Abstract
Phenolic resin based activated carbon fibers (ACFs) were applied for the first time as a reversed-dispersive solid-phase extraction (r-DSPE) sorbent. A modified quick, easy, cheap, effective, rugged, and safe (QuEChERS) method was applied to determine 26 pesticides (organophosphates, organochlorines, synthetic pyrethroids, and herbicides) in different complex matrices, including cauliflower, cucumber, banana, apple, wheat, and black gram. Different physicochemical characterization techniques were used to investigate the engineering and structural properties of the r-DSPE sorbent. All the chromatographic analyses were performed with a gas chromatograph equipped with an electron capture detector. The recoveries of all 26 pesticides were acceptable (70–120%), with relative standard deviations of less than 15%. The limit of detection and the limit of quantification were 1.13–5.48 ng/g and 3.42–16.60 ng/g, respectively. In the original QuEChERS method, primary secondary amine is extensively used as the r-DSPE sorbent in the cleanup process, but it is eightfold more expensive than the ACFs used in this study. Therefore, the modified QuEChERS method using ACFs during the cleanup process is more efficient, cheaper, and more robust to determine pesticides from different types of matrices, including vegetables, grains, and fruits, and ACFs could be used as a cost-effective alternative to primary secondary amine.

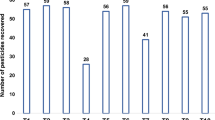
Sample clean-up using PSA and ACF as r-DSPE sorbent in QuEChERS method







Similar content being viewed by others
References
Pareja L, Cesio V, Heinzen H, Fernández-Alba AR. Evaluation of various QuEChERS based methods for the analysis of herbicides and other commonly used pesticides in polished rice by LC–MS/MS. Talanta. 2011;83(5):1613–22. https://doi.org/10.1016/j.talanta.2010.11.052.
Han Y, Song L, Zou N, Chen R, Qin Y, Pan C. Multi-residue determination of 171 pesticides in cowpea using modified QuEChERS method with multi-walled carbon nanotubes as reversed-dispersive solid-phase extraction materials. J Chromatogr B. 2016;1031:99–108. https://doi.org/10.1016/j.jchromb.2016.07.043.
García-Reyes JF, Gilbert-López B, Molina-Díaz A, Fernández-Alba AR. Determination of pesticide residues in fruit-based soft drinks. Anal Chem. 2008;80(23):8966–74. https://doi.org/10.1021/ac8012708.
Andrascikova M, Hrouzkova S. A comparative study of three modifications of the QuEChERS method for determination of endocrine disrupting pesticide residues in lemon matrices by fast GC-MS. Anal Methods. 2013;5(6):1374–84. https://doi.org/10.1039/C3AY26434C.
Zhao P, Wang L, Zhou L, Zhang F, Kang S, Pan C. Multi-walled carbon nanotubes as alternative reversed-dispersive solid phase extraction materials in pesticide multi-residue analysis with QuEChERS method. J Chromatogr A. 2012;1225:17–25. https://doi.org/10.1016/j.chroma.2011.12.070.
Whiteaker JR, Prather KA. Detection of pesticide residues on individual particles. Anal Chem. 2003;75(1):49–56. https://doi.org/10.1021/ac025771v.
Srivastava A, Rai S, Kumar Sonker A, Karsauliya K, Pandey CP, Singh SP. Simultaneous determination of multiclass pesticide residues in human plasma using a mini QuEChERS method. Anal Bioanal Chem. 2017;409(15):3757–65. https://doi.org/10.1007/s00216-017-0317-7.
Yang T, Zhang Z, Zhao B, Hou R, Kinchla A, Clark JM, et al. Real-time and in situ monitoring of pesticide penetration in edible leaves by surface-enhanced Raman scattering mapping. Anal Chem. 2016;88(10):5243–50. https://doi.org/10.1021/acs.analchem.6b00320.
Al-Degs YS, Al-Ghouti MA, El-Sheikh AH. Simultaneous determination of pesticides at trace levels in water using multiwalled carbon nanotubes as solid-phase extractant and multivariate calibration. J Hazard Mater. 2009;169(1–3):128–35. https://doi.org/10.1016/j.jhazmat.2009.03.065.
