Abstract
The mechanisms how Giardias attach to the intestinal epithelium remain unclear. None of the methods currently being used to measure the attachment force could provide a continuous nutrition supply and a micro-aerobic atmosphere to the Giardia. Besides, they are all labor-intensive. In the present research, a microfluidic method based on electric circuit analogy was developed. The input fluid flowed through the inlet channel with different lengths and was distributed in four assay chambers. Shear force gradients were generated in chambers, too. This allowed an easy control of fluids and the shear forces. Most importantly, the shear stress large enough to detach Giardia could be generated in laminar flow regime. Moreover, analysis could be accomplished in one single test. By applying inlet flow rates of 30, 60, and 120 μL ml–1, shear force gradients ranging from 19.47 to 60.50 Pa were generated. The adhesion forces of trophozoites were analyzed and the EC50 of the force that caused 50% trophozoites detachment was calculated as 36.60 Pa. This paper presents a novel method for measurement of Giardia adhesion force.

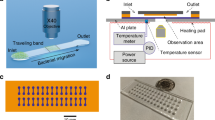
Measurement of Giardia adhesion force. Various of flow rates were applied to generate different shear forces and Giardia trophozoites remaining attached were counted (a-c). The percentages of attachment vs shear stress were plotted and the EC50 of adhesion force was calculated (d)





Similar content being viewed by others
References
Escobedo AA, Hanevik K, Almirall P, Cimerman S, Alfonso M. Management of chronic Giardia infection. Expert Rev Anti Infect Ther. 2014;12:1143–57.
Chavez B, Martinez-Palomo A. Giardia lamblia: freeze-fracture ultrastructure of the ventral disc plasma membrane. J Eukaryot Microbiol. 1995;42:136–41.
Lauwaet T, Andersen Y, Van de Ven L, Eckmann L, Gillin FD. Rapid detachment of Giardia lamblia trophozoites as a mechanism of antimicrobial action of the isoflavone formononetin. J Antimicrob Chemother. 2010;65:531–4.
Hansen WR, Tulyathan R, Dawson SC, Cande WZ, Fletcher DA. Giardia lamblia attachment force is insensitive to surface treatments. Eukaryot Cell. 2006;5:781–3.
Magne D, Favennec L, Chochillon C, Gorenflot A, Meillet D. Role of cytoskeleton and surface lectins in Giardia duodenalis attachment to Caco2 cells. J Parasitol Res. 1991;77:659–62.
Sousa MC, Gonçalves CA, Bairos VA, Poiares-Da-Silva J. Adherence of Giardia lamblia trophozoites to int-407 human intestinal cells. Clin Diagn Lab Immunol. 2001;8:258–65.
Erlandsen SL, Chase DG. Morphological alterations in the microvillous border of villous epithelial cells produced by intestinal microorganisms. Am J Clin Nutr. 1994;27:1277–86.
Hansen WR, Fletcher DA. Tonic shock induces detachment of Giardia lamblia. PLoS Negl Trop Dis. 2008;2, e169.
Elmendorf HG, Dawson SC, McCaffery JM. The cytoskeleton of Giardia lamblia. Int J Parasitol. 2003;33(1):3–28.
Mariante RM, Vancini RG, Melo AL, Benchimol M. Giardia lamblia: evaluation of the in vitro effects of nocodazole and colchicine on trophozoites. Exp Parasitol. 2005;110(1):62–72.
Holberton DV. Attachment of Giardia-a hydrodynamic model based on flagellar activity. J Exp Biol. 1974;60(1):207–21.
Lenaghan SC, Davis CA, Henson WR, Zhang Z, Zhang M. High-speed microscopic imaging of flagella motility and swimming in. Giardia Lamblia tro Phozoites PNAS. 2011;108(34):E550–8.
House SA, Richter DJ, Pham JK, Dawson SC. Giardia flagellar motility is not directly required to maintain attachment to surfaces. PLoS Pathog. 2011;7, e1002167.
Woessner DJ, Dawson SC. The Giardia median body protein is a ventral disc protein that is critical for maintaining a domed disc conformation during attachment. Eukaryot Cell. 2012;11(3):292–301.
