Abstract
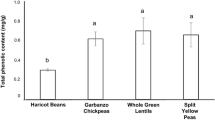
Recent interest in pulses has resulted in the application of their cooking water (PCW) as replacement for egg white in meringues. Nonetheless, scientific understanding of their chemical composition, physicochemical properties and effect on food quality is lacking. This study analysed the PCW of haricot beans, garbanzo chickpeas, whole green lentils and split yellow peas, determining their composition and effect on meringues’ quality. The PCW tested contained different amounts of sugar, soluble and insoluble fibre, protein, ash and saponins. All PCW exerted foaming ability (39–97%), directly correlated to their protein content and lower than that of egg white (400%). Moreover, gelling ability was observed, resulting in the development of egg-like meringues, with their hardness inversely correlated to the content of insoluble fibre. Whole green lentils expressed the highest foaming ability while garbanzo chickpeas resulted in the strongest gelling ability. Sensory analysis of meringues depicted low acceptance for the taste of meringues made with the PCW of haricot beans and whole green lentils. On the contrary, high acceptance was determined for garbanzo chickpeas and split yellow peas, similarly to that of the egg white.

Similar content being viewed by others
References
Liu XD, Han RX, Yun H, Jung KC, Jin DI, Lee BD, Min TS, Jo C (2009) Effect of irradiation on foaming properties of egg white proteins. Poult Sci 88(11):2435–2441
Wilde PJ, Clarke DC (1996) Foam formation and stability. In: Hall GM (ed) Methods of testing protein functionality. Blackie Academic and Professional, London, pp 110–149
Mariotti M, Pagani MA, Lucisano M (2013) The role of buckwheat and HPMC on the breadmaking properties of some commercial gluten-free bread mixtures. Food Hydrocolloid 30(1):393–400
Li JM, Nie SP (2016) The functional and nutritional aspects of hydrocolloids in foods. Food Hydrocolloid 53:46–61
Hüttner EK, Arendt EK (2010) Recent advances in gluten-free baking and the current status of oats. Trends Food Sci Tech 21(6):303–312
The Official Aquafaba Website (2017) http: www.aquafaba.com. Accessed 3 April 2017
Vidal-Valverde C, Frías J, Valverde S (1993) Changes in the carbohydrate composition of legumes after soaking and cooking. J Am Diet Assoc 93(5):547–550
El-Adawy TA (2002) Nutritional composition and antinutritional factors of chickpeas (Cicer arietinum L.) undergoing different cooking methods and germination. Plant Food Hum Nutr 57(1):83–97
Ghribi AM, Sila A, Gafsi IM, Blecker C, Danthine S, Attia H, Bougatef A, Besbes S (2015) Structural, functional, and ACE inhibitory properties of water-soluble polysaccharides from chickpea flours. Int J Biol Macromol 75:276–282
Alajaji SA, El-Adawy TA (2006) Nutritional composition of chickpea (Cicer arietinum L.) as affected by microwave cooking and other traditional cooking methods. J Food Compos Anal 19(8):806–812
Cherian BM, Leao AL, Caldeira MDS, Chiarelli D, de Souza SF, Narine S, de Morais Chaves MR (2012) Use of saponins as an effective surface modifier in cellulose nanocomposites. Mol Cryst Liq Cryst 556(1):233–245
Güçlü-Üstündağ Ö, Mazza G (2007) Saponins: properties, applications and processing. Crit Rev Food Sci 47(3):231–258
Klamczynska B, Czuchajowska Z, Baik BK (2001) Composition, soaking, cooking properties and thermal characteristics of starch of chickpeas, wrinkled peas and smooth peas. Int J Food Sci Tech 36(5):563–572
AACC International (2001) Approved methods of analysis, 11th Edn. Method 44-19.01. Moisture—Air-oven method, drying at 135°. AACC International: St. Paul
Pollock CJ, Jones T (1979) Seasonal patterns of fructan metabolism in forage grasses. New Phytol 83(1):9–15
Jermyn MA (1956) A new method for determining ketohexoses in the presence of aldohexoses. Nature 177(4497):38–39
AOAC (1995) Official methods of analysis of AOAC International, 16th edn. AOAC International Publ, Aarlington
Ncube B, Ngunge VNP, Finnie JF, Van Staden J (2011) A comparative study of the antimicrobial and phytochemical properties between outdoor grown and micropropagated Tulbaghia violacea Harv. plants. J Ethnopharmacol 134(3):775–780
Sathe SK, Salunkhe DK (1981) Functional properties of the great northern bean (Phaseolus vulgaris L.) proteins: emulsion, foaming, viscosity, and gelation properties. J Food Sci 46(1):71–81
AACC International (2000) Approved methods of analysis, 10th Ed. Method 10-05.01. Guidelines for measurement of volume by rapeseed displacement. AACC International, St. Paul
Sánchez-Mata MC, Peñuela-Teruel MJ, Cámara-Hurtado M, Díez-Marqués C, Torija-Isasa ME (1998) Determination of mono-, di-, and oligosaccharides in legumes by high-performance liquid chromatography using an amino-bonded silica column. J Agr Food Chem 46(9):3648–3652
Knudsen KEB (1997) Carbohydrate and lignin contents of plant materials used in animal feeding. Anim Feed Sci Technol 67(4):319–338
Rehinan ZU, Rashid M, Shah WH (2004) Insoluble dietary fibre components of food legumes as affected by soaking and cooking processes. Food Chem 85(2):245–249
Vasishtha H, Srivastava RP (2013) Effect of soaking and cooking on dietary fibre components of different type of chickpea genotypes. J Food Sci Technol Mys 50(3):579–584
Oenning G, Juillerat MA, Fay L, Asp NG (1994) Degradation of oat saponins during heat processing-effect of pH, stainless steel, and iron at different temperatures. J Agr Food Chem 42(11):2578–2582
Fenwick DE, Oakenfull D (1983) Saponin content of food plants and some prepared foods. J Sci Food Agric 34(2):186–191
Song HP, Kim B, Choe JH, Jung S, Kim KS, Kim DH, Jo C (2009) Improvement of foaming ability of egg white product by irradiation and its application. Radiat Phys Chem 78(3):217–221
Iqbal A, Khalil IA, Ateeq N, Khan MS (2006) Nutritional quality of important food legumes. Food Chem 97(2):331–335
Imeson AP (2012) Thickening and gelling agents for food. Springer Science & Business Media, Berlin
Ngouémazong ED, Christiaens S, Shpigelman A, Loey A, Hendrickx M (2015) The emulsifying and emulsion-stabilizing properties of pectin: a review. Compr Rev Food Sci J 14(6):705–718
Rao MR, Warrier DU, Rao SH (2015) Evaluation of phosphorylated psyllium seed polysaccharide as a release retardant. Indian J Pharm Educ 77(5):605–612
Gross J (2012) Pigments in vegetables: chlorophylls and carotenoids. Springer Science & Business Media, Berlin
Xu BJ, Yuan SH, Chang SKC (2007) Comparative analyses of phenolic composition, antioxidant capacity, and color of cool season legumes and other selected food legumes. J Food Sci 72(2):S167–S177
Drewnowski A, Gomez-Carneros C (2000) Bitter taste, phytonutrients, and the consumer: a review. Am J Clin Nutr 72(6):1424–1435
Boye JI, Aksay S, Roufik S, Ribéreau S, Mondor M, Farnworth E, Rajamohamed SH (2010) Comparison of the functional properties of pea, chickpea and lentil protein concentrates processed using ultrafiltration and isoelectric precipitation techniques. Food Res Int 43(2):537–546
Acknowledgements
This research has been sponsored by Lincoln University. The authors would like to thank Roger Cresswell and Vicky Zhang for performing the protein analysis; Andrea Hogan, Shuang Jiang and Rosy Tung for their support with the quantification of total soluble carbohydrates and for performing the ash and fat analyses, as well as Joan Narciso for helping with the carbohydrate extraction. Finally, we acknowledge Libby Bird for her literature review on hydrocolloids functionality and Weihan Zhang for taking pictures of the meringues and performing their sensory analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Compliance with ethical requirements
Sensory analysis was conducted in compliance with ethics standards upon approval by the ethics committee at Lincoln University.
Rights and permissions
About this article
Cite this article
Stantiall, S.E., Dale, K.J., Calizo, F.S. et al. Application of pulses cooking water as functional ingredients: the foaming and gelling abilities. Eur Food Res Technol 244, 97–104 (2018). https://doi.org/10.1007/s00217-017-2943-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-017-2943-x