Zhou Q, Xiao J, Wang W. Using multi-walled carbon nanotubes as solid phase extraction adsorbents to determine dichlorodiphenyltrichloroethane and its metabolites at trace level in water samples by high performance liquid chromatography with UV detection. J Chromatogr A. 2006;1125(2):152–8. https://doi.org/10.1016/j.chroma.2006.05.047.
Song X-Y, Shi Y-P, Chen J. Carbon nanotubes-reinforced hollow fibre solid-phase microextraction coupled with high performance liquid chromatography for the determination of carbamate pesticides in apples. Food Chem. 2013;139(1–4):246–52. https://doi.org/10.1016/j.foodchem.2013.01.112.
Hou X, Lei S, Qiu S, Guo L, Yi S, Liu W. A multi-residue method for the determination of pesticides in tea using multi-walled carbon nanotubes as a dispersive solid phase extraction absorbent. Food Chem. 2014;153:121–9. https://doi.org/10.1016/j.foodchem.2013.12.031.
Rodrigues FM, Mesquita PRR, de Oliveira LS, de Oliveira FS, Menezes Filho A, de P. Pereira PA, et al. Development of a headspace solid-phase microextraction/gas chromatography–mass spectrometry method for determination of organophosphorus pesticide residues in cow milk. Microchem J. 2011;98(1):56–61. https://doi.org/10.1016/j.microc.2010.11.002.
Rai S, Singh AK, Srivastava A, Yadav S, Siddiqui MH, Mudiam MKR. Comparative evaluation of QuEChERS method coupled to DLLME extraction for the analysis of multiresidue pesticides in vegetables and fruits by gas chromatography-mass spectrometry. Food Anal Methods. 2016;9(9):2656–69. https://doi.org/10.1007/s12161-016-0445-2.
Zhao P, Wang L, Luo J, Li J, Pan C. Determination of pesticide residues in complex matrices using multi-walled carbon nanotubes as reversed-dispersive solid-phase extraction sorbent. J Sep Sci. 2012;35(1):153–8. https://doi.org/10.1002/jssc.201100566.
Anastassiades M, Maštovská K, Lehotay SJ. Evaluation of analyte protectants to improve gas chromatographic analysis of pesticides. J Chromatogr A. 2003;1015(1–2):163–84. https://doi.org/10.1016/S0021-9673(03)01208-1.
Anastassiades M, Lehotay SJ, Štajnbaher D, Schenck FJ. Fast and easy multiresidue method employing acetonitrile extraction/partitioning and “dispersive solid-phase extraction” for the determination of pesticide residues in produce. J AOAC Int. 2003;86(2):412–31.
Rajski Ł, Lozano A, Uclés A, Ferrer C, Fernández-Alba AR. Determination of pesticide residues in high oil vegetal commodities by using various multi-residue methods and clean-ups followed by liquid chromatography tandem mass spectrometry. J Chromatogr A. 2013;1304:109–20. https://doi.org/10.1016/j.chroma.2013.06.070.
Han Y, Zou N, Song L, Li Y, Qin Y, Liu S, et al. Simultaneous determination of 70 pesticide residues in leek, leaf lettuce and garland chrysanthemum using modified QuEChERS method with multi-walled carbon nanotubes as reversed-dispersive solid-phase extraction materials. J Chromatogr B. 2015;1005:56–64. https://doi.org/10.1016/j.jchromb.2015.10.002.
Wu Y-L, Chen R-X, Zhu Y, Zhao J, Yang T. Simultaneous determination of sixteen amide fungicides in vegetables and fruits by dispersive solid phase extraction and liquid chromatography–tandem mass spectrometry. J Chromatogr B. 2015;989:11–20. https://doi.org/10.1016/j.jchromb.2015.02.038.
Singh S, Singh A, Bais VSS, Prakash B, Verma N. Multi-scale carbon micro/nanofibers-based adsorbents for protein immobilization. Mater Sci Eng C. 2014;38:46–54. https://doi.org/10.1016/j.msec.2014.01.042.
