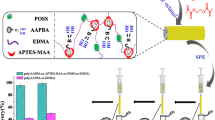
Abstract
A novel strategy for preparation of a boronate affinity hybrid monolith was developed using a Cu(I)-catalyzed 1,3-dipolar azide–alkyne cycloaddition (CuAAC) reaction of an alkyne–boronate ligand with an azide-functionalized monolithic intermediate. An azide-functionalized hybrid monolith was first synthesized via a single-step procedure to provide reactive sites for click chemistry; then the alkyne–boronate ligands were covalently immobilized on the azide-functionalized hybrid monolith via an in-column CuAAC reaction to form a boronate affinity hybrid monolith under mild conditions. The boronate affinity monolith was characterized and evaluated by means of elemental analysis, Fourier transform infrared spectroscopy, and scanning electron microscopy. The boronate affinity hybrid monolith exhibited excellent specificity toward nucleosides and glycoproteins, which were chosen as test cis-diol-containing compounds under neutral conditions. The binding capacity of the monolith for the glycoprotein ovalbumin was 2.36 mg · g-1 at pH 7.0. The practicability of the boronate affinity hybrid monolithic material was demonstrated by specific capture of the glycoproteins ovalbumin and ovotransferrin from an egg sample.

A novel strategy for preparation of boronate affinity hybrid monolith was developed by utilizing Cu(I)-catalyzed 1,3-dipolar azide-alkyne cycloaddition reaction (CuAAC). The obtained boronate affinity hybrid monolith exhibited excellent performance for isolation and enrichment of nucleosides and glycoproteins and was successfully employed to specific capture of glycoproteins from the egg sample









Similar content being viewed by others
References
Parodi AJ (2000) Annu Rev Biochem 69:69–93
Helenius A, Aebi M (2001) Science 291:2364–2369
Reis CA, Osorio H, Silva L, Gomes C, David L (2010) J Clin Pathol 63:322–329
Narimatsu H, Sawaki H, Kuno A, Kaji H, Ito H, Ikehara Y (2010) FEBS J 277:95–105
Hägglund P, Bunkenborg J, Elortza F, Jensen ON, Roepstorff P (2004) J Proteome Res 3:556–566
Jmeian Y, Hammad LA, Mechref Y (2012) Anal Chem 84:8790–8796
Chen R, Jiang XN, Sun DG, Han GH, Wang FJ, Ye ML, Wang LM, Zou HF (2009) J Proteome Res 8:651–661
Monzo A, Bonn GK, Guttman A (2007) Trends Anal Chem 26:423–432
Feng S, Yang N, Pennathur S, Goodison S, Lubman DM (2009) Anal Chem 81:3776–3783
Kaji H, Saito H, Yamauchi Y, Shinkawa T, Taoka M, Hirabayashi J, Kasai K, Takahashi N, Isobe T (2003) Nat Biotechnol 21:667–672
Tang J, Liu YC, Qi DW, Yao GP, Deng CH, Zhang XM (2009) Proteomics 9:5046–5055
Lin ZA, Zheng JN, Lin F, Zhang L, Cai ZW, Chen GN (2011) J Mater Chem 21:518–524
Lin ZA, Zheng JG, Xia ZW, Yang HH, Zhang L, Chen GN (2012) RSC Adv 2:5062–5065
Zhang XH, He XW, Chen LX, Zhang YK (2012) J Mater Chem 22:16520–16526
Li HY, Liu Z (2012) Trends Anal Chem 37:148–161
Liu X-C, Scouten WH (2000) In: Bailon P, Ehrlich GK, Fung W-J, Berthold W (eds) Affinity chromatography: methods and protocols. Methods in molecular biology, vol 147. Humana, Totowa, pp 119–128
Lin ZA, Pan JL, Yang HH, Cai ZW, Zhang L, Chen GN (2011) Chem Commun 47:9675–9677
Yang F, Lin ZA, He XW, Chen LX, Zhang YK (2011) J Chromatogr A 1218:9194–9201
Liu YC, Ren LB, Liu Z (2011) Chem Commun 47:5067–5069
Chen M, Lu Y, Ma Q, Guo L, Feng YQ (2009) Analyst 134:2158–2164
Lin ZA, Pan JL, Lin Y, Huang H, Cai ZW, Zhang L, Chen GN (2011) Analyst 136:3281–3288
Ren LB, Liu YC, Dong MM, Liu Z (2009) J Chromatogr A 1216:8421–8425
Li HY, Wang HY, Liu YC, Liu Z (2012) Chem Commun 48:4115–4117
Li QJ, Lü CC, Li HY, Liu YC, Wang HY, Wang X, Liu Z (2012) J Chromatogr A 1256:114–120
Kolb HC, Finn MG, Sharpless KB (2001) Angew Chem Int Ed 40:2004–2021
Moses JE, Moorhouse AD (2007) Chem Soc Rev 36:1249–1262
Slater M, Snauko M, Svec F, Fréchet JMJ (2006) Anal Chem 78:4969–4975
Huang HX, Jin Y, Xue MY, Yu L, Fu Q, Ke YX, Chu CH, Liang XM (2009) Chem Commun 6973–6975
Moni L, Ciogli A, Acquarica ID, Dondoni A, Gasparrini F, Marra A (2010) Chem Eur J 16:5712–5722
Suksrichavalit T, Yoshimatsu K, Prachayasittikul V, Bulow L, Ye L (2010) J Chromatogr A 1217:3635–3641
White MA, Johnson JA, Koberstein JT, Turro NJ (2006) J Am Chem Soc 128:11356–11357
Jian GQ, Liu YX, He XW, Chen LX, Zhang YK (2012) Nanoscale 4:6336–6342
Chu CH, Liu RH (2011) Chem Soc Rev 40:2177–2188
Guerrouache M, Millot MC, Carbonnier B (2009) Macromol Rapid Commun 30:109–113
Slater MD, Fréchet JMJ, Svec F (2009) J Sep Sci 32:21–28
Sun XL, Lin D, He XW, Chen LX, Zhang YK (2010) Talanta 82:404–408
Sun XL, He XW, Chen LX, Zhang YK (2011) Anal Bioanal Chem 399:3407–3413
Wang FJ, Dong J, Jiang XG, Ye ML, Zou HF (2007) Anal Chem 79:6599–6606
Acknowledgments
The authors are grateful to the National Basic Research Program of China (no. 2012CB910601), the National Natural Science Foundation of China (no. 20935001, 21275080), the Research Fund for the Doctoral Program of Higher Education of China (no. 20120031110007), and the Natural Science Foundation of Tianjin (no. 10JCZDJC17600).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Yang, F., Mao, J., He, X.W. et al. Preparation of a boronate-functionalized affinity hybrid monolith for specific capture of glycoproteins. Anal Bioanal Chem 405, 5321–5331 (2013). https://doi.org/10.1007/s00216-013-6917-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-013-6917-y