Abstract
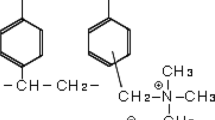
A simple method for recovery of dissolved vanillin from aqueous solutions by the adsorption–regeneration technique has been developed. Three macroporous adsorption resins with crosslinked-polystyrene framework were NKA-2, S-8 and H103, respectively. Static equilibrium adsorption studies were used for comparing the adsorption capacities of the three resins. The results showed that H103 resin had the best adsorption capacity because of its large and nonpolar surface areas. The effects of pH, temperature and salt concentration on the adsorption capacity of H103 were investigated. The results revealed that the resin had a maximum adsorption capacity, 416 mg/g (vanillin/resin) in acidic condition when the molecule of vanillin is neutral. Furthermore, its adsorption capacity increased with the increase of temperature and salt concentration. The mass transfer zone motion model was used for analyzing the fixed bed adsorption. H103 resin had a shorter mass transfer zone of 24.7 cm and both NKA-2 and S-8 were higher than 30 cm in the experiment conditions. More than 95.6% of adsorbed vanillin can be recovered by use of 3–5 bed volumes of absolute ethanol. The resin can be used repeatedly by simple regeneration and its adsorption capacity was almost unchangeable.







Similar content being viewed by others
References
Walton NJ, Narbad A, Faulds CB, Williamson G (2000) Novel approaches to the biosynthesis of vanillin. Curr Opin Biotechnol 11(5):490–496
Lomascolo A, Stentelaire C, Asther M, Meessen LL (1999) Basidiomycetes as new biotechnological tools to generate natural aromatic flavours for the food industry. Trends Biotechnol 17:282–289
Pak FE, Gropper S, Dai WD, Frenkel DH, Belanger FC (2004) Characterization of a multifunctional methyltransferase from the orchid vanilla planifolia. Plant Cell Rep 22:959–966
Dignum MJW, Kerler J, Verpoorte R (2001) Vanilla production: technological, chemical, and biosynthetic aspects. Food Rev Int 17:199–219
Priefert H, Rabenhorst J, Steinbüchel A (2001) Biotechnological production of vanillin. Appl Microbiol Biotechnol 56:296–314
Walton NJ, Mayer M, Narbad A (2003) Vanillin. Phytochemistry 63:505–515
Krings U, Berger RG (1998) Biotechnological production of flavours and fragrances. Appl Microbiol Biotechnol 49:1–8
Ribeiro MHL, Ribeiro IAC (2005) Recovery of erythromycin from fermentation broth by adsorption onto neutral and ion-exchange resins. Sep Purif Technol 45(3):232–239
Kim CH, Kim SW, Hong SI (2000) An integrated fermentation–separation process for the production of red pigment by Serratia sp KH-95. Process Biochem 35(5):485–490
Sun Y, Li XT, Xu C, Chen JL, Li AM, Zhang QX (2005) Adsorption of catechol from aqueous solution by aminated hypercrosslinked polymers. J Environ Sci China 17(4):584–588
Fu BQ, Liu J, Li H, Li L, Lee FSC, Wang XR (2005) The application of macroporous resins in the separation of licorice flavonoids and glycyrrhizic acid. J Chromatogr A 1089(1–2):18–24
Zhang WM, Chen JL, Pan BC, Zhang QX (2005) Binary competitive and cooperative adsorption of aromatic compounds on polymeric resins. Chin J Polym Sci 23(5):503–511
Xu ZY, Zhang QX, Chen JL, Wang LS, Anderson GK (1999) Adsorption of naphthalene derivatives on hypercrosslinked polymeric adsorbents. Chemosphere 38(9):2003–2011
Li AM, Wu HS, Zhang QX, Zhang GC, Long C, Fei ZH, Liu FQ, Chen JL (2004) Thermodynamic study of adsorption of phenolic compounds from aqueous solution by a water-compatible hypercrosslinked polymeric adsorbent. Chin J Polym Sci 22(3):259–267
Lee JW, Shim WG, Ko JY, Moon H (2004) Adsorption equilibria, kinetics, and column dynamics of chlorophenols on a nonionic polymeric sorbent, XAD-1600. Sep Sci Technol 39(9):2041–2065
Wang XJ, Zhao JF, Xia SQ, Li AM, Chen L (2004) Adsorption mechanism of phenolic compounds from aqueous solution on hypercrosslinked polymeric adsorbent. J Environ Sci China 16(6):919–924
Pan BC, Zhang X, Zhang WM, Zheng JZ, Pan BJ, Chen JL, Zhang QX (2005) Adsorption of phenolic compounds from aqueous solution onto a macroporous polymer and its aminated derivative: isotherm analysis. J Hazard Mater 121(1–3):233–241
Jogdeo DA, Niranjan K, Pangarkar VG (2000) Recovery of allyl isothiocyanate from steam distillation condensate using adsorption. J Chem Technol Biotechnol 75:673–680
Meessen LL, Lomascolo A, Bonnin E, Thibault JF, Buleon A, RollerM, Asther M, Record E, Ceccaldi BC, Asther M (2002) A biotechnological process involving filamentous fungi to produce natural crystalline vanillin from maize bran. Appl Biochem Biotechnol 102:141–153
Nilvebrant NO, Reimann A, Larsson S, Jonson LJ (2001) Detoxification of lignocellulose hydrolysates with ion-exchange resins. Appl Biochem Biotechnol 91(3):35–49
Stentelaire C, Meessen LL, Oddou J, Bernard O, Bastin G, Ceccaldi BC, Asther M (2000) Design of a fungal bioprocess for vanillin production from vanillic acid at scalable level by Pycnoporus cinnabarinus. J Biosci Bioeng 89(3):223–230
Wang CL, Li SL, Zhou QL, Zhang M, Zeng JH, Zhang Y (2005) Extraction of vanillin in fermented liquor by macroporous adsorption resin. Jingxi Huagong 22(6):458–460
Romo A, Penas FJ, Isasi JR (2004) Sorption of dibenzofuran derivatives from aqueous solutions by β-cyclodextrin polymers: an isosteric heat approach. J Colloid Interf Sci 279:55–60
China national research institute of food & fermentation industries, Southern Yangtze University (2004) Food Engineering. China Light Industry Press, Beijing, pp 682–689
Li R, Jiang ZT, Mao LY, Shen HX (1998) Adsorbed resin phase spectrophotometric determination of vanillin or/and its derivatives. Talanta 47(5):1121–1127
Zhu BY, Zhao ZG (1996) Interface chemistry basic. Chemical Industry Press, Beijing pp 254
Acknowledgments
The authors acknowledge Natural Science Foundation of Tianjin for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, QF., Jiang, ZT., Gao, HJ. et al. Recovery of vanillin from aqueous solutions using macroporous adsorption resins. Eur Food Res Technol 226, 377–383 (2008). https://doi.org/10.1007/s00217-006-0548-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-006-0548-x