Abstract
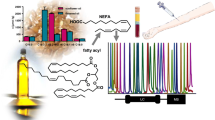
A combinative method using fingerprint analysis (FA) and multi-ingredients quantification (MIQ) was developed and validated for the quality control of Yinhuang (YH) preparations including granule, capsule, and lozenge by high-performance liquid chromatography coupled with diode array detection (HPLC-DAD). Common peaks with or without standard references in FA were confirmed or identified by electrospray ionization tandem mass spectrometry (ESI-MS/MS). The chromatographic separations were achieved on a Sepax GP-C18 column (250 mm × 4.6 mm i.d., 5 μm) with a gradient elution using a mixture of 0.1 % formic acid methanol solution and 0.1 % formic acid water solution. In quantitative analysis, nine bioactive constituents (chlorogenic acid, caffeic acid, luteoloside, baicalin, luteolin, wogonoside, baicalein, wogonin, and oroxylin A) were simultaneously determined. The detection wavelength was set at 275 nm, 320 nm, and 350 nm according to the absorption properties of the nine quantified compounds. The linearity, recovery, intraday and interday precision, accuracy, limit of detection (LOD) and quantification (LOQ), repeatability and stability were all tested and good results were obtained. In the FA, 320 nm was selected. The correlation coefficients of similarity were determined on the basis of the relative retention time (RRT) and relative peak area (RPA) of 20 common peaks in chromatographic fingerprints. Results indicated that both the RRT and RPA of 20 common peaks shared a close similarity. From the 20 common peaks, 18 compounds, including the nine quantified compounds, were identified or tentatively characterized by comparing their retention times, UV spectra, and MS spectra with those of standard compounds or literature data. The study not only presents a powerful and reliable analytical tool for the quality control of YH preparations, but also provides the chemical evidence for revealing the material basis of their therapeutic effects.

The HPLC-DAD and LC-ESI-MS/MS chromatogram of Yinhuang granule







Similar content being viewed by others
References
Huang ZL, Huang XY, Zheng Q (2010) Medical Recapitulate 16:386–388
Ma HD, Shi GB (2009) Pharmaceut Care Res 9:374–376
Li P, Qi LW, Wen XD, Sheng LH (2007) Chin J Nat Med 5:1–9
Su W, Xu ZY (2007) Shanghai J Tradit Chin Med 41(9):87–88
Li YF, Liu L, Cheng YY (2007) Chromatographia 65:749–755
He Q, Hu XJ, Cheng YY (2006) J Pharm Biomed Anal 41:485–492
Xie J, Wang W, Zhang Y, Bai Y, Yang Q (2007) J Pharm Biomed Anal 45:450–455
Liu AH, Lin YH, Yang M, Guo H, