Abstract
DNA adductome analysis using liquid chromatography–tandem mass spectrometry is a promising tool to exhaustively search DNA modifications. Given that the molecular weight of chemical-specific adducts is determined by the total molecular weights of the active form and nucleotide bases, we developed a new method of comprehensive analysis for chemical-specific DNA adducts based on the principle of adductome analysis. The actual analytical mass range was 50 mass units up or down from the average molecular weight of the four DNA bases plus the molecular weight of the expected active form of the chemical. Using lucidin-3-O-primeveroside (LuP), lucidin-modified bases formed by its active form were exhaustively searched using this new method. Various DNA adducts, including Luc-N 2-dG and Luc-N 6-dA, were identified in the kidneys of rats given LuP. Together with measurement of 8-hydroxydeoxyguanosine (8-OHdG) levels, the combined application of this new method with a reporter gene mutation assay was performed to clarify renal carcinogenesis induced by madder color (MC) that includes LuP and alizarin (Alz) as constituent agents. A DNA adductome map derived from MC-treated rats was almost identical to that of LuP-treated rats, but not Alz-treated rats. Although 8-OHdG levels were elevated in MC- and Alz-treated rats, significant increases in gpt and Spi− mutant frequencies were observed only in MC- and LuP-treated rats. In addition, the spectrum of gpt mutants in MC-treated rats showed almost the same pattern as those in LuP-treated rats. The overall data suggest that LuP may be responsible for MC-induced carcinogenicity and that the proposed methodology is appropriate for exploring and understanding mechanisms of chemical carcinogenesis.

DNA adductome map of kidneys from F344 gpt delta rats in the control and LuP-treated groups. The peaks detected in control and LuP-treated rats are represented as black and blue spots, respectively




Similar content being viewed by others
References
Weston A, Harris CC (2000) Holland-Frei cancer medicine, 5th edn. BC Decker, Canada
Nohmi T, Suzuki T, Masumura K (2000) Recent advances in the protocols of transgenic mouse mutation assays. Mutat Res 455:191–215
Suzuki Y, Umemura T, Hibi D, Inoue T, Jin M, Ishii Y, Sakai H, Nohmi T, Yanai T, Nishikawa A, Ogawa K (2012) Possible involvement of genotoxic mechanisms in estragole-induced hepatocarcinogenesis in rats. Arch Toxicol 86:1593–1601
Hibi D, Suzuki Y, Ishii Y, Jin M, Watanabe M, Sugita-Konishi Y, Yanai T, Nohmi T, Nishikawa A, Umemura T (2011) Site-specific in vivo mutagenicity in the kidney of gpt delta rats given a carcinogenic dose of ochratoxin A. Toxicol Sci 122:406–414
Jin M, Kijima A, Hibi D, Ishii Y, Takasu S, Matsushita K, Kuroda K, Nohmi T, Nishikawa A, Umemura T (2013) In vivo genotoxicity of methyleugenol in gpt delta transgenic rats following medium-term exposure. Toxicol Sci 131:387–394
Baguley BC, Ferguson LR (1998) Mutagenic properties of topoisomerase-targeted drugs. Biochem Biophys Acta 1400:213–222
Lagerqvist A, Håkansson D, Lundin C, Prochazka G, Dreij K, Segerbäck D, Jernström B, Törnqvist M, Frank H, Seidel A, Erixon K, Jenssen D (2011) DNA repair and replication influence the number of mutations per adduct of polycyclic aromatic hydrocarbons in mammalian cells. DNA Repair 10:877–886
Alvi NK, Foiles PG, Williams GM (1990) Inhibition of repair of O6-methyldeoxguanosine and enhanced mutagenesis in rat-liver epithelial cells. Mutat Res 230:219–226
Seo KY, Nagalingam A, Tiffany M, Loechler EL (2005) Mutagenesis studies with four stereoisomeric N2-dG benzo[a]pyrene adducts in the identical 5′-CGC sequence used in NMR studies: G>T mutations dominate in each case. Mutagenesis 20:441–448
Zhao B, Wang J, Geacintow NE, Wang Z (2006) Poleta, Polzeta and Rev1 together are required for G to T transversion mutations induced by the (+)-and (−)-trans-anti-BPDE-N2-dG DNA adducts in yeast cells. Chem Res Toxicol 34:417–425
Cheng KC, Hahill DS, Kasai H, Nishimura S, Loeb LA (1992) 8-Hydroxyguanine, an abundant form of oxidative DNA damage, causes G-T and A-C substitutions. J Biol Chem 267:166–172
Kalam MA, Haraguchi K, Chandani S, Loechler EL, Moriya M, Greenberg MM, Basu AK (2006) Genetic effects of oxidative DNA damages: comparative mutagenesis of the imidazole ring-opened formamidopyrimidines (Fapy lesions) and 8-oxo-purines in simian kidney cells. Nucleic Acids Res 43:2305–2315
Shibata MA, Shirai T, Ogawa K, Takahashi S, Wild CP, Montesano R, Tsuda H, Ito N (1994) DNA methylation adduct formation and H-ras gene mutations in progression of N-buthyl-N-(4-hydroxybutyl)nitrosamine-induced bladder tumors caused by a single exposure to N-methyl-N-nitrosourea. Carcinogenesis 15:2965–2968
Hamid S, Eckert KA (2005) Effect of DNA polymerase beta loop variants on discrimination of O6-methyldeoxyguaosine modification present in the nucleotide versus template substrate. Biochemistry 44:10378–10387
Snyderwine EG, Roller PP, Adamson RH, Sato S, Thorgeirsson SS (1988) Reaction of N-hydroxylamine and N-acetoxy derivatives of 2-amino-3-methylimidazolo[4,5-f]quinolone with DNA. Synthesis and identification of N-(deoxyguanosin-8-yl)-IQ. Carcinogenesis 9:1061–1065
Turesky RJ, Markovic J, Aeschlimann JM (1996) Formation and differential removal of C-8 and N2-guanine adducts of the food carcinogen 2-amino-3-methylimidazo[4,5-f]quinolone in the liver, kidney, and colorectum of the rat. Chem Res Toxicol 9:397–402
Jamin EL, Arquier D, Canlet C, Rathahao E, Tulliez J, Debrauwer L (2007) New insights in the formation of deoxynucleoside adducts with the heterocyclic aromatic amines PhIP and IQ by means of ion trap MSn and accurate mass measurement of fragment ions. J Am Soc Mass Spectrom 18:2107–2118
Kanaly RA, Hanaoka T, Sugimura H, Toda H, Matsui S, Matsuda T (2006) Development of the adductome approach to detect DNA damage in humans. Antioxid Redox Signal 8:993–1001
Inoue K, Yoshida M, Takahashi M, Shibutani M, Takagi H, Hirose M, Nishikawa A (2009) Induction of kidney and liver cancers by the natural food additive madder color in a 2-year rat carcinogenicity study. Food Chem Toxicol 47:1400
Kawasaki Y, Goda Y, Yoshihira K (1992) The mutagenic constituents of Rubia tinctorum. Chem Pharm Bull 40:1504–1509
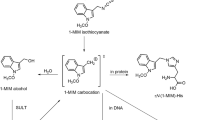
Ishii Y, Okamura T, Inoue T, Fukuhara K, Umemura T, Nishikawa A (2010) Chemical structure determination of DNA bases modified by active metabolites of lucidin-3-O-primeveroside. Chem Res Toxicol 23:134–141
Inoue K, Yoshida M, Takahashi M, Fujimoto H, Ohnishi K, Nakashima K, Shibutani M, Hirose M, Nishikawa A (2009) Possible contribution of rubiadin, a metabolite of madder color, to renal carcinogenesis in rats. Food Chem Toxicol 47:752–759
Ishii Y, Inoue K, Takasu S, Jin M, Matsushita K, Kuroda K, Fukuhara K, Nishikawa A, Umemura T (2012) Determination of lucidin-specific DNA adducts by liquid chromatography with tandem mass spectrometry in the livers and kidneys of rats given lucidin-3-O-primeveroside. Chem Res Toxicol 25:1112–1118
Tasaki M, Umemura T, Suzuki Y, Hibi D, Inoue T, Okamura T, Ishii Y, Maruyama S, Nohmi T, Nishikawa A (2010) Oxidative DNA damage and reporter gene mutation in the livers of gpt delta rats given non-genotoxic hepatocarcinogens with cytochrome P450-inducible potency. Cancer Sci 101:2525–2530
Poginsky B, Westendorf J, Blomeke B, Marquardt H, Hewer A, Grover PL, Phillips DH (1991) Evaluation of DNA-binding activity of hydroxyanthraquinones occurring in Rubia tinctrum L. Carcinogenesis 12:1265–1271
Kaur P, Chandel M, Kumar S, Kumar N, Kaur S (2010) Modulatory role of alizarin from Rubia cordifolia L. against genotoxicity of mutagens. Food Chem Toxicol 48:320–325
Acknowledgments
This work was supported part by a Grant-in-Aid from the Ministry of Health, Labour and Welfare, Japan.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 353 kb)
Rights and permissions
About this article
Cite this article
Ishii, Y., Takasu, S., Kuroda, K. et al. Combined application of comprehensive analysis for DNA modification and reporter gene mutation assay to evaluate kidneys of gpt delta rats given madder color or its constituents. Anal Bioanal Chem 406, 2467–2475 (2014). https://doi.org/10.1007/s00216-014-7621-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-014-7621-2