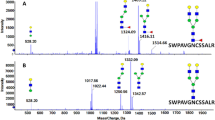
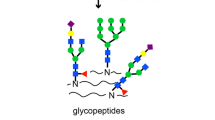
Abstract
Fucosylation of N-glycoproteins has been implicated in various diseases, such as hepatocellular carcinoma (HCC). However, few studies have performed site-specific analysis of fucosylation in liver-secreted proteins. In this study, we characterized the fucosylation patterns of liver-secreted proteins in HCC plasma using a workflow to identify site-specific N-glycoproteins, where characteristic B- and/or Y-ion series with and without fucose in collision-induced dissociation were used in tandem mass spectrometry. In total, 71 fucosylated N-glycopeptides from 13 major liver-secreted proteins in human plasma were globally identified by LC-MS/MS. Additionally, 37 fucosylated N-glycopeptides were newly identified from nine liver-secreted proteins, including alpha-1-antichymotrypsin, alpha-1-antitrypsin, alpha-2-HS-glycoprotein, ceruloplasmin, alpha-1-acid glycoprotein 1/2, alpha-2-macroglobulin, serotransferrin, and beta-2-glycoprotein 1. Of the fucosylated N-glycopeptides, bi- and tri-antennary glycoforms were the most common ones identified in liver-secreted proteins from HCC plasma. Therefore, we suggest that this analytical method is effective for characterizing fucosylation in liver-secreted proteins.

A global map of fucosylated and non-fucosylated glycopeptides from 13 liver-secreted glycoproteins in hepatocellular carcinoma plasma





Similar content being viewed by others
Abbreviations
- A2M:
-
Alpha-2-macroglobulin
- AACT:
-
Alpha-1-antichymotrypsin
- AAT:
-
Alpha-1-antitrypsin
- AGP1:
-
Alpha-1-acid glycoprotein 1
- AGP2:
-
Alpha-1-acid glycoprotein 2
- APOH:
-
Beta-2-glycoprotein 1
- CID:
-
Collision-induced dissociation
- CP:
-
Ceruloplasmin
- FETUA:
-
Fetuin A
- Fuc:
-
Fucose
- HCC:
-
Hepatocellular carcinoma
- HCD:
-
Higher-energy collision dissociation
- Hex:
-
Hexose
- HexNAc:
-
N-Acetyl hexosamine
- HILIC:
-
Hydrophilic interaction liquid chromatography
- HP:
-
Haptoglobin
- HPX:
-
Hemopexin
- KNG1:
-
Kininogen-1
- MS:
-
Mass spectrometry
- N:
-
Asparagine
- Sia:
-
Sialic acid
- TF:
-
Serotransferrin
- VTN:
-
Vitronectin
References
Walsh G, Jefferis R. Post-translational modifications in the context of therapeutic proteins. Nat Biotechnol. 2006;24(10):1241–52.
Kornfeld R, Kornfeld S. Assembly of asparagine-linked oligosaccharides. Annu Rev Biochem. 1985;54(1):631–64.
An HJ, Peavy TR, Hedrick JL, Lebrilla CB. Determination of N-glycosylation sites and site heterogeneity in glycoproteins. Anal Chem. 2003;75(20):5628–37.
Fuster MM, Esko JD. The sweet and sour of cancer: glycans as novel therapeutic targets. Nat Rev Cancer. 2005;5(7):526–42.
Cheng L, Luo S, Jin C, Ma H, Zhou H, Jia L. FUT family mediates the multidrug resistance of human hepatocellular carcinoma via the PI3K/Akt signaling pathway. Cell Death Dis. 2013;4(11):e923. doi:10.1038/cddis.2013.450.
Kang X, Wang N, Pei C, Sun LU, Sun R, Chen JIE, et al. Glycan-related gene expression signatures in human metastatic hepatocellular carcinoma cells. Exp Ther Med. 2012;3(3):415–22.
Nakagawa T, Uozumi N, Nakano M, Mizuno-Horikawa Y, Okuyama N, Taguchi T, et al. Fucosylation of N-glycans regulates the secretion of hepatic glycoproteins into bile ducts. J Biol Chem. 2006;281(40):29797–806.
