Abstract
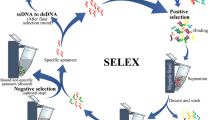
This study reports a fluorescence microscope-imaging assay for determining the binding characteristics of single-stranded DNA aptamers selected against the antibacterial agent, triclosan. The imaging assay utilises fluorescently labelled aptamers and target-immobilised matrices. Upon binding of triclosan-specific aptamers to triclosan-conjugated matrices, the binding complex was visualised and the image was captured with the aid of a fluorescence microscope. Subsequently, the fluorescent intensities of aptamer-bound matrices were analysed using dedicated image-processing software and correlated to known concentrations of selected input aptamers. Thus, by plotting fluorescence intensities against different aptamer concentrations, binding isotherms were generated to determine aptamer Kd values. The imaging assay was applied to characterise the binding affinities and specificities of ten triclosan-specific aptamers H1–H10. One of the candidate aptamers, H6, showed a Kd value of 378 nM, which was comparable with previously published Kd values for aptamer—generated against triclosan analogous. In addition, the utility of the imaging assay for aptamer characterisation was compared with a commonly used affinity column-binding assay. It was concluded that the imaging assay was superior to alternative assays in terms of accuracy, simplicity, and reproducibility.






Similar content being viewed by others
References
Dann AB, Hontela A. Triclosan: environmental exposure, toxicity and mechanisms of action. J Appl Toxicol. 2011;31:285–311.
Zhao C, Xie H, Xu J, Xu X, Zhang J, Hu Z, et al. Bacterial community variation and microbial mechanism of triclosan (TCS) removal by constructed wetlands with different types of plants. Sci Total Environ. 2015;505:633–9.
Wang H, Zhang J, Gao F, Yang Y, Duan H, Wu Y, et al. Simultaneous analysis of synthetic musks and triclosan in human breast milk by gas chromatography tandem mass spectrometry. J Chromatogr B. 2011;879:1861–9.
Azzouz A, Rascón AJ, Ballesteros E. Simultaneous determination of parabens, alkylphenols, phenylphenols, bisphenol A and triclosan in human urine, blood and breast milk by continuous solid-phase extraction and gas chromatography–mass spectrometry. J Pharm Biomed Anal. 2016;119:16–26.
Asimakopoulos AG, Wang L, Thomaidis NS, Kannan K. A multi-class bioanalytical methodology for the determination of bisphenol A diglycidyl ethers, P-hydroxybenzoic acid esters, benzophenone-type ultraviolet filters, triclosan, and triclocarban in human urine by liquid chromatography–tandem mass spectrometry. J Chromatogr A. 2014;1324:141–8.
Henry ND, Fair PA. Comparison of in vitro cytotoxicity, estrogenicity and anti-estrogenicity of triclosan, perfluorooctane sulfonate and perfluorooctanoic acid. J Appl Toxicol. 2013;33:265–72.
Kumar V, Chakraborty A, Kural MR, Roy P. Alteration of testicular steroidogenesis and histopathology of reproductive system in male rats treated with triclosan. Reprod Toxicol. 2019;27:177–85.
Yueh MF, Taniguchi K, Chen S, Evans RM, Hammock BD, Karin M, et al. The commonly used antimicrobial additive triclosan is a liver tumor promoter. Proc Natl Acad Sci. 2014;111:17200–5.
Li S. Selection and characterisation of single-stranded DNA aptamers for triclosan. A PhD thesis submitted to the library of Victoria University of Wellington, New Zealand. 2016;URI:http://hdl.handle.net/10063/5017.
Ahn KC, Ranganathan A, Bever CS, Hwang SH, Holland EB, Morisseau K, et al. Detection of the antimicrobial triclosan in environmental samples by immunoassay. Environ Sci Technol. 2016;50:3754–61.
Cha M, Lee HJ, Kim JS, Kim E. Environmental assessment of estrogenic pollutants in Nam River of Korea using indirect competitive Elisa and E-screen assay. Toxicol Environ Heal Sci. 2013;4:262–8.
