SC, Lee WR, Hou WC, Yang LL, Lee TJ (2001) Inhibition of NOS, nitric oxide synthase inhibitors and lipopolysaccharide induced inducible and cyclooxygenase-2 gene expressions by rutin, quercetin, and quercetin 82, pentaacetate in RAW 264.7 macrophages. J Cell Biochem 82:537–548
Nishiumi S, Mukai R, Ichiyanagi T, Ashida H (2012) Suppression of lipopolysaccharide and galactosamine-induced hepatic inflammation by red grape pomace. J Agric Food Chem 60:9315–9320. https://doi.org/10.1021/jf302298n
Panico AM, Cardile V, Avondo S, Garufi F, Gentile B, Puglia C, Bonina F, Santagati NA, Ronsisvalle G (2006) The in vitro effect of a lyophilized extract of wine obtained from Jacquez grapes on human chondrocytes. Phytomedicine 13(7):522–526. https://doi.org/10.1016/J.PHYMED.2005.06.009
Gonçalves GA, Soares AA, Correa RCG, Barros L, Haminiuk CWI, Peralta RM, Ferreira ICFR, Bracht A (2017) Merlot grape pomace hydroalcoholic extract improves the oxidative and inflammatory states of rats with adjuvant-induced arthritis. J Funct Foods 33:408–418. https://doi.org/10.1016/j.jff.2017.04.009
Acknowledgements
Funding is acknowledged from Agencia Nacional de Investigación e Innovación (Grant POS_NAC_2016_1_130292), program EMHE-CSIC (Enhancing Mobility between Latin America, Caribbean, and the European Union in Health and Environment) and Programa de Desarrollo de las Ciencias Básicas (PEDECIBA-UDELAR). The SUSCOFFEE Project (AGL2014-57239-R) also funded this work. A. Iriondo-DeHond is a fellow of the FPI predoctoral program of MINECO (BES-2015-072191).
Author information
Authors and Affiliations
Contributions
AMFF performed the chemical and biological assays and data processing, and also contributed to the interpretation and global integration of the results and manuscript preparation; ED contributed to the critical manuscript revision; AMF and AID contributed to the interpretation of the global results and critical manuscript revision; MDC contributed to the study design, the interpretation of the global results, and manuscript preparation.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interest.
Compliance with ethics requirements
This article does not contain any studies with human or animal subjects.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fernández-Fernández, A.M., Iriondo-DeHond, A., Dellacassa, E. et al. Assessment of antioxidant, antidiabetic, antiobesity, and anti-inflammatory properties of a Tannat winemaking by-product. Eur Food Res Technol 245, 1539–1551 (2019). https://doi.org/10.1007/s00217-019-03252-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-019-03252-w