Lu H, Koo LY, Wang WM, Lauffenburger DA, Griffith LG, Jensen KF. Microfluidic shear devices for quantitative analysis of cell adhesion. Anal Chem. 2004;76(18):5257–64.
Gutierrez E, Groisman A. Quantitative measurements of the strength of adhesion of human neutrophils to a substratum in a microfluidic device. Anal Chem. 2007;79(6):2249–58.
Culbertson CT, Mickleburgh TG, Stewart-James SA, Sellens KA, Pressnall M. Micro total analysis systems: fundamental advances and biological applications. Anal Chem. 2014;86(1):95–118.
Oh KW, Lee K, Ahn B, Furlani EP. Design of pressure-driven microfluidic networks using electric circuit analogy. Lab Chip. 2012;12(3):515–45.
Whitesides GM. The origins and the future of microfluidics. Nature. 2006;442(7101):368–73.
Salieb-Beugelaar GB, Simone G, Arora A, Philippi A, Manz A. Latest developments in microfluidic cell biology and analysis systems. Anal Chem. 2010;82(12):4848–64.
Hayt WH, Kemmerly JE (2012) Engineering Circuit Analysis, Durbin 8th ed. McGraw-Hill Book Company Inc., New York
Beebe DJ, Mensing GA, Walker GM. Physics and applications of microfluidics in biology. Annu Rev Biomed Eng. 2002;4:261–86.
Bruus H. Acoustofluidics 1: Governing equations in microfluidics. Lab Chip. 2011;11:3742–51.
Zheng GX, Zhang XM, Yang YS, Zeng SR, Wei JF, Wang YH, et al. An integrated microfluidic device for culturing and screening of Giardia lamblia. Exp Parasitol. 2014;137:1–7.
Wei CJ, Tian XF, Adam RD, Lu SQ. Giardia lamblia: intracellular localization of alpha8-giardin. Exp Parasitol. 2010;126(4):489–96.
Gut J, Ang KK, Legac J, Arkin MR, Rosenthal PJ, McKerrow JH. An image-based assay for high throughput screening of Giardia lamblia. J Microbiol Methods. 2011;84(3):398–405.
Wolfe DB, Qin D, Whitesides GM. Rapid prototyping of microstructures by soft lithography for biotechnology. Methods Mol Biol. 2010;583:81–107.
McCann JA, Peterson SD, Plesniak MW, Webster TJ, Haberstroh KM. Non-uniform flow behavior in a parallel plate flow chamber alters endothelial cell responses. Ann Biomed Eng. 2005;33(3):328–36.
Christ KV, Williamson KB, Masters KS, Turner KT. Measurement of single-cell adhesion strength using a microfluidic assay. Biomed Microdev. 2010;12(3):443–55.
Zhong W, Ma H, Wang S, Gao X, Zhang W, Qin J. An integrated microfluidic device for characterizing chondrocyte metabolism in response to distinct levels of fluid flow stimulus. Microfluid Nanofluid. 2013;15:763–73.
MacDonald MP, Spalding GC, Dholakia K. Microfluidic sorting in an optical lattice. Nature. 2003;426(6965):421–4.
Ye N, Qin J, Shi W, Liu X, Lin B. Cell-based high content screening using an integrated microfluidic device. Lab Chip. 2007;7(12):1696–704.
Acknowledgements
The authors thank Dr. Xian-Ming Liu (Dalian Institute of Chemical Physics, Chinese Academy of Sciences) for proofreading the manuscript. This work was supported by the National Natural Science Foundation of China (no. 41476085, no 81471807), Scientific Research Project of Liaoning Education Department (L2013478, L2013479), and Development Plan for Distinguished Young Scholars of Liaoning Province (LJQ2015005).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interests
The authors declare no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 777 kb)
Rights and permissions
About this article
Cite this article
Lu, L., Zheng, GX., Yang, YS. et al. Measurement of Giardia lamblia adhesion force using an integrated microfluidic assay. Anal Bioanal Chem 409, 1451–1459 (2017). https://doi.org/10.1007/s00216-016-0080-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-016-0080-1