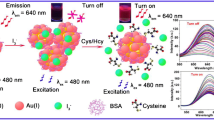
Abstract
Balanced level of hemin in the body is fundamentally important for normal human organ function. Therefore, environmentally benign, stable, and fluorescent metal nanoclusters (NCs) for selective and sensitive detection of hemin have been investigated and reported. Herein, highly orange red emissive gold NCs are successfully synthesized using glutathione as a reducing and stabilizing agent (GSH-Au NCs). The clusters are characterized using various techniques like Fourier transform infrared spectroscopy (FTIR), transmission electron microscopy (TEM), UV-vis spectroscopy, and fluorescence spectrometer. The fluorescence intensity of as-synthesized Au NCs strongly quenched upon addition of different concentrations of hemin. The decrease in fluorescence intensity of GSH-Au NCs has been applied for determination of hemin concentration in the linear range from 1 to 25 nM with a low limit of detection (LOD) of 0.43 nM. The method was also successfully applied for quantification of hemin in human serum sample. In view of this reality, the system can be considered as a possible strategy and excellent platform for determination of hemin in various areas of application.
Graphical abstract








Similar content being viewed by others
References
Lee JM, Lee WH, Kay HY, E-s K, Moon A, Kim SG. Hemin, an iron-binding porphyrin, inhibits HIF-1α induction through its binding with heat shock protein 90. Int J Cancer. 2012;130:716–27. https://doi.org/10.1002/ijc.26075.
Ni P, Chen C, Jiang Y, Lu Y, Chen W. A simple and sensitive fluorescent assay for hemin detection based on artemisinin-thiamine. Sensor Actuat B: Chem. 2018;273:198–203. https://doi.org/10.1016/j.snb.2018.06.052.
Gao W, Wang C, Muzyka K, Kitte SA, Li J, Zhang W, et al. Artemisinin-luminol chemiluminescence for forensic bloodstain detection using a smart phone as a detector. Anal Chem. 2017;89:6160–5. https://doi.org/10.1021/acs.analchem.7b01000.
Fereja TH, Kitte SA, Gao W, Yuan F, Snizhko D, Qi L, et al. Artesunate-luminol chemiluminescence system for the detection of hemin. Talanta. 2019;204:379–85. https://doi.org/10.1016/j.talanta.2019.06.007.
Jahromi Z, Shamspur T, Mostafavi A, Mohamadi M. Separation and preconcentration of hemin from serum samples followed by voltammetric determination. J Mol Liq. 2017;242:91–7. https://doi.org/10.1016/j.molliq.2017.07.008.
Han J, Zhou Z, Bu X, Zhu S, Zhang H, Sun H, et al. Employing aqueous CdTe quantum dots with diversified surface functionalities to discriminate between heme (Fe(ii)) and hemin (Fe(iii)). Analyst. 2013;138:3402–8. https://doi.org/10.1039/C3AN00310H.
Ikariyama Y, Suzuki S, Aizawa M. Luminescence immunoassay of human serum albumin with hemin as labeling catalyst. Anal Chem. 1982;54:1126–9. https://doi.org/10.1021/ac00244a026.
Han S, Liu E, Li H. Flow injection chemiluminescence determination of hemin using the rhodamine B–H2O2–NaOH system. Microchim Acta. 2005;149:281–6. https://doi.org/10.1007/s00604-004-0312-5.
Sawicki KT, Chang H-C, Ardehali H. Role of heme in cardiovascular physiology and disease. J Am Heart Assoc. 2015;4:e001138–e. https://doi.org/10.1161/JAHA.114.001138.
Kang BH, Li N, Liu SG, Li NB, Luo HQ. A label-free, highly sensitive and selective detection of hemin based on the competition between hemin and protoporphyrin IX binding to G-quadruplexes. Anal Sci. 2016;32:887–92. https://doi.org/10.2116/analsci.32.887.
Rong X-J, Tang M-Q, He J-H, Cai X-C, Kang Y-T, Mao Y. RP-HPLC determination of related substances of hemin. Chin J Pharl Anal. 2013;33:274–7.
Lombardo ME, Araujo LS, Ciccarelli AB, Batlle A. A spectrophotometric method for estimating hemin in biological systems. Anal Biochem. 2005;341:199–203. https://doi.org/10.1016/j.ab.2004.11.002.
Takahashi S, Bhowmik S, Sugimoto N. Volumetric analysis of formation of the complex of G-quadruplex DNA with hemin using high pressure. J Inorg Biochem. 2017;166:199–207. https://doi.org/10.1016/j.jinorgbio.2016.08.011.
