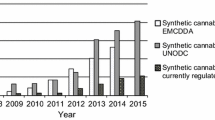
Abstract
The popularity of new psychoactive substances among drug users has become a public health concern worldwide. Among them, synthetic cannabinoids (SCs) represent the largest, most diversified and fastest growing group. Commonly known as ‘synthetic marijuana’ as an alternative to cannabis, these synthetic compounds are easily accessible via the internet and are sold as ‘herbal incenses’ under different brand names with no information about the chemical composition. In the present work, we aim to integrate gas chromatography-tandem mass spectrometry (GC-MS) and nuclear magnetic resonance (NMR) data as useful strategy for the identification and confirmation of synthetic cannabinoids present in nine seized herbal incenses. The analysis of all samples allowed the initial identification of 9 SCs, namely 5 napthoylindoles (JWH-018, JWH-073, JWH-122, JWH-210, MAM-2201), APINACA, XLR-11 and CP47,497-C8 and its enantiomer. JWH-018 was the most frequently detected synthetic compound (8 of 9 samples), while APINACA and XLR-11 were only identified in one herbal product. Other non-cannabinoid drugs, including oleamide, vitamin E and vitamin E acetate, have also been detected. Oleamide and vitamin E are two adulterants, frequently added to herbal products to mask the active ingredients or added as preservatives. However, to our knowledge, no analytical data about vitamin E acetate was reported in herbal products, being the first time that this compound is identified on this type of samples. The integration data obtained from the used analytical technologies proved to be useful, allowing the preliminary identification of the different SCs in the mixture. Furthermore, the examination of mass spectral fragment ions, as well as the results of both 1D and 2D NMR experiments, enabled the identification and confirmation of the molecular structure of SCs.





Similar content being viewed by others
References
Alves VL, Gonçalves JL, Aguiar J, Teixeira HM, Câmara JS. The synthetic cannabinoids phenomenon: from structure to toxicological properties. A review. Crit Rev Toxicol. 2020;50(5):1–24. https://doi.org/10.1080/10408444.2020.1762539.
Dronova M, Smolianitski E, Lev O. Electrooxidation of new synthetic cannabinoids: voltammetric determination of drugs in seized street samples and artificial saliva. Anal Chem. 2016;88(8):4487–94. https://doi.org/10.1021/acs.analchem.6b00368.
EMCDDA. Perspectives on drugs: synthetic cannabinoids in Europe. 2017. http://www.emcdda.europa.eu/publications/pods/synthetic-cannabinoids_en. Accessed 12 April 2019.
EMCDDA. EU drug markets report 2019. 2019. https://www.emcdda.europa.eu/publications/joint-publications/eu-drug-markets-report-2019_en. Accessed 15 December 2019.
Papaseit E, Pérez-Mañá C, Pérez-Acevedo AP, Hladun O, Torres-Moreno MC, Muga R, et al. Cannabinoids: from pot to lab. Int J Med Sci. 2018;15(12):1286–95. https://doi.org/10.7150/ijms.27087.
Duccio P, Fabrizio S, Laura O. Synthetic cannabinoids and synthetic cannabinoid-induced psychotic disorders. In: Compton MT, Manseau MW, editors. The complex connection between cannabis and schizophrenia. San Diego: Academic Press; 2018. p. 199–220.
Britannica E. Cannabis. Encyclopædia Britannica, inc. 2018. https://www.britannica.com/plant/cannabis-plant. Accessed 17 April 2019.
Solimini R, Busardo FP, Rotolo MC, Ricci S, Mastrobattista L, Mortali C, et al. Hepatotoxicity associated to synthetic cannabinoids use. Eur Rev Med Pharmacol Sci. 2017;21(1 Suppl):1–6.
Lefever TW, Marusich JA, Thomas BF, Barrus DG, Peiper NC, Kevin RC, et al. Vaping synthetic cannabinoids: a novel preclinical model of E-cigarette use in mice. Subst Abus. 2017;11:1178221817701739. https://doi.org/10.1177/1178221817701739.