Bikshapathi M, Mandal S, Mathur GN, Sharma A, Verma N. Modification of activated carbon fiber by metal dispersion and surface functionalization for the removal of 2-chloroethanol. Ind Eng Chem Res. 2011;50(23):13092–104.
Bikshapathi M, Singh S, Bhaduri B, Mathur GN, Sharma A, Verma N. Fe-nanoparticles dispersed carbon micro and nanofibers: surfactant-mediated preparation and application to the removal of gaseous VOCs. Colloids Surf A. 2012;399:46–55. https://doi.org/10.1016/j.colsurfa.2012.02.023.
Singh S, Verma N. Graphitic carbon micronanofibers asymmetrically dispersed with alumina-nickel nanoparticles: a novel electrode for mediatorless microbial fuel cells. Int J Hydrogen Energy. 2015;40(17):5928–38. https://doi.org/10.1016/j.ijhydene.2015.03.010.
Prajapati YN, Bhaduri B, Joshi HC, Srivastava A, Verma N. Aqueous phase adsorption of different sized molecules on activated carbon fibers: effect of textural properties. Chemosphere. 2016;155:62–9. https://doi.org/10.1016/j.chemosphere.2016.04.040.
ALOthman ZA. A review: fundamental aspects of silicate mesoporous materials. Materials. 2012;5(12):2874–902. https://doi.org/10.3390/ma5122874.
Sing KSW, Williams RT. Physisorption hysteresis loops and the characterization of nanoporous materials. Adsorpt Sci Technol. 2004;22(10):773–82.
Haress NG, Govindarajan M, Al-Wabli RI, Almutairi MS, Al-Alshaikh MA, Al-Saadi AA, et al. Spectroscopic (FT-IR, FT-Raman, UV, 1H and 13C NMR) profiling and theoretical calculations of (2E)-2-[3-(1H-imidazol-1-yl)-1-phenylpropylidene]hydrazinecarboxamide: an anticonvulsant agent. J Mol Struct. 2016;1118:219–32. https://doi.org/10.1016/j.molstruc.2016.04.026.
Singh S, Ashfaq M, Singh RK, Joshi HC, Srivastava A, Sharma A, et al. Preparation of surfactant-mediated silver and copper nanoparticles dispersed in hierarchical carbon micro-nanofibers for antibacterial applications. New Biotechnol. 2013;30(6):656–65. https://doi.org/10.1016/j.nbt.2013.05.002.
Lehotay SJ, Son KA, Kwon H, Koesukwiwat U, Fu W, Mastovska K, et al. Comparison of QuEChERS sample preparation methods for the analysis of pesticide residues in fruits and vegetables. J Chromatogr A. 2010;1217(16):2548–60. https://doi.org/10.1016/j.chroma.2010.01.044.
Koesukwiwat U, Lehotay SJ, Mastovska K, Dorweiler KJ, Leepipatpiboon N. Extension of the QuEChERS method for pesticide residues in cereals to flaxseeds, peanuts, and doughs. J Agric Food Chem. 2010;58(10):5950–8. https://doi.org/10.1021/jf902988b.
Acknowledgements
The authors are grateful for the support of the Department of Science and Technology (New Delhi, India) in the form of research grants (DST/INSPIRE/04/2015/001869 and GAP-306) for this research work. The authors are also obliged to CSIR-Indian Institute of Toxicology Research (CSIR-IITR) for providing necessary infrastructural facilities. The authors appreciatively acknowledge Gun Ei Chemical Industry Co. Ltd (Japan) for supplying the activated carbon fibers. The CSIR-IITR communication number for this article is 3459.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Electronic supplementary material
ESM 1
(PDF 473 kb)
Rights and permissions
About this article
Cite this article
Singh, S., Srivastava, A. & Singh, S.P. Inexpensive, effective novel activated carbon fibers for sample cleanup: application to multipesticide residue analysis in food commodities using a QuEChERS method. Anal Bioanal Chem 410, 2241–2251 (2018). https://doi.org/10.1007/s00216-018-0894-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-018-0894-0