Benavides J, Quijada-Garrido I, García O. The synthesis of switch-off fluorescent water-stable copper nanocluster Hg2+ sensors via a simple one-pot approach by an in situ metal reduction strategy in the presence of a thiolated polymer ligand template. Nanoscale. 2020;12(2):944–55. https://doi.org/10.1039/C9NR08439H.
Feng X, Zhang J, Wang J, Han A, Fang G, Liu J, et al. The stabilization of fluorescent copper nanoclusters by dialdehyde cellulose and their use in mercury ion sensing. Anal Methods. 2020;12(24):3130–6. https://doi.org/10.1039/D0AY00657B.
Zhao Y, Zhou H, Zhang S, Xu J. The synthesis of metal nanoclusters and their applications in bio-sensing and imaging. Methods and Applications in Fluorescence. 2019;8(1):012001. https://doi.org/10.1088/2050-6120/ab57e7.
Chen Y, Dong X, Zheng Y, Wang Y, Guo Z, Jiang H, et al. A novel turn-on fluorescent sensor for the sensitive detection of glutathione via gold nanocluster preparation based on controllable ligand-induced etching. Analyst. 2020;145(12):4265–75. https://doi.org/10.1039/D0AN00807A.
Zhang G, Li Y, Xu J, Zhang C, Shuang S, Dong C, et al. Glutathione-protected fluorescent gold nanoclusters for sensitive and selective detection of Cu2+. Sensor Actuat B: Chem. 2013;183:583–8. https://doi.org/10.1016/j.snb.2013.04.023.
Li Y, Wen Q-L, Liu A-Y, Long Y, Liu P, Ling J, et al. One-pot synthesis of green-emitting gold nanoclusters as a fluorescent probe for determination of 4-nitrophenol. Microchim Acta. 2020;187:106. https://doi.org/10.1007/s00604-019-4090-5.
Jia Y, Sun T, Jiang Y, Sun W, Zhao Y, Xin J, et al. Green, fast, and large-scale synthesis of highly fluorescent au nanoclusters for Cu2+ detection and temperature sensing. Analyst. 2018;143(21):5145–50. https://doi.org/10.1039/C8AN01617H.
Liu R, Duan S, Bao L, Wu Z, Zhou J, Yu R. Photonic crystal enhanced gold-silver nanoclusters fluorescent sensor for Hg2+ ion. Anal Chim Acta. 2020;1114:50–7. https://doi.org/10.1016/j.aca.2020.04.011.
Zhang G, Xiang M, Kong R-M, Qu F. Fluorescent and colorimetric determination of glutathione based on the inner filter effect between silica nanoparticle–gold nanocluster nanocomposites and oxidized 3,3′,5,5′-tetramethylbenzidine. Analyst. 2020;145(19):6254–61. https://doi.org/10.1039/D0AN01392G.
Halawa MI, Lai J, Xu G. Gold nanoclusters: synthetic strategies and recent advances in fluorescent sensing. Mater Today Nano. 2018;3:9–27. https://doi.org/10.1016/j.mtnano.2018.11.001.
Wang J, Lin X, Su L, Yin J, Shu T, Zhang X. Chemical etching of pH-sensitive aggregation-induced emission-active gold nanoclusters for ultra-sensitive detection of cysteine. Nanoscale. 2019;11(1):294–300. https://doi.org/10.1039/C8NR08526A.
Bian R-X, Wu X-T, Chai F, Li L, Zhang L-Y, Wang T-T, et al. Facile preparation of fluorescent Au nanoclusters-based test papers for recyclable detection of Hg2+ and Pb2+. Sensor Actuat B: Chem. 2017;241:592–600. https://doi.org/10.1016/j.snb.2016.10.120.
Deng H-H, Deng Q, Li K-L, Zhuang Q-Q, Zhuang Y-B, Peng H-P, et al. Fluorescent gold nanocluster-based sensor for detection of alkaline phosphatase in human osteosarcoma cells. Spectrochim Acta A. 2020;229:117875. https://doi.org/10.1016/j.saa.2019.117875.
Yahia-Ammar A, Sierra D, Mérola F, Hildebrandt N, Le Guével X. Self-assembled gold nanoclusters for bright fluorescence imaging and enhanced drug delivery. ACS Nano. 2016;10:2591–9. https://doi.org/10.1021/acsnano.5b07596.