Anderson NL, Anderson NG. The human plasma proteome: history, character, and diagnostic prospects. Mol Cell Proteomics. 2002;2(1):847–67.
Farrah T, Deutsch EW, Omenn GS, Campbell DS, Sun Z, Bletz JA, et al. High-confidence human plasma proteome reference set with estimated concentrations in PeptideAtlas. Mol Cell Proteomics. 2011;10(9), M110.006353.
Boron WF, Boulpaep EL. Medical physiology: a cellular and molecular approach. Philadelphia: Elsevier/Saunders; 2003. p. 1300.
Uhlén M, Fagerberg L, Hallström BM, Lindskog C, Oksvold P, Mardinoglu A, et al. Proteomics. Tissue-based map of the human proteome. Science. 2015;6220(6220):1260419.
Ludwig JA, Weinstein JN. Biomarkers in cancer staging, prognosis and treatment selection. Nat Rev Cancer. 2005;5(11):845–56.
Miyaaki H, Nakashima O, Kurogi M, Eguchi K, Kojiro M. Lens culinaris agglutinin-reactive alpha-fetoprotein and protein induced by vitamin K absence II are potential indicators of a poor prognosis: a histopathological study of surgically resected hepatocellular carcinoma. J Gastroenterol. 2007;42(12):962–8.
Khien VV, Mao HV, Chinh TT, Ha PT, Bang MH, Lac BV, et al. Clinical evaluation of lentil lectin-reactive alpha-fetoprotein-L3 in histology-proven hepatocellular carcinoma. Int J Biol Markers. 2001;16(2):105–11.
Breborowicz J, Mackiewicz A, Breborowicz D. Microheterogeneity of alpha-fetoprotein in patient serum as demonstrated by lectin affino-electrophoresis. Scand J Immunol. 1981;14(1):15–20.
Okuda H, Saito A, Shiratori K, Yamamoto M, Takasaki K, Nakano M. Clinicopathologic features of patients with primary malignant hepatic tumors seropositive for alpha-fetoprotein-L3 alone in comparison with other patients seropositive for alpha-fetoprotein-L3. J Gastroenterol Hepatol. 2005;20(5):759–64.
Naitoh A, Aoyagi Y, Asakura H. Highly enhanced fucosylation of serum glycoproteins in patients with hepatocellular carcinoma. J Gastroenterol Hepatol. 1999;14(5):436–45.
Li D, Mallory T, Satomura S. AFP-L3: a new generation of tumor marker for hepatocellular carcinoma. Clin Chim Acta. 2001;313(1-2):15–9.
Taylor AD, Hancock WS, Hincapie M, Taniguchi N, Hanash SM. Towards an integrated proteomic and glycomic approach to finding cancer biomarkers. Genome Med. 2009;1(6):57.
Zhao J, Qiu W, Simeone DM, Lubman DM. N-linked glycosylation profiling of pancreatic cancer serum using capillary liquid phase separation coupled with mass spectrometric analysis. J Proteome Res. 2007;6(3):1126–38.
Pedersen JW, Blixt O, Bennett EP, Tarp MA, Dar I, Mandel U, et al. Seromic profiling of colorectal cancer patients with novel glycopeptide microarray. Int J Cancer. 2011;128(8):1860–71.
Barrabés S, Pagès-Pons L, Radcliffe CM, Tabarés G, Fort E, Royle L, et al. Glycosylation of serum ribonuclease 1 indicates a major endothelial origin and reveals an increase in core fucosylation in pancreatic cancer. Glycobiology. 2007;17(4):388–400.
Comunale MA, Lowman M, Long RE, Krakover J, Philip R, Seeholzer S, et al. Proteomic analysis of serum associated fucosylated glycoproteins in the development of primary hepatocellular carcinoma. J Proteome Res. 2006;5(2):308–15.
Schiel JE, Au J, He H-J, Phinney KW. LC-MS/MS biopharmaceutical glycoanalysis: identification of desirable reference material characteristics. Anal Bioanal Chem. 2012;403(8):2279–89.
Pompach P, Brnakova Z, Sanda M, Wu J, Edwards N, Goldman R. Site-specific glycoforms of haptoglobin in liver cirrhosis and hepatocellular carcinoma. Mol Cell Proteomics. 2013;12(5):1281–93.
