Abstract
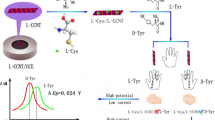
Chiral recognition is an emerging field of modern chemical analysis, and the development of health-related fields depends on the production of enantiomers. Cellulose is a kind of natural polymer material with certain chiral recognition ability. Limited by the chiral recognition ability of natural cellulose itself, more cellulose derivatives have been gradually developed for chiral recognition and separation. Based on the difference in action between cellulose derivatives and enantiomers, this work synthesized cellulose-tris(4-methylphenylcarbamate) (CMPC) chiral recognition mediators and a CMPC-functionalized extended-gate organic field effect transistor (EG-OFET) was constructed for the first time. Three chiral molecules were selected as model analytes to evaluate the enantiomeric recognition ability of the platform, including threonine (Thr), 2-chloromandelic acid (CA), and 1,2-diphenylethylenediamine (DPEA). The detection limit for 1,2-diphenylethylenediamine (DPEA) is down to 10−13 M. Through the amplification effect of the EG-OFET platform, the difference in the interaction between CMPC and three chiral molecules with different structures is converted into a current signal output. At the same time, the enantiomer discrimination mechanism of CMPC was further studied by means of spectroscopy and nuclear magnetic resonance.
Graphical abstract






Similar content being viewed by others
References
Wang ZQ, Liu JD, Chen W, Bai ZW. Enantioseparation characteristics of biselector chiral stationary phases based on derivatives of cellulose and amylose [J]. J Chromatogr A. 2014;1346:57–68.
Ward TJ, Ward KD. Chiral separations: a review of current topics and trends[J]. Anal Chem. 2012;84(2):626–35.
Zhu QF, Scriba GKE. Advances in the use of cyclodextrins as chiral selectors in capillary electrokinetic chromatography: fundamentals and applications [J]. Chromatographia. 2016;79(21–22):1403–35.
Ali I, Suhail M, Al-Othman ZA, Alwarthan A, Aboul-Enein HY. Enantiomeric resolution of multiple chiral centers racemates by capillary electrophoresis [J]. Biomed Chromatogr. 2016;30(5):683–94.
Zhu HTZ, Li Q, Gao ZC, Wang HL, Shi BB, Wu YT, et al. Pillararene host-guest complexation induced chirality amplification: a new way to detect cryptochiral compounds [J]. Angew Chem Int Ed. 2020;59(27):10868–72.
Alwera S, Alwera V, Sehlangia S. An efficient method for the determination of enantiomeric purity of racemic amino acids using micellar chromatography, a green approach [J]. Biomed Chromatogr. 2020;34(11):e4943.
Bao JJ, Jia F, Li Y, Liang Q, Wang Y. Synthesis and applications of sulfopropyl ether gamma-cyclodextrin polymer as chiral selector in capillary electrophoresis [J]. Anal Bioanal Chem. 2016;408(13):3639–49.
Mohankumar P, Ajayan J, Mohanraj T, Yasodharan R. Recent developments in biosensors for healthcare and biomedical applications: a review [J]. Measurement. 2021;167:108293.
Pedroza Dias Mello HJN, Bachour Junior B, Mulato M. Polyanine-based field effect transistor for DNA/RNA biomarker sensing: comparison to electrochemical impedance and inorganic layer [J]. Sensors Actuators A Phys. 2021;318:112481.
Pan TM, Lin CH. High performance NiOx extended-gate field-effect transistor biosensor for detection of uric acid [J]. J Electrochem Soc. 2021;168(1):017511.
Mulla MY, Tuccori E, Magliulo M, Lattanzi G, Palazzo G, Persaud K, et al. Capacitance-modulated transistor detects odorant binding protein chiral interactions [J]. Nat Commun. 2015;6:6010.
Torricelli F, Adrahtas D Z, Bao Z N, Berggren M, Biscarini F, Bonfiglio A, et al. Electrolyte-gated transistors for enhanced performance bioelectronics [J]. Nat Rev Methods Primers. 2021;1:66.
Sun YW, Wang Y, Wu YF, Wang XP, Li XG, Wang SY, et al. A chiral organic field-effect transistor with a cyclodextrin modulated copper hexadecafluorophthalocyanine semiconductive layer as the sensing unit [J]. Anal Chem. 2018;90(15):9264–71.