Chang H-C, Ho J-aA. Gold nanocluster-assisted fluorescent detection for hydrogen peroxide and cholesterol based on the inner filter effect of gold nanoparticles. Anal Chem. 2015;87:10362–7. https://doi.org/10.1021/acs.analchem.5b02452.
Alkilany A, Alsotari S, Alkawareek M, Abulateefeh S. Facile hydrophobication of glutathione-protected gold nanoclusters and encapsulation into poly(lactide-co-glycolide) nanocarriers. Sci Rep. 2019;9. https://doi.org/10.1038/s41598-019-47543-4.
Govindaraju S, Ankireddy SR, Viswanath B, Kim J, Yun K. Fluorescent gold nanoclusters for selective detection of dopamine in cerebrospinal fluid. Sci Rep. 2017;7:40298. https://doi.org/10.1038/srep40298.
Thakur NS, Mandal N, Banerjee UC. Esterase-mediated highly fluorescent gold nanoclusters and their use in ultrasensitive detection of mercury: synthetic and mechanistic aspects. ACS Omega. 2018;3(12):18553–62. https://doi.org/10.1021/acsomega.8b02505.
Chen Y-S, Kamat PV. Glutathione-capped gold nanoclusters as photosensitizers. Visible light-induced hydrogen generation in neutral water. J Am Chem Soc. 2014;136(16):6075–82. https://doi.org/10.1021/ja5017365.
Lu F, Yang H, Tang Y, Yu C-J, Wang G, Yuan Z, et al. 11-Mercaptoundecanoic acid capped gold nanoclusters with unusual aggregation-enhanced emission for selective fluorometric hydrogen sulfide determination. Microchim Acta. 2020;187(4):200. https://doi.org/10.1007/s00604-020-4159-1.
Zhang H, Liu Q, Wang T, Yun Z, Li G, Liu J, et al. Facile preparation of glutathione-stabilized gold nanoclusters for selective determination of chromium (III) and chromium (VI) in environmental water samples. Anal Chim Acta. 2013;770:140–6. https://doi.org/10.1016/j.aca.2013.01.042.
Feng B, Xing Y, Lan J, Su Z, Wang F. Synthesis of MUC1 aptamer-stabilized gold nanoclusters for cell-specific imaging. Talanta. 2020;212:120796. https://doi.org/10.1016/j.talanta.2020.120796.
Qian H, Zhu M, Wu Z, Jin R. Quantum sized gold nanoclusters with atomic precision. Acc Chem Res. 2012;45(9):1470–9. https://doi.org/10.1021/ar200331z.
Sang F, Zhang X, Shen F. Fluorescent methionine-capped gold nanoclusters for ultra-sensitive determination of copper(II) and cobalt(II), and their use in a test strip. Microchim Acta. 2019;186(6):373. https://doi.org/10.1007/s00604-019-3428-3.
Talavera C, Kamat PV. Glutathione-capped gold nanoclusters: photoinduced energy transfer and singlet oxygen generation. J Chem Sci. 2018;130:143. https://doi.org/10.1007/s12039-018-1549-6.
Gao L, Xiao Y, Wang Y, Chen X, Zhou B, Yang X. A carboxylated graphene and aptamer nanocomposite-based aptasensor for sensitive and specific detection of hemin. Talanta. 2015;132:215–21. https://doi.org/10.1016/j.talanta.2014.09.010.
Wu Z, Jin R. On the ligand’s role in the fluorescence of gold nanoclusters. Nano Lett. 2010;10(7):2568–73. https://doi.org/10.1021/nl101225f.
Su X, Jiang H, Wang X. Thiols-induced rapid photoluminescent enhancement of glutathione-capped gold nanoparticles for intracellular thiols imaging applications. Anal Chem. 2015;87(20):10230–6. https://doi.org/10.1021/acs.analchem.5b02559.
Khataee A, Jalili R, Dastborhan M, Karimi A, Ebadi Fard Azar A. Ratiometric visual detection of tetracycline residues in milk by framework-enhanced fluorescence of gold and copper nanoclusters. Spectrochim Acta A. 2020;242:118715. https://doi.org/10.1016/j.saa.2020.118715.
Meng L, Wu Y, Xu N. Gold nanoclusters fluorescence probe for monitoring chloramphenicol and study of two-dimensional correlation fluorescence spectroscopy. J Mol Struc. 2021;1223:128875. https://doi.org/10.1016/j.molstruc.2020.128875.
