Abstract
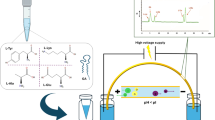
A two-dimensional achiral-chiral LC-LC method in heart-cut mode for ketoprofen and its enantiomeric fraction determination was proposed. A C8 column was used in the first dimension, and the chiral column was an α1-acid glycoprotein. The mobile phase of the chiral system was optimized by a factorial design. The effect of temperature on retention and on enantiomeric resolution was studied. Particular attention was paid to mobile phase compatibility for the two columns and to transferring time, using ketoprofen standards. The R-(−) and S-(+)-ketoprofen retention times were 9 and 11 min, respectively; the resolution was higher than 1.1 and enantiomeric fraction close to 0.5. The method was applied to capsules and gels containing ketoprofen. Factorial design was also used to establish the best conditions for gel sample preparation. Recoveries were 84 and 105 % for capsules and gels, respectively.

Two-dimensional chromatogram for KPF and its enantiomers



Similar content being viewed by others
References
Maier NM, Franco P, Lindner W. Separation of enantiomers: needs, challenges, perspectives. J Chromatogr A. 2001;906:3–33.
Lämmerhofer M. Chiral recognition by enantioselective liquid chromatography: mechanisms and modern chiral stationary phases. J Chromatogr A. 2010;1217:814–56.
Mondello L, Lewis AC, Bartle KD. Multidimensional chromatography. Chichester: John Wiley & Sons; 2002.
Meloun M, Bordovská S, Galla L. The thermodynamic dissociation constants of four non-steroidal anti-inflammatory drugs by the least-squares nonlinear regression of multiwavelength spectrophotometric pH-titration data. J Pharm Biomed Anal. 2007;45:552–64.
Sangster J. Octanol-water partition coefficients: fundamentals and physical chemistry. Chichester: John Willey & Sons; 1997.
Lagrange F, Pehourcq F, Bannwarth B, Leng JJ, Saux MC. Passage of S-(+)- and R-(−)-ketoprofen across the human isolated perfused placenta. Fundam Clin Pharmacol. 1998;12:286–91.
Rudy AC, Liu X, Brater DC, Hall SD. Stereoselective pharmacokinetics and inversion of R-KPF in healthy volunteers. J Clin Pharmacol. 1988;38:3S–10S.
British Pharmacopoeia 2013. Version 17.0. Seventh edition. Supplements 7.1 to 7.5.
Özlü C, Basan H, Satana E, Ertas N, Göger NG. Quantitative determination of ketoprofen in gels and ampules by using flow-injection UV spectrophotometry and HPLC. J Pharm Biomed Anal. 2005;39:606–11.
Mullangi R, Yao M, Srinivas NR. Resolution of enantiomers of ketoprofen by HPLC: a review. Biomed Chromatogr. 2003;17:423–34.
Menzel-Soglowek S, Geisslinger G, Brune K. Stereoselective high-performance liquid chromatographic determination of ketoprofen, ibuprofen and fenoprofen in plasma using a chiral α1-acid glycoprotein column. J Chromatogr. 1990;532:295–303.
Oda Y, Asakawa N, Abe S, Yoshida Y, Sato T. Avidin protein-conjugated column for direct injection analysis of drug enantiomers in plasma by high-performance liquid chromatography. J Chromatogr B. 1991;572:133–41.
Oda Y, Asakawa N, Yoshida Y, Sato T. On-line determination and resolution of the enantiomers of ketoprofen in plasma using coupled achiral-chiral high-performance liquid chromatography. J Pharm Biomed Anal. 1992;10:81–7.
Haginaka J, Murashima T, Fujima H, Wada H. Direct injection assay of drug enantiomers in serum on ovomucoid-bonded silica materials by liquid chromatography. J Chromatrogr. 1993;620:199–204.
Ekborg-Ott KH, Wang X, Armstrong DW. Effect of selector coverage and mobile phase composition on enantiomeric separations with ristocetin A chiral stationary phases. Microchem J. 1999;62:26–49.
Péhourcq F, Jarry C, Bannawarth B. Chiral resolution of flurbiprofen and ketoprofen enantiomers by HPLC on a glycopeptide-type column chiral stationary phase. Biomed Chromatogr. 2001;15:217–22.
Ôi N, Kitahara H, Aoki F, Kisu N. Direct separation of carboxilic acid enantiomers by high-performance liquid chromatography with amide and urea derivatives bonded to silica gel as chiral stationary phases. J Chromatogr A. 1995;689:195–201.
Yoon TH, Kim IH. Chiral separation of ketoprofen racemate by using Chirex® 3005 and Kromasil® CHI-II chiral column. Korean J Chem Eng. 2004;21:521–6.
Yagi M, Shibukawa A, Nakagawa T. Direct injection analysis of ketoprofen enantiomers in plasma using column-switching high-performance liquid chromatography system. Chem Pharm Bull. 1990;38:2513–7.
Al-Katheeri NA, Wasfi IA, Lambert M, Saeed A, Khan IA. Pharmacokinetics of ketoprofen enantiomers after intravenous administration of racemate in camels: effect on gender. J Vet Pharmacol Therap. 2000;23:137–43.
Hoke SH, Pinkston JD, Bailey RE, Tanguay SL, Eichhold TH. Comparison of packed column supercritical fluid chromatography-tandem mass spectrometry with liquid chromatography-tandem mass spectrometry for bioanalytical determination of (R)- and (S)-ketoprofen in human plasma following automated 96-well solid phase extraction. Anal Chem. 2000;72:4235–41.
Lovlin R, Vakily M, Jamali F. Rapid, sensitive and direct chiral high-performance liquid chromatographic method for ketoprofen enantiomers. J Chromatogr B. 1996;679:196–8.
Van Overbeke A, Baeyens W, Dewaele C. Comparative study on the enantiomeric separation of several non-steroidal anti-inflammatory drugs on two cellulose-based chiral stationary phases. J Liq Chromatogr. 1995;18:2427–43.
Ribeiro AE, Graça NS, Pais LS, Rodrigues AE. Preparative separation of ketoprofen enantiomers: choice of mobile phase composition and measurement of competitive adsorption isotherms. Sep Purif Technol. 2008;61:375–83.
Zhou Y, Ma C, Wang Y, Zhang QM, Zhang YY, Fu J, et al. High performance liquid chromatographic separation of thirteen drugs collected in Chinese Pharmacopoeia 2010 (Ch.P2010) on cellulose ramification chiral stationary phase. J Pharm Anal. 2012;2:48–55.
Mauleon D, Mis R, Ginesta J, Ortega E, Vilageliu J, Basi N, et al. Pharmacokinetics of ketoprofen enantiomers in monkeys following single and multiple oral administration. Chirality. 1994;6:537–42.
Carr RA, Caillé G, Ngoc AH, Foster RT. Stereospecific high-performance liquid chromatographic assay of ketoprofen in human plasma and urine. J Chromatogr B. 1995;668:175–81.
Harner T, Wiberg K, Norstrom R. Enantiomer fractions are preferred to enantiomer ratios for describing chiral signatures in environmental. Environ Sci Technol. 2000;34:218–20.
Zurita-Pérez J, Santos-Delgado MJ, Crespo-Corral E, Polo-Díez LM, Aguilar-Gallardo A. Separation of para- and meta-imazamethabenz-methyl enantiomers by direct chiral HPLC using a protein chiral selector. Chromatographia. 2012;75:847–55.
Inezédy J, Lengyel T, Ure AM. Compendium of analytical nomenclature (Orange book), chapter 9.2.4.4. 3rd ed. Oxford: IUPAC; 1997.
Evans SE, Kasprzyk-Hordern B. Aplications of chiral chromatography coupled with mass spectrometry in the analysis of chiral pharmaceuticals in the environment. Trends Environ Anal Chem (TrEAC). 2014;1:e34–51.
Gatti R, Gioia MG, Cavrini V. Analysis and stability study of retinoids in pharmaceuticals by LC with fluorescence detection. J Pharm Biomed Anal. 2000;23:147–59.
Acknowledgment
The present work received financial support from Project 152GR10581, supported by Complutense University of Madrid (Spain).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 498 kb)
Rights and permissions
About this article
Cite this article
Peñín-Ibáñez, M., Santos-Delgado, M.J. & Polo-Díez, L.M. Heart-cut achiral-chiral LC-LC method development using factorial design: application to the chiral separation of ketoprofen. Anal Bioanal Chem 409, 1135–1144 (2017). https://doi.org/10.1007/s00216-016-0039-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-016-0039-2