Abstract
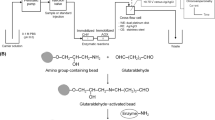
Previous works on organic phase enzyme electrodes (OPEE) applied methods that worked mostly under stirred conditions. The aim of our present work was to develop a flow-through measuring set-up for determination of cholesterol content in organic media. For determination of free cholesterol content cholesterol oxidase (COD) was used, while for measuring the total cholesterol content a bi-enzyme cell containing immobilised cholesterol esterase (CE) and cholesterol oxidase was developed. Enzyme immobilisation took place on a natural protein membrane, by a glutaraldehyde cross-linking method, in a thin-layer enzyme cell made from Teflon. The enzyme cell was connected into a stopped-flow injection system (SFIA), with a flow-through amperometric detector. The parameters of biochemical and electrochemical reactions were measured. The effect on amperometric detection of different organic salts as electron mediator or conducting salts was studied. The optimal concentration of (TBATS) was 2.4 mg L−1 while for (FMCA) an optimal concentration was found at 0.4 mg L−1. The minimum amount of water, necessary for enzymatic activity in the organic phase, was also determined. Changing the concentration of toluene in acetonitrile carrier solution, the peaks increased definitely in the range 10–40% toluene. Since CE and COD were immobilised together in the enzyme cell, the conversion rate was found to be about 0.7–0.8 when the toluene content was higher then 30%. The linear measuring range for cholesterol oleate and cholesterol was 0.1–0.5 mM. Total cholesterol content of lard, butter and pasta samples were determined. It is concluded that an organic phase bi-enzyme cell may be suitable for cholesterol determination in food.







Similar content being viewed by others
References
Karam WG, Chiang JYL (1994) J Lipid Res 35:1222–2131
Ram MK, Bertoncello P, Ding H, Paddeu S, Nicolini C (2001) Biosens Bioelectr 16:849–856
Vidal JC, Garcia-Ruiz E, Castillo JR (2000) J Pharm Biomed Anal 24:51–63
Gobi K, Mizutani F (2001) Sensors Actuators B 4062:1-6
Situmorang M, Alexander PW, Hibbert DB (1999) Talanta 49/3:639–649
Bongiovanni C, Ferri T, Poscia A, Varalli M, Santucci R, Desideri A (2001) Bioelectrochemistry 54:17–22
Yao T, Sato M, Kobayashi Y, Wasa T (1985) Anal Biochem 149:387–391
Krug A, Suleiman AA, Guilbault GG, Kellner R (1992) Enzyme Microb Technol 14:313–316
Baticz O, Tömösközi S (2002) Nahrung 46:46–50
Buckland BC, Dunnill P, Lilly D (1975) Biotechnol Bioeng 17:815–826
Liu WH, Houng WC, Tsai MS (1996) Enzyme Microb Technol 18:184–189
Narayan VS, Klibanov AM (1993) Biotech Bioeng 41:390–393
Kazandjian RZ, Dordick JS, Klibanov AM (1986) Biotechnol Bioeng 28:417–421
Zaks A, Klibanov AM (1988) Biol Chem 263:3194–3201
Dordick JS (1989) Enzyme Microb Technol 11:194–211
Pineiro-Availa G, Salvador A, de la Guardia M (1998) Analyst 123:999–1003
Kumar H, Kumar A, Kumari P, Jyotirmai S, Tulsani NB (1999) Biotechnol Appl Biochem 30:231–233
Hall GF, Turner APF (1991) Anal Lett 24:1375–1388
Campanella L, Tomassetti M (1996) Food Technol Biotechnol 34:131–141
Pena N, Ruiz G, Reviejo AJ, Pingarron JM (2001) Anal Chem 73(6):1190–1195
Klibanov AM (1986) Chemtech 354–358
Rong-Zhen-Zhang, Long-Li, Shu-Tao-Liu, Ru-Ming-Chen, Ping-Fan-Rao, (1999) J Food Biochem 23:351–361
Johnson JH, McIntyre P, Zdunek J (1995) J Chromatogr A 718:371–81
Váradi M, Adányi N, Nagy G, Rezessy-Szabó J (1993) Biosens Bioelectr 8:339–345
Adányi N, Tóth-Markus M, Szabó EE, Váradi M, Sammartino MP, Tomassetti M, Campanella L (2003) Anal Chim Acta (submitted)
Holland B, Welch AA, Unwin ID, Buss DH, Paul AA, Southgate DAT (eds) (1992) McCance and Widdowson’s The composition of foods. RSC, Cambridge
Choong YM, Lin HJ, Wang ML (1999) J Food Drug Analysis 7(4):279–290
Lee SY, Rhee HI, Tae WC, Shin JC, Park BK (1989) Appl. Microbiol Biotechnol 31:542–546
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Adányi, N., Váradi, M. Development of organic phase amperometric biosensor for measuring cholesterol in food samples. Eur Food Res Technol 218, 99–104 (2003). https://doi.org/10.1007/s00217-003-0805-1
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-003-0805-1