Bilici R. Synthetic cannabinoids. North Clin Istanb. 2014;1(2):121–6. https://doi.org/10.14744/nci.2014.44153.
Zuba D, Byrska B, Maciow M. Comparison of “herbal highs” composition. Anal Bioanal Chem. 2011;400(1):119–26. https://doi.org/10.1007/s00216-011-4743-7.
Cooper ZD. Adverse effects of synthetic cannabinoids: management of acute toxicity and withdrawal. Curr Psychiatry Rep. 2016;18(5):52. https://doi.org/10.1007/s11920-016-0694-1.
de la Asunción-Nadal V, Armenta S, Garrigues S, de la Guardia M. Identification and determination of synthetic cannabinoids in herbal products by dry film attenuated total reflectance-infrared spectroscopy. Talanta. 2017;167:344–51. https://doi.org/10.1016/j.talanta.2017.02.026.
Henriques S, Silva JP, Hsu K. Overview on new psychoactive substances in Portugal. Centro de Investigação e Estudos de Sociologia Instituto Universitário de Lisboa2018.
Liu C, Jia W, Hua Z, Qian Z. Identification and analytical characterization of six synthetic cannabinoids NNL-3, 5F-NPB-22-7N, 5F-AKB-48-7N, 5F-EDMB-PINACA, EMB-FUBINACA, and EG-018. Drug Test Anal. 2017;9(8):1251–61. https://doi.org/10.1002/dta.2160.
Marino MA, Voyer B, Cody RB, Dane AJ, Veltri M, Huang L. Rapid identification of synthetic cannabinoids in herbal incenses with DART-MS and NMR. J Forensic Sci. 2016;61(Suppl 1):S82–91. https://doi.org/10.1111/1556-4029.12932.
Gonçalves JL, Alves VL, Aguiar J, Teixeira HM, Câmara JS. Synthetic cathinones: an evolving class of new psychoactive substances. Crit Rev Toxicol. 2019;49(7):549–66. https://doi.org/10.1080/10408444.2019.1679087.
Angerer V, Bisel P, Moosmann B, Westphal F, Auwarter V. Separation and structural characterization of the new synthetic cannabinoid JWH-018 cyclohexyl methyl derivative "NE-CHMIMO" using flash chromatography, GC-MS, IR and NMR spectroscopy. Forensic Sci Int. 2016;266:e93–e8. https://doi.org/10.1016/j.forsciint.2016.05.031.
Dunne SJ, Rosengren-Holmberg JP. Quantification of synthetic cannabinoids in herbal smoking blends using NMR. Drug Test Anal. 2017;9(5):734–43. https://doi.org/10.1002/dta.2032.
Akutsu M, Sugie K-i, Saito K. Analysis of 62 synthetic cannabinoids by gas chromatography–mass spectrometry with photoionization. Forensic Toxicol 2017;35(1):94–103. doi:https://doi.org/10.1007/s11419-016-0342-9.
Carlsson A. Synthesis and spectroscopic characterization of emerging synthetic cannabinoids and cathinones [dissertation]. Sweden: Linköping University; 2016.
UNODC. Recommended methods for the identification and analysis of synthetic cannabinoid receptor agonists in seized materials. United Nations. 2013. https://www.unodc.org/unodc/en/scientists/recommended-methods-for-the-identification-and-analysis-of-synthetic-cannabinoid-receptor-agonists-in-seized-materials.html. Accessed 16 April 2019.
Uchiyama N, Kikura-Hanajiri R, Ogata J, Goda Y. Chemical analysis of synthetic cannabinoids as designer drugs in herbal products. Forensic Sci Int. 2010;198(1):31–8. https://doi.org/10.1016/j.forsciint.2010.01.004.
Ogata J, Uchiyama N, Kikura-Hanajiri R, Goda Y. DNA sequence analyses of blended herbal products including synthetic cannabinoids as designer drugs. Forensic Sci Int. 2013;227(1):33–41. https://doi.org/10.1016/j.forsciint.2012.09.006.