Ellington AD, Szostak JW. In vitro selection of RNA molecules that bind specific ligands. Nature. 1990;346:818–22.
Tuerk C, Gold L. Systematic evolution of ligands by exponential enrichment - RNA ligands to bacteriophage-T4 DNA-polymerase. Science. 1990;249:505–10.
Alsager OA, Kumar S, Zhu B, Travas-Sejdic J, McNatty KP, Hodgkiss JM. Ultrasensitive colorimetric detection of 17β-estradiol: the effect of shortening DNA aptamer sequences. Anal Chem. 2015;87:4201–9.
Cruz-Aguado JA, Penner G. Fluorescence polarization based displacement assay for the determination of small molecules with aptamers. Anal Chem. 2008;80:8853–5.
Mehta J, Rouah-Martin E, Van Dorst B, Maes B, Herrebout W, Scippo ML, et al. Selection and characterization of PCB-binding DNA aptamers. Anal Chem. 2012;84:1669–76.
Ma H, Liu J, Ali MM, Mahmood MA, Labanieh L, Lu M, et al. Nucleic acid aptamers in cancer research, diagnosis and therapy. Chem Soc Rev. 2015;44:1240–56.
Sundberg L, Porath J. Preparation of adsorbents for biospecific affinity chromatography. 1. Attachment of group-containing ligands to insoluble polymers by means of bifunctional oxiranes. J Chromatogr A. 1974;90:87–98.
Porath I, Fornstedt N. Group fractionation of plasma proteins on dipolar ion exchangers. J Chromatogr A. 1970;51:479–89.
Zuker M. Mfold web server for nucleic acid folding and hybridization prediction. Nucleic Acids Res. 2003;31:3406–15.
Mann D, Reinemann C, Stoltenburg R, Strehlitz B. In vitro selection of DNA aptamers binding ethanolamine. Biochem Biophys Res Commun. 2005;338:1928–34.
Mehta J, Van Dorst B, Rouah-Martin E, Herrebout W, Scippo ML, Blust R, et al. In vitro selection and characterization of DNA aptamers recognizing chloramphenicol. J Biotechnol. 2011;155:361–9.
Cheng R, Liu S, Shi H, Zhao G. A highly sensitive and selective aptamer-based colorimetric sensor for the rapid detection of PCB 77. J Hazard Mater. 2018;341:373–80.
Harada K, Frankel AD. Identification of two novel arginine binding DNAs. EMBO J. 1995;14:5798–811.
Paramasivan S, Rujan L, Bolton PH. Circular dichroism of quadruplex DNAs: applications to structure, cation effects and ligand binding. Methods. 2007;43:324–31.
Chang YM, Chen CK, Hou MH. Conformational changes in DNA upon ligand binding monitored by circular dichroism. Int J Mol Sci. 2012;13:3394–413.
Xu S, Yuan H, Chen S, Xu A, Wang J, Wu L. Selection of DNA aptamers against polychlorinated biphenyls as potential biorecognition elements for environmental analysis. Anal Biochem. 2012;423:195–201.
Acknowledgments
The authors would like to thank Dr. Alan Clark for guidance in the preparation of affinity matrices, Dr. Junsheng Lin for scientific discussions and aid in designing the processes of image capturing and analysing, and Dr. Guozheng Liu for proofreading the manuscript.
Funding
This work was financially supported by the Ministry of Science and Innovation of New Zealand (contract number C08X0708).
Author information
Authors and Affiliations
Contributions
All researchers who contributed to this study have consented to its submission.
Corresponding author
Ethics declarations
No experimental human or animals involved in this study.
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
The research results herein have not been submitted or published in any other journals.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 960 kb)
Rights and permissions
About this article
Cite this article
Li, S., Clarkson, M. & McNatty, K. Selection and characterisation of triclosan-specific aptamers using a fluorescence microscope-imaging assay. Anal Bioanal Chem 412, 7285–7294 (2020). https://doi.org/10.1007/s00216-020-02863-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-020-02863-7