Iskierko Z, Checinska A, Sharma PS, Golebiewska K, Noworyta K, Borowicz P, et al. Molecularly imprinted polymer based extended-gate field-effect transistor chemo sensors for phenylalanine enantioselective sensing [J]. J Mater Chem C. 2017;5(4):969–77.
Pullano SA, Critello CD, Mahbub I, Tasneem NT, Shamsir S, Islam SK, et al. EGFET-based sensors for bioanalytical applications: a review [J]. Sensors. 2018;18(11):4042.
He Y, Pang H, Liao B. Progress in cellulose-based chiral stationary phases [J]. Prog Chem. 2006;18(7):957–65.
Wang XP, Li H, Quan KJ, Zhao L, Qiu HD, Li ZG. Preparation and applications of cellulose-functionalized chiral stationary phases: a review [J]. Talanta. 2021;225:121987.
Chenl X, Yamamoto C, Okamoto Y. Polysaccharide derivatives as useful chiral stationary phases in high-performance liquid chromatography [J]. Pure Appl Chem. 2007;79(9):1561–73.
Li L, Yuan X, Shi ZG, Xu LY. Chiral stationary phase based on cellulose derivative coated polymer microspheres and its separation performance [J]. J Chromatogr A. 2020;1623:461154.
Agathokleous EA, Stavrou IJ, Kapnissi-Christodoulou C. Comparison of cyclofructan-, cyclodextrin-, and polysaccharide-based chiral stationary phases for the separation of pharmaceuticals[J]. Anal Bioanal Chem. 2022;414(3):1323–33.
Li WZ, Zhang WQ, Zhao QY, Li YM, Ma CL, Chen LR, et al. Direct optical resolution of the enantiomers of novel chiral tetrahedral metal clusters by HPLC on a cellulose tris-(3,5-dimethylphenylcarbamate) stationary phase [J]. Anal Bioanal Chem. 2004;378(5):1364–8.
Cavazzini A, Pasti L, Massi A, Marchetti N, Dondi F. Recent applications in chiral high performance liquid chromatography: a review [J]. Anal Chim Acta. 2011;706(2):205–22.
Felix G. Regioselectively modified polysaccharide derivatives as chiral stationary phases in high-performance liquid chromatography [J]. J Chromatogr A. 2001;906(1):171–84.
Shen J, Wang F, Bi W, Liu B, Liu SY, Okamoto Y. Synthesis of cellulose carbamates bearing regioselective substituents at 2,3-and 6-positions for efficient chromatographic enantioseparation [J]. J Chromatogr A. 2018;1572:54–61.
Yin CC, Yang TT, Zhang JM, Zhang J. Homogeneous synthesis of cellulose phenylcarbamates and evaluation of their enantioseparation capabilities [J]. Chin J Chromatogr. 2020;38(4):476–83.
Yin CC, Zhang JM, Chang LM, Zhang M, Yang TT, Zhang XC, et al. Regioselectively substituted cellulose mixed esters synthesized by two-steps route to understand chiral recognition mechanism and fabricate high-performance chiral stationary phases [J]. Anal Chim Acta. 2019;1073:90–8.
Chen WW, Zhang M, Feng Y, Wu J, Gao X, Zhang JM, et al. Homogeneous synthesis of partially substituted cellulose phenylcarbamates aiming at chiral recognition [J]. Polym Int. 2015;64(8):1037–44.
Zhang JJ, Wang SY, Zhang P, Fan SC, Dai HT, Xiao Y, et al. Engineering a cationic supramolecular charge switch for facile amino acids enantiodiscrimination based on extended-gate field effect transistors [J]. Chin Chem Lett. 2022. https://doi.org/10.1016/j.cclet.2021.11.081.
Funding
This work was financially funded by the National Key R&D Program of China (No. 2019YFC1905500), the National Natural Science Foundation of China (Nos. 21922409, 21976131), and the Tianjin Research Program of Application Foundation and Advanced Technology (No. 18JCZDJC37500).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Published in the topical collection Young Investigators in (Bio-)Analytical Chemistry 2023 with guest editors Zhi-Yuan Gu, Beatriz Jurado-Sánchez, Thomas H. Linz, Leandro Wang Hantao, Nongnoot Wongkaew, and Peng Wu.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, JJ., Wang, SY., Dai, HT. et al. Construction of cellulose-based highly sensitive extended-gate field effect chiral sensor. Anal Bioanal Chem 415, 4245–4254 (2023). https://doi.org/10.1007/s00216-022-04306-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-022-04306-x