Namera A, Kawamura M, Nakamoto A, Saito T, Nagao M. Comprehensive review of the detection methods for synthetic cannabinoids and cathinones. Forensic Toxicol. 2015;33(2):175–94. https://doi.org/10.1007/s11419-015-0270-0.
Moran CL, Le V-H, Chimalakonda KC, Smedley AL, Lackey FD, Owen SN, et al. Quantitative measurement of JWH-018 and JWH-073 metabolites excreted in human urine. Anal Chem. 2011;83(11):4228–36. https://doi.org/10.1021/ac2005636.
Xing Y, Xu X, Liu X, Xu B, Ma Q, Lei H. Study on the mass fragmentation pathway of the synthetic cannabinoids JWH-018 and JWH-073. Int J Mass Spectrom. 2015;379:139–45. https://doi.org/10.1016/j.ijms.2015.01.007.
Dresen S, Ferreiros Bouzas N, Pütz M, Westphal F, Zimmermann R, Auwärter V. Monitoring of herbal mixtures potentially containing synthetic cannabinoids as psychoactive compounds. J Mass Spectrom : JMS. 2010;45:1186–94. https://doi.org/10.1002/jms.1811.
Thaxton A, Belal TS, Smith F, DeRuiter J, Abdel-Hay KM, Clark CR. GC–MS studies on the six naphthoyl-substituted 1-n-pentyl-indoles: JWH-018 and five regioisomeric equivalents. Forensic Sci Int. 2015;252:107–13. https://doi.org/10.1016/j.forsciint.2015.04.023.
Grabenauer M, Krol WL, Wiley JL, Thomas BF. Analysis of synthetic cannabinoids using high-resolution mass spectrometry and mass defect filtering: implications for nontargeted screening of designer drugs. Anal Chem. 2012;84(13):5574–81. https://doi.org/10.1021/ac300509h.
Thaxton A, Belal TS, Smith F, DeRuiter J, Abdel-Hay KM, Clark CR. Mass spectral studies on 1-n-pentyl-3-(1-naphthoyl)indole (JWH-018), three deuterium-labeled analogues and the inverse isomer 1-naphthoyl-3-n-pentylindole. Rapid Commun Mass Spectrom. 2015;29(9):871–7. https://doi.org/10.1002/rcm.7171.
Harris DN, Hokanson S, Miller V, Jackson GP. Fragmentation differences in the EI spectra of three synthetic cannabinoid positional isomers: JWH-250, JWH-302, and JWH-201. Int J Mass Spectrom. 2014;368:23–9. https://doi.org/10.1016/j.ijms.2014.05.005.
Ma Q, Bai H, Li W, Wang C, Cooks RG, Ouyang Z. Rapid analysis of synthetic cannabinoids using a miniature mass spectrometer with ambient ionization capability. Talanta. 2015;142:190–6. https://doi.org/10.1016/j.talanta.2015.04.044.
Uchiyama N, Kawamura M, Kikura-Hanajiri R, Goda Y. Identification of two new-type synthetic cannabinoids, N-(1-adamantyl)-1-pentyl-1H-indole-3-carboxamide (APICA) and N-(1-adamantyl)-1-pentyl-1H-indazole-3-carboxamide (APINACA), and detection of five synthetic cannabinoids, AM-1220, AM-2233, AM-1241, CB-13 (CRA-13), and AM-1248, as designer drugs in illegal products. Forensic Toxicol. 2012;30(2):114–25. https://doi.org/10.1007/s11419-012-0136-7.
Asada A, Doi T, Tagami T, Takeda A, Sawabe Y. Isomeric discrimination of synthetic cannabinoids by GC-EI-MS: 1-adamantyl and 2-adamantyl isomers of N-adamantyl carboxamides. Drug Test Anal. 2017;9(3):378–88. https://doi.org/10.1002/dta.2124.
Gandhi AS, Zhu M, Pang S, Wohlfarth A, Scheidweiler KB, Liu H-F, et al. First characterization of AKB-48 metabolism, a novel synthetic cannabinoid, using human hepatocytes and high-resolution mass spectrometry. AAPS J. 2013;15(4):1091–8. https://doi.org/10.1208/s12248-013-9516-0.
