Abstract
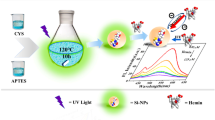
A new type of fluorescent silicon nanoparticles (SiNPs) were prepared via a facile one-pot hydrothermal method by using N-[3-(trimethoxysilyl)propyl]-ethylenediamine (DAMO) and glucose as reagents, and were subsequently applied to construct a ratiometric fluorescence assay for sensitive and rapid determination of xanthine in human serum. Two catalytic oxidation reactions were employed to induce a fluorescence response of the testing system towards xanthine. Under the catalysis of xanthine oxidase (XOD), xanthine in serum samples was oxidized and produced hydrogen peroxide (H2O2). By utilizing o-phenylenediamine (OPD) as the substrate for horseradish peroxidase (HRP) in the presence of H2O2, fluorescent 2,3-diaminophenazine (DAP) was finally generated. A ratiometric fluorescence assay for xanthine was established by determining the ratio of the green-yellow fluorescence emission of DAP and the blue fluorescence emitted from SiNPs under the inner filter effect (IFE) of DAP. Instead of traditional multi-step procedures for adding reacting reagents to the testing solution, all the reaction reagents were mixed with serum samples in a single step for this assay to shorten the total reaction time. This assay demonstrates superiority over a solo DAP fluorescence-based assay as well as other reported methods, with excellent sensitivity and reduced testing time. The strategies proposed in this work for both synthesis and application of fluorescent SiNPs can be used in future fabrication of novel fluorescent probes, especially for sensing biological metabolites involved in H2O2-generation or consumption reactions.
Graphical abstract










Similar content being viewed by others
References
Luo A, Lian Q, An Z, Li Z, Guo Y, Zhang D, et al. Simultaneous determination of uric acid, xanthine and hypoxanthine based on sulfonic groups functionalized nitrogen-doped graphene. J Electroanal Chem. 2015;756:22–9.
Bas SZ, Gulce H, Yildiz S, Gulce A. Amperometric biosensors based on deposition of gold and platinum nanoparticles on polyvinylferrocene modified electrode for xanthine detection. Talanta. 2011;87:189–96.
Wang Y, Deng M, Deng B, Ye L, Fei X, Huang Z. Study on the diagnosis of gout with xanthine and hypoxanthine. J Clin Lab Anal. 2019;33(5):e22868.
Pierini GD, Robledo SN, Zon MA, Di Nezio MS, Granero AM, Fernández H. Development of an electroanalytical method to control quality in fish samples based on an edge plane pyrolytic graphite electrode. Simultaneous determination of hypoxanthine, xanthine and uric acid. Microchem J. 2018;138:58–64.
Dervisevic M, Dervisevic E, Cevik E, Senel M. Novel electrochemical xanthine biosensor based on chitosan-polypyrrole-gold nanoparticles hybrid bio-nanocomposite platform. J Food Drug Anal. 2017;25(3):510–9.
Li Z, Liu X, Liang XH, Zhong J, Guo L, Fu F. Colorimetric determination of xanthine in urine based on peroxidase-like activity of WO3 nanosheets. Talanta. 2019;204:278–84.
Bory C, CC RB. Comparison of capillary electrophoretic and liquid chromatographic determination of hypoxanthine and xanthine for the diagnosis of xanthinuria. J Chromatogr A. 1996;730:329–31.
Cooper N, Khosravan R, Erdmann C, Fiene J, Lee JW. Quantification of uric acid, xanthine and hypoxanthine in human serum by HPLC for pharmacodynamic studies. J Chromatogr B Anal Technol Biomed Life Sci. 2006;837(1–2):1–10.
Kant R, Tabassum R, Gupta BD. Xanthine oxidase functionalized Ta2O5 nanostructures as a novel scaffold for highly sensitive SPR based fiber optic xanthine sensor. Biosens Bioelectron. 2018;99:637–45.
Ma Y, Cen Y, Sohail M, Xu G, Wei F, Shi M, et al. A ratiometric fluorescence universal platform based on N, Cu codoped carbon dots to detect metabolites participating in H2O2-generation reactions. ACS Appl Mater Interfaces. 2017;9(38):33011–9.
Menon S, Girish Kumar K. A fluorescent biosensor for the determination of xanthine in tea and coffee via enzymatically generated uric acid. LWT. 2017;86:8–13.
Zhang H, Chen Y, Liang M, Xu L, Qi S, Chen H, et al. Solid-phase synthesis of highly fluorescent nitrogen-doped carbon dots for sensitive and selective probing ferric ions in living cells. Anal Chem. 2014;86(19):9846–52.
Qian Z, Chai L, Tang C, Huang Y, Chen J, Feng H. Carbon quantum dots-based recyclable real-time fluorescence assay for alkaline phosphatase with adenosine triphosphate as substrate. Anal Chem. 2015;87(5):2966–73.
Peng F, Su Y, Zhong Y, Fan C, Lee S, He Y. Silicon nanomaterials platform for bioimaging, biosensing, and cancer therapy. Acc Chem Res. 2014;47(2):612–23.
Folarin E, Yong KT, Roy I, Hu R, Law WC, Zhao WW, et al. In vivo targeted cancer imaging, sentinel lymph node mapping and multi-channel imaging with biocompatible silicon nanocrystals. ACS Nano. 2011;5(1):413–23.
Li Q, Luo TY, Zhou M, Abroshan H, Huang J, Kim HJ, et al. Silicon nanoparticles with surface nitrogen: 90% quantum yield with narrow luminescence bandwidth and the ligand structure based energy law. ACS Nano. 2016;10(9):8385–93.