Pompach P, Ashline DJ, Brnakova Z, Benicky J, Sanda M, Goldman R. Protein and site specificity of fucosylation in liver-secreted glycoproteins. J Proteome Res. 2014;13(12):5561–9.
Lee H-J, Cha H-J, Lim J-S, Lee SH, Song SY, Kim H, et al. Abundance-ratio-based semiquantitative analysis of site-specific N-linked glycopeptides present in the plasma of hepatocellular carcinoma patients. J Proteome Res. 2014;13(5):2328–38.
Hwang H, Lee JY, Lee HK, Park GW, Jeong HK, Moon MH, et al. In-depth analysis of site-specific N-glycosylation in vitronectin from human plasma by tandem mass spectrometry with immunoprecipitation. Anal Bioanal Chem. 2014;406(30):7999–8011.
Kuo C-W, Wu I-L, Hsiao H-H, Khoo K-H. Rapid glycopeptide enrichment and N-glycosylation site mapping strategies based on amine-functionalized magnetic nanoparticles. Anal Bioanal Chem. 2012;402(9):2765–76.
Zhao Y, Yu L, Guo Z, Li X, Liang X. Reversed-phase depletion coupled with hydrophilic affinity enrichment for the selective isolation of N-linked glycopeptides by using Click OEG-CD matrix. Anal Bioanal Chem. 2011;399(10):3359–65.
Zhu J, Lin Z, Wu J, Yin H, Dai J, Feng Z, et al. Analysis of serum haptoglobin fucosylation in hepatocellular carcinoma and liver cirrhosis of different etiologies. J Proteome Res. 2014;13(6):2986–97.
Asazawa H, Kamada Y, Takeda Y, Takamatsu S, Shinzaki S, Kim Y, et al. Serum fucosylated haptoglobin in chronic liver diseases as a potential biomarker of hepatocellular carcinoma development. Clin Chem Lab Med. 2015;53(1):95–102.
Mayampurath A, Song E, Mathur A, Yu C-y, Hammoud Z, Mechref Y, et al. Label-free glycopeptide quantification for biomarker discovery in human sera. J Proteome Res. 2014;13(11):4821–32.
Mayampurath A, Yu C-Y, Song E, Balan J, Mechref Y, Tang H. Computational framework for identification of intact glycopeptides in complex samples. Anal Chem. 2014;86(4):453–63.
Ahn YH, Shin PM, Oh NR, Park GW, Kim H, Yoo JS. A lectin-coupled, targeted proteomic mass spectrometry (MRM MS) platform for identification of multiple liver cancer biomarkers in human plasma. J Proteomics. 2012;75(17):5507–15.
Ahn YH, Shin PM, Kim Y-S, Oh NR, Ji ES, Kim KH, et al. Quantitative analysis of aberrant protein glycosylation in liver cancer plasma by AAL-enrichment and MRM mass spectrometry. Analyst. 2013;138(21):6454–62.
Ahn YH, Shin PM, Ji ES, Kim H, Yoo JS. A lectin-coupled, multiple reaction monitoring based quantitative analysis of human plasma glycoproteins by mass spectrometry. Anal Bioanal Chem. 2012;402(6):2101–12.
Lee JY, Kim JY, Park GW, Cheon MH, Kwon K-H, Ahn YH, et al. Targeted mass spectrometric approach for biomarker discovery and validation with nonglycosylated tryptic peptides from N-linked glycoproteins in human plasma. Mol Cell Proteomics. 2011;10(12):M111.009290.1–13.
Link AJ, Eng J, Schieltz DM, Carmack E, Mize GJ, Morris DR, et al. Direct analysis of protein complexes using mass spectrometry. Nat Biotechnol. 1999;17(7):676–82.
Kronewitter SR, An HJ, de Leoz ML, Lebrilla CB, Miyamoto S, Leiserowitz GS. The development of retorosynthetic glycan libraries to profile and classify the human serum N-linked glycome. Proteomics. 2009;9(11):2986–94.
Ozohanics O, Krenyacz J, Ludanyi K, Pollreisz F, Vekey K, Drahos L. GlycoMiner: a new software tool to elucidate glycopeptide composition. Rapid Commun Mass Spectrom. 2008;22(20):3245–54.