Jia W, Meng X, Qian Z, Hua Z, Li T, Liu C. Identification of three cannabimimetic indazole and pyrazole derivatives, APINACA 2H-indazole analogue, AMPPPCA, and 5F-AMPPPCA. Drug Test Anal. 2017;9(2):248–55. https://doi.org/10.1002/dta.1967.
Banister SD, Stuart J, Kevin RC, Edington A, Longworth M, Wilkinson SM, et al. Effects of bioisosteric fluorine in synthetic cannabinoid designer drugs JWH-018, AM-2201, UR-144, XLR-11, PB-22, 5F-PB-22, APICA, and STS-135. ACS Chem Neurosci. 2015;6(8):1445–58. https://doi.org/10.1021/acschemneuro.5b00107.
Arar SH, ALSoufi H, Abu-Nameh E, Deeb A, Habahbeh A. Screening of (aminoalkylindols) cannabinoids in smoking products by GC-EI/MS in Jordan: liquid-liquid extraction optimization. J Egypt J Chem. 2020;63(2):373–87. https://doi.org/10.21608/ejchem.2019.11591.1737.
Thomas BF, Lefever TW, Cortes RA, Grabenauer M, Kovach AL, Cox AO, et al. Thermolytic degradation of synthetic cannabinoids: chemical exposures and pharmacological consequences. J Pharmacol Exp Ther. 2017;361(1):162–71. https://doi.org/10.1124/jpet.116.238717.
Hiang Ng C, Ani SAC, Mustafa K, Desa S, Sulaiman M. Identification of synthetic cannabinoids 5F-ADB and XLR-11 in seized sample in Penang, Malaysia. Malaysian J Forensic Sci. 2019;9(1):1–6.
Choi H, Heo S, Kim E, Hwang BY, Lee C, Lee J. Identification of (1-pentylindol-3-yl)-(2,2,3,3-tetramethylcyclopropyl)methanone and its 5-pentyl fluorinated analog in herbal incense seized for drug trafficking. Forensic Toxicol. 2013;31(1):86–92. https://doi.org/10.1007/s11419-012-0170-5.
Atwood B, Lee D, Straiker A, Widlanski T, Mackie K. CP47,497-C8 and JWH073, commonly found in ‘Spice’ herbal blends, are potent and efficacious CB1 cannabinoid receptor agonists. Eur J Pharmacol. 2011;659:139–45. https://doi.org/10.1016/j.ejphar.2011.01.066.
EMCDDA. EMCDDA, Understanding the ‘Spice’ phenomenon. 2009. http://www.emcdda.europa.eu/publications/thematic-papers/understanding-spice-phenomenon_en. Accessed 23 February 2019.
Uchiyama N, Kikura-Hanajiri R, Shoda T, Fukuhara K, Goda Y. Isomeric analysis of synthetic cannabinoids detected as designer drugs. Yakugaku Zasshi. 2011;131(7):1141–7. https://doi.org/10.1248/yakushi.131.1141.
Gwak S, Arroyo-Mora LE, Almirall JR. Qualitative analysis of seized synthetic cannabinoids and synthetic cathinones by gas chromatography triple quadrupole tandem mass spectrometry. Drug Test Anal. 2015;7(2):121–30. https://doi.org/10.1002/dta.1667.
UNODC. Synthetic cannabinoids in herbal products. 2011. https://www.unodc.org/documents/scientific/Synthetic_Cannabinoids.pdf. Accessed 10 March 2019.
Zawilska JB, Wojcieszak J. Spice/K2 drugs – more than innocent substitutes for marijuana. Int J Neuropsychopharmacol. 2014;17(3):509–25. https://doi.org/10.1017/S1461145713001247.
Duffy B, Li L, Lu S, Durocher L, Dittmar M, Delaney-Baldwin E, et al. Analysis of cannabinoid-containing fluids in illicit vaping cartridges recovered from pulmonary injury patients: identification of vitamin E acetate as a major diluent. Toxics. 2020;8(1):8. https://doi.org/10.3390/toxics8010008.
