Abstract.
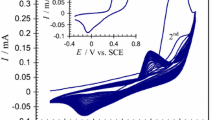
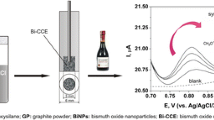
9,10-Phenanthrenequinone (PQ) supported on graphite powder by adsorption was dispersed in propyltrimethoxysilane-derived gels to yield a conductive composite which was used as electrode material to fabricate a PQ-modified carbon ceramic electrode. In this configuration, PQ acts as a catalyst, graphite powder guarantees conductivity by percolation, the silicate provides a rigid porous backbone, and the propyl groups endow hydrophobicity and thus limit the wetting region of the modified electrode. Square-wave voltammetry was exploited to investigate the pH-dependent electrochemical behavior of the composite electrode and an almost Nernstian response was obtained from pH 0.42 to 6.84. Because the chemically modified electrode can electrocatalyze the reduction of iodate in acidic aqueous solution (pH 2.45), it was used as an amperometric sensor for the determination of iodate in table salt. The advantages of the electrode are that it can be polished in the event of surface fouling, it is simple to prepare, has excellent chemical and mechanical stability, and the reproducibility of surface-renewal is good.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Yang, Z., Wang, P., Zhang, W. et al. Sol–gel-derived carbon ceramic electrode containing 9,10-phenanthrenequinone, and its electrocatalytic activity toward iodate. Fresenius J Anal Chem 371, 337–341 (2001). https://doi.org/10.1007/s002160100950
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s002160100950