Abstract
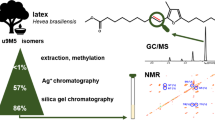
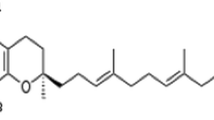
Countercurrent chromatography (CCC) was used for the enrichment of α-tocodienol (α-T2), a rare vitamin E–related minor compound previously tentatively detected in palm oil. Hitherto, only one isomer has been mentioned to occur at traces in palm oil. However, CCC fractionation followed by GC/MS measurements of all fractions resulted in the detection of two α-T2 isomers in five different palm oil vitamin E dietary supplement capsules. Five repetitive CCC separations of ~ 1 g sample and additional purification steps by column chromatography provided ~ 2 mg of two equally abundant α-T2 isomers with a purity of ~ 85%. The positions of the double bonds in the alkyl side chain could be assigned by means of two characteristic chemical shifts in the 1H NMR spectrum. Accordingly, the structures of the α-T2 isomers were 2,5,7,8-tetramethyl-2-(4,8,12-trimethyltridec-3,11-dienyl)chroman-6-ol (double bonds in 3′,11′-position) and 2,5,7,8-tetramethyl-2-(4,8,12-trimethyltridec-7,11-dienyl)chroman-6-ol (double bonds in 7′,11′-position). Natural occurrence of both isomers was proven by GC/MS screening of crude palm oil after saponification and CCC separation. Moreover, GC/MS analysis allowed the tentative assignment of γ-tocomonoenol (γ-T1) and β-tocomonoenol (β-T1) as trace compounds in palm oil.







Similar content being viewed by others
References
Evans HM, Bishop KS. On the existence of a hitherto unrecognized dietary factor essential for reproduction. Science. 1922;56:650–1.
Sure B. Dietary requirements for reproduction II. The existence of a specific vitamin for reproduction. J Biol Chem. 1924;58:693–709.
Brigelius-Flohe R, Traber MG. Vitamin E: function and metabolism. FASEB J. 2016;10:1145–55.
Prasad K. Tocotrienols and cardiovascular health. Curr Pharm Des. 2011;17:2147–54.
Saremi A, Arora R. Vitamin E and cardiovascular disease. Am J Ther. 2010;17:56–65.
Rizvi S, Raza ST, Ahmed F, Ahmad A, Abbas S, Mahdi F. The role of vitamin E in human health and some diseases. Sultan Qaboos Univ Med J. 2014;14:157–65.
Birringer M, Siems K, Maxones A, Frank J, Lorkowski S. Natural 6-hydroxy-chromanols and -chromenols: structural diversity, biosynthetic pathways and health implications. RSC Adv. 2018;8:4803–41.
Kamal-Eldin A, Appelqvist L-Å. The chemistry and antioxidant properties of tocopherols and tocotrienols. Lipids. 1996;31:671–701.
Evans HM, Emerson OH, Emerson GA. The isolation from wheat germ oil of an alcohol, α-tocopherol, having the properties of vitamin E. J Biol Chem. 1936;113:319–32.
Fernholz E. On the constitution of α-tocopherol. J Am Chem Soc. 1938;603:700–5.
Sen CK, Khanna S, Roy S. Tocotrienols: vitamin E beyond tocopherols. Life Sci. 2006;78:2088–98.
IUPAC-IUB Joint Commission on Biochemical Nomenclature. Nomenclature of tocopherols and related compounds. Pure Appl Chem. 1982;54:1507–10.
Matsumoto A, Takahashi S, Nakano K, Kijima S. Identification of new vitamin E in plant oil. J Oleo Sci. 1995;44:593–7.
Ng MH, Choo YM, Ma AN, Chuah CH, Hashim MA. Separation of vitamin E (tocopherol, tocotrienol, and tocomonoenol) in palm oil. Lipids. 2004;39:1031–5.
Yamamoto Y, Maita N, Fujisawa A, Takashima J, Ishii Y, Dunlap WCA. New vitamin E (α-tocomonoenol) from eggs of the Pacific salmon Oncorhynchus keta. J Nat Prod. 1999;62:1685–7.
Butinar B, Bučar-Miklavčič M, Mariani C, Raspor P. New vitamin E isomers (gamma-tocomonoenol and alpha-tocomonoenol) in seeds, roasted seeds and roasted seed oil from the Slovenian pumpkin variety ‘Slovenska golica’. Food Chem. 2011;128:505–12.