Naqi H, Woodman T, Husbands S, Blagbrough I. 19F and 1H quantitative-NMR spectroscopic analysis of fluorinated third-generation synthetic cannabinoids. Anal Methods. 2019. https://doi.org/10.1039/C9AY00814D.
Amato J, Iaccarino N, Pagano B, Compagnone V, Rosa F, Peluso G et al. NMR assignment of N-(1-adamantyl)-1-pentyl-1H-indazole-3-carboxamide seized as herbal incense for the first time in Italy. Journal of Forensic Science & Criminology. 2014;2(1). doi:https://doi.org/10.15744/2348-9804.1.403.
Fowler F, Voyer B, Marino M, Finzel J, Veltri M, Wachter N, et al. Rapid screening and quantification of synthetic cannabinoids in herbal products with NMR spectroscopic methods. Anal Methods. 2015;7. https://doi.org/10.1039/C5AY01754H.
Assemat G, Dubocq F, Balayssac S, Lamoureux C, Malet-Martino M, Gilard V. Screening of “spice” herbal mixtures: from high-field to low-field proton NMR. Forensic Sci Int. 2017;279:88–95. https://doi.org/10.1016/j.forsciint.2017.08.006.
Langer N, Lindigkeit R, Schiebel H-M, Ernst L, Beuerle T. Identification and quantification of synthetic cannabinoids in ‘spice-like’ herbal mixtures: a snapshot of the German situation in the autumn of 2012. Drug Test Anal. 2014;6(1–2):59–71. https://doi.org/10.1002/dta.1499.
Simolka K, Lindigkeit R, Schiebel H-M, Papke U, Ernst L, Beuerle T. Analysis of synthetic cannabinoids in “spice-like” herbal highs: snapshot of the German market in summer 2011. Anal Bioanal Chem. 2012;404(1):157–71. https://doi.org/10.1007/s00216-012-6122-4.
Shevyrin V, Melkozerov V, Nevero A, Eltsov O, Morzherin Y, Shafran Y. Identification and analytical properties of new synthetic cannabimimetics bearing 2,2,3,3-tetramethylcyclopropanecarbonyl moiety. Forensic Sci Int. 2013;226(1):62–73. https://doi.org/10.1016/j.forsciint.2012.12.009.
Sekuła K, Zuba D, Stanaszek R. Identification of naphthoylindoles acting on cannabinoid receptors based on their fragmentation patterns under ESI-QTOFMS. J Mass Spectrom. 2012;47(5):632–43. https://doi.org/10.1002/jms.3004.
Acknowledgments
The authors would like to thank LPC-PJ for their collaboration in providing seized herbal products.
Funding
This work was supported by FCT-Fundação para a Ciência e a Tecnologia through the CQM Base Fund (UIDB/00674/2020, and Programmatic Fund - UIDP/00674/2020), and the PhD fellowships SFRH/BD/117426/2016 and SFRH/BD/116895/2016 granted to Vera Alves and João Gonçalves respectively, and through Madeira 14-20 Program, project PROEQUIPRAM - Reforço do Investimento em Equipamentos e Infraestruturas Científicas na RAM (M1420-01-0145-FEDER-000008), and by ARDITI-Agência Regional para o Desenvolvimento da Investigação Tecnologia e Inovação, through the project M1420-01-0145-FEDER-000005 - Centro de Química da Madeira - CQM+ (Madeira 14–20 Program).
Author information
Authors and Affiliations
Contributions
Vera Alves: Investigation, methodology, data curation, writing—original draft. João Gonçalves: Visualization, data curation, methodology. Joselin Aguiar: Investigation and methodology. Helena Teixeira: Conceptualization, supervision, review and editing. José S. Câmara: Supervision, resources, project administration, funding acquisition, supervision, review and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(PDF 478 kb)
Rights and permissions
About this article
Cite this article
Alves, V.L., Gonçalves, J.L., Aguiar, J. et al. Highly sensitive screening and analytical characterization of synthetic cannabinoids in nine different herbal mixtures. Anal Bioanal Chem 413, 2257–2273 (2021). https://doi.org/10.1007/s00216-021-03199-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-021-03199-6