Mayampurath AM, Wu Y, Segu ZM, Mechref Y, Tang H. Improving confidence in detection and characterization of protein N-glycosylation sites and microheterogeneity. Rapid Commun Mass Spectrom. 2011;25(14):2007–19.
Yuan W, Sanda M, Wu J, Koomen J, Goldman R. Quantitative analysis of immunoglobulin subclasses and subclass specific glycosylation by LC-MS-MRM in liver disease. J Proteome. 2015;116:24–33.
Takakura D, Harazono A, Hashii N, Kawasaki N. Selective glycopeptide profiling by acetone enrichment and LC/MS. J Proteomics. 2014;101:17–30.
Hortin GL, Sviridov D, Anderson NL. High-abundance polypeptides of the human plasma proteome comprising the top 4 logs of polypeptide abundance. Clin Chem. 2008;54(10):1608–16.
Song E, Mayampurath A, Yu C-Y, Tang H, Mechref Y. Glycoproteomics: identifying the glycosylation of prostate specific antigen at normal and high isoelectric points by LC-MS/MS J. Proteome Res. 2014;13(12):5570–80.
Clerc F, Reiding KR, Jansen BC, Kammeijer GSM, Bondt A, Wuhrer M. Human plasma protein N-glycosylation. Glycoconj J. 2015. doi:10.1007/s10719-015-9626-2.
Reusch D, Haberger M, Falck D, Peter B, Maier B, Gassner J, et al. Comparison of methods for the analysis of therapeutic immunoglobulin G Fc-glycosylation profiles—Part 2: mass spectrometric methods. mAbs. 2015;7(4):732–42.
Gomaa AI, Khan SA, Leen ELS, Waked I, Taylor-Robinson SD. Diagnosis of hepatocellular carcinoma. World J Gastroenterol. 2009;15(11):1301–14.
Imre T, Kremmer T, Heberger K, Molnar-Szollosi E, Ludanyi K, Pocsfalvi G, et al. Mass spectrometric and linear discriminant analysis of N-glycans of human serum alpha-1-acid glycoprotein in cancer patients and healthy individuals. J Proteome. 2008;71(2):186–97.
Kondo A, Miyamoto T, Yonekawa O, Giessing AM, Osterlund EC, Jensen ON. Glycopeptide profiling of beta-2glycoprotein I by mass spectrometry reveals attenuated sialylation in patients with antiphospholipid syndrome. J Proteome. 2009;73(1):123–33.
Satomi Y, Shimonishi Y, Hase T, Takao T. Site-specific carbohydrate profiling of human transferrin by nano-flow liquid chromatography/electrospray ionization mass spectrometry. Rapid Commun Mass Spectrom. 2004;18(24):2983–8.
Arnold JN, Wallis R, Willis AC, Harvey DJ, Royle L, Dwek RA, et al. Interaction of mannan binding lectin with alpha 2 macroglobulin via exposed oligomannose glycans. J Biol Chem. 2006;281(11):6955–63.
Acknowledgments
This research was supported by the National Research Council of Science and Technology (NTM2371511, the Creative Allied Project (CAP)), the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI); it was funded by the Ministry of Health &Welfare, Republic of Korea (grant number: HI13C2098), and by the Ministry of Trade, Industry & Energy (MOTIE, 1415139249) through the Osong Academy–Industry Convergence (BAIO) fostering project.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Ethics approval and consent to participate
This study was performed in accordance with ethical standards. Plasma samples were obtained from voluntary blood donors with informed consent and approval in accordance with IRB guidelines from Yonsei University College of Medicine (Seoul, Korea).
Additional information
Eun Sun Ji and Heeyoun Hwang contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 5758 kb)
Rights and permissions
About this article
Cite this article
Ji, E.S., Hwang, H., Park, G.W. et al. Analysis of fucosylation in liver-secreted N-glycoproteins from human hepatocellular carcinoma plasma using liquid chromatography with tandem mass spectrometry. Anal Bioanal Chem 408, 7761–7774 (2016). https://doi.org/10.1007/s00216-016-9878-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-016-9878-0