Kruk J, Pisarski A, Szymańska R. Novel vitamin E forms in leaves of Kalanchoe daigremontiana and Phaseolus coccineus. J Plant Physiol. 2011;168:2021–7.
Fiorentino A, Mastellone C, D’Abrosca B, Pacifico S, Scognamiglio M, Cefarelli G, et al. δ-Tocomonoenol. A new vitamin E from kiwi (Actinidia chinensis) fruits. Food Chem. 2009;115:187–92.
Gee PT, Liew CY, Thong MC, Gay MCL. Vitamin E analysis by ultra-performance convergence chromatography and structural elucidation of novel a-tocodienol by high-resolution mass spectrometry. Food Chem. 2016;196:367–73.
Ito Y. Golden rules and pitfalls in selecting optimum conditions for high-speed counter-current chromatography. J Chromatogr A. 2005;1065:145–68.
Pauli GF, Pro SM, Friesen JB. Countercurrent separation of natural products. J Nat Prod. 2008;71:1489–508.
Marston A, Hostettmann K. Counter-current chromatography as a preparative tool—applications and perspectives. J Chromatogr A. 1994;658:315–41.
Sutherland IA, Brown L, Forbes S, Games G, Hawes D, Hostettmann K, et al. Countercurrent chromatography (CCC) and its versatile application as an industrial purification & production process. J Liq Chromatogr Relat Technol. 1998;21:279–98.
Zhao C-X, He C-H. Sample capacity in preparative high-speed counter-current chromatography. J Chromatogr A. 2007;1146:186–92.
Ito Y, Bowman RL. Countercurrent chromatography: liquid-liquid partition chromatography without solid support. Science. 1970;167:281–3.
Ito Y. Origin and evolution of the coil planet centrifuge: a personal reflection of my 40 years of CCC research and development. Sep Purif Rev. 2005;34:131–54.
Müller M, Hammann S, Vetter W. Countercurrent chromatographic isolation and purification of 11′-α-tocomonoenol from the vitamin E extract of palm oil. Food Chem. 2018;256:327–32.
Vetter W, Hammann S, Müller M, Englert M, Huang Y. The use of countercurrent chromatography in the separation of nonpolar lipid compounds. J Chromatogr A. 2017;1501:51–60.
Schröder M, Vetter W. Detection of 430 fatty acid methyl esters from a transesterified butter sample. J Am Oil Chem Soc. 2013;90:771–90.
Hammann S, Kröpfl A, Vetter W. More than 170 polyunsaturated tocopherol-related compounds in a vitamin E capsule: countercurrent chromatographic enrichment, gas chromatography/mass spectrometry analysis and preliminary identification of the potential artefacts. J Chromatogr A. 2016;1476:77–87.
Hammann S, Englert M, Müller M, Vetter W. Accelerated separation of GC-amenable lipid classes in plant oils by countercurrent chromatography in the co-current mode. Anal Bioanal Chem. 2015;407:9019–28.
Englert M, Brown L, Vetter W. Heart-cut two-dimensional countercurrent chromatography with a single instrument. Anal Chem. 2015;87:10172–7.
Hammann S, Wendlinger C, Vetter W. Analysis of intact cholesteryl esters of furan fatty acids in cod liver. Lipids. 2015;50:611–20.
Hammann S, Vetter W. Method development for the determination of free and esterified sterols in button mushrooms (Agaricus bisporus). J Agric Food Chem. 2016;64:3437–44.
Englert M, Hammann S, Vetter W. Isolation of β-carotene, α-carotene and lutein from carrots by countercurrent chromatography with the solvent system modifier benzotrifluoride. J Chromatogr A. 2015;1388:119–25.
Vetter W, Müller M, Sommer K, Schröder M, Hammann S. Development of equivalent chain length (ECL) rules for lipid compounds. J Chromatogr A. 2019;1599:187–95.
Hammann S, Korf A, Bull ID, Hayen H, Cramp LJE. Lipid profiling and analytical discrimination of seven cereals using high temperature gas chromatography coupled to high resolution quadrupole time-of-flight mass spectrometry. Food Chem. 2019;282:27–35.
Ohnmacht S, West R, Simionescu R, Atkinson J. Assignment of the 1H and 13C NMR of tocotrienols. Magn Reson Chem. 2008;46:287–94.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 624 kb)
Rights and permissions
About this article
Cite this article
Müller, M., Kröpfl, A. & Vetter, W. Identification of two α-tocodienol isomers in palm oil after countercurrent chromatographic enrichment. Anal Bioanal Chem 412, 795–802 (2020). https://doi.org/10.1007/s00216-019-02303-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-019-02303-1