Yi Y, Zhu G, Liu C, Huang Y, Zhang Y, Li H, et al. A label-free silicon quantum dots-based photoluminescence sensor for ultrasensitive detection of pesticides. Anal Chem. 2013;85(23):11464–70.
Li D, Jiang Y, Chen S, Zhao Q, Zhang Y, Wang W, et al. A simple and sensitive assay of alkaline phosphatase activity in serum by fluorescent silicon nanoparticles based on inner filter effect. Sensors Actuators B Chem. 2020;307:127589.
Chen S, Yu YL, Wang JH. Inner filter effect-based fluorescent sensing systems: a review. Anal Chim Acta. 2018;999:13–26.
Han Y, Chen Y, Feng J, Liu J, Ma S, Chen X. One-pot synthesis of fluorescent silicon nanoparticles for sensitive and selective determination of 2,4,6-trinitrophenol in aqueous solution. Anal Chem. 2017;89(5):3001–8.
Wu F, Zhang X, Kai S, Zhang M, Wang H, Myers JN, et al. One-step synthesis of superbright water-soluble silicon nanoparticles with photoluminescence quantum yield exceeding 80%. Adv Mater Interfaces. 2015;2(16):1500360.
Song W, Duan W, Liu Y, Ye Z, Chen Y, Chen H, et al. Ratiometric detection of intracellular lysine and pH with one-pot synthesized dual emissive carbon dots. Anal Chem. 2017;89(24):13626–33.
Nsanzamahoro S, Mutuyimana FP, Han Y, Ma S, Na M, Liu J, et al. Highly selective and sensitive detection of catechol by one step synthesized highly fluorescent and water-soluble silicon nanoparticles. Sensors Actuators B Chem. 2019;281:849–56.
Han Y, Chen Y, Feng J, Na M, Liu J, Ma Y, et al. Investigation of nitrogen content effect in reducing agent to prepare wavelength controllable fluorescent silicon nanoparticles and its application in detection of 2-nitrophenol. Talanta. 2019;194:822–9.
Ma SD, Chen YL, Feng J, Liu JJ, Zuo XW, Chen XG. One-step synthesis of water-dispersible and biocompatible silicon nanoparticles for selective heparin sensing and cell imaging. Anal Chem. 2016;88(21):10474–81.
Chen X, Zhang X, Xia LY, Wang HY, Chen Z, Wu FG. One-step synthesis of ultrasmall and ultrabright organosilica nanodots with 100% photoluminescence quantum yield: long-term lysosome imaging in living, fixed, and permeabilized cells. Nano Lett. 2018;18(2):1159–67.
Zhang M, Su R, Zhong J, Fei L, Cai W, Guan Q, et al. Red/orange dual-emissive carbon dots for pH sensing and cell imaging. Nano Res. 2019;12(4):815–21.
Wang X, Cao L, Yang ST, Lu F, Meziani MJ, Tian L, et al. Bandgap-like strong fluorescence in functionalized carbon nanoparticles. Angew Chem Int Ed Engl. 2010;49(31):5310–4.
Sharma B, Tanwar S, Sen T. One pot green synthesis of Si quantum dots and catalytic Au nanoparticle–Si quantum dot nanocomposite. ACS Sustain Chem Eng. 2019;7(3):3309–18.
Liu J, Chen Y, Wang W, Feng J, Liang M, Ma S, et al. “Switch-on” fluorescent sensing of ascorbic acid in food samples based on carbon quantum dots-MnO2 probe. J Agric Food Chem. 2016;64(1):371–80.
Gauthier TD, Shane EC, Guerin WF, Rudolf Seitz W, Grant CL. Fluorescence quenching method for determining equilibrium constants for polycyclic aromatic hydrocarbons binding to dissolved humic materials. Environ Sci Technol. 1986;20:1162–6.
Kubista M, Sjoback R, Eriksson S, Albinsson B. Experimental correction for the inner-filter effect in fluorescence spectra. Analyst. 1994;119:417–9.
Boulieu R, Bory C, Baltassat P, Gonnet C. Hypoxanthine and xanthine levels determined by high-performance liquid chromatography in plasma, erythrocyte, and urine samples from healthy subjects: the problem of hypoxanthine level evolution as a function of time. Anal Biochem. 1983;129(2):398–404.
Wu X, Chen T, Wang J, Yang G. Few-layered MoSe2 nanosheets as an efficient peroxidase nanozyme for highly sensitive colorimetric detection of H2O2 and xanthine. J Mater Chem B. 2018;6(1):105–11.
Acknowledgements
This work was supported by the State Scholarship Fund of China (No. 201806175003), Industrialization Project of the Education Department of Jilin Province of China (No. JJKH20200944KJ), the National Natural Science Foundation of China (31771093), the Project of International Collaboration of Jilin Province (201180414085GH), the Fundamental Research Funds for the Central Universities, the Program for JLU Science and Technology Innovative Research Team (2017TD-27, 2019TD-36), and “The 13th Five-Year” industrialization project of Jilin Education Department (JJKH20190109KJ, 2019C023, 2018SCZ038).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
All applicable international, national, and/or institutional guidelines for the collection and use of human blood and serum samples were followed.
Human ethics
The study was approved by the Institutional Ethics Committee of China-Japan Union Hospital of Jilin University, Changchun, China (No. 2019040811).
Conflict of interest
The authors declare that there is no conflict of interest regarding the publication of this article.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(PDF 766 kb)
Rights and permissions
About this article
Cite this article
Li, D., Chen, F., Li, N. et al. Sensitive ratiometric fluorescence assay for detecting xanthine in serum based on the inner filter effect of enzyme-catalyzed oxidation products to silicon nanoparticles. Anal Bioanal Chem 413, 1405–1415 (2021). https://doi.org/10.1007/s00216-020-03104-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-020-03104-7