Liu G, Feng D-Q, Hua D, Liu T, Qi G, Wang W. Fluorescence enhancement of terminal amine assembled on gold nanoclusters and its application to ratiometric lysine detection. Langmuir. 2017;33(51):14643–8. https://doi.org/10.1021/acs.langmuir.7b02614.
Soleilhac A, Bertorelle F, Comby-Zerbino C, Chirot F, Calin N, Dugourd P, et al. Size characterization of glutathione-protected gold nanoclusters in the solid, liquid and gas phases. J Phys Chem C. 2017;121:27733–40. https://doi.org/10.1021/acs.jpcc.7b09500.
Xu X, Ji J, Chen P, Wu J, Jin Y, Zhang L, et al. Salt-induced gold nanoparticles aggregation lights up fluorescence of DNA-silver nanoclusters to monitor dual cancer markers carcinoembryonic antigen and carbohydrate antigen 125. Anal Chim Acta. 2020;1125:41–9. https://doi.org/10.1016/j.aca.2020.05.027.
Wang Y, Chen T, Zhang Z, Ni Y. Cytidine-stabilized copper nanoclusters as a fluorescent probe for sensing of copper ions and hemin. RSC Adv. 2018;8(17):9057–62. https://doi.org/10.1039/C7RA11383H.
Jiang H, Zhang W, Li J, Nie L, Wu K, Duan H, et al. Inner-filter effect based fluorescence-quenching immunochromotographic assay for sensitive detection of aflatoxin B1 in soybean sauce. Food Control. 2018;94:71–6. https://doi.org/10.1016/j.foodcont.2018.06.030.
Goldstein L, Teng Z-P, Zeserson E, Patel M, Regan RF. Hemin induces an iron-dependent, oxidative injury to human neuron-like cells. J Neurosci Res. 2003;73(1):113–21. https://doi.org/10.1002/jnr.10633.
Gao S, Wang R, Bi Y, Qu H, Chen Y, Zheng L. Identification of frozen/thawed beef based on label-free detection of hemin (Iron Porphyrin) with solution-gated graphene transistor sensors. Sensor Actuat B: Chem. 2020;305:127167. https://doi.org/10.1016/j.snb.2019.127167.
Guo Z, Li B, Zhang Y, Zhao Q, Zhao J, Li L, et al. Acid-treated graphitic carbon nitride nanosheets as fluorescence probe for detection of hemin. ChemistrySelect. 2019;4(28):8178–82. https://doi.org/10.1002/slct.201901841.
Shi Y, Huang WT, Luo HQ, Li NB. A label-free DNA reduced graphene oxide-based fluorescent sensor for highly sensitive and selective detection of hemin. Chem Commun. 2011;47(16):4676–8. https://doi.org/10.1039/C0CC05518B.
Zhang Z, Hu B, Zhuang Q, Wang Y, Luo X, Xie Y, et al. Green synthesis of fluorescent nitrogen–sulfur co-doped carbon dots from scallion leaves for hemin sensing. Anal Lett. 2020;53(11):1704–18. https://doi.org/10.1080/00032719.2020.1716782.
Baruah U, Gogoi N, Majumdar G, Chowdhury D. Capped fluorescent carbon dots for detection of hemin: role of number of -OH groups of capping agent in fluorescence quenching. Sci World J. 2013;2013:529159. https://doi.org/10.1155/2013/529159.
Shekari Z, Zare HR, Falahati A. An ultrasensitive aptasensor for hemin and hemoglobin based on signal amplification via electrocatalytic oxygen reduction. Anal Biochem. 2017;518:102–9. https://doi.org/10.1016/j.ab.2016.11.016.
Neugebauer U, März A, Henkel T, Schmitt M, Popp J. Spectroscopic detection and quantification of heme and heme degradation products. Anal Bioanal Chem. 2012;404:2819–29. https://doi.org/10.1007/s00216-012-6288-9.
Funding
This work was funded by the National Natural Science Foundation of China (Nos. 21575134, 21633008, 21773224), National Key Research and Development Plan (2016YFA0203200), K. C. Wong Education Foundation, Natural Science Foundation of Guangxi Province (2019GXNSFGA245003), and Chinese Government Scholarship under China Scholarships council (CSC).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fereja, S.L., Fang, Z., Li, P. et al. “Turn-off” sensing probe based on fluorescent gold nanoclusters for the sensitive detection of hemin. Anal Bioanal Chem 413, 1639–1649 (2021). https://doi.org/10.1007/s00216-020-03126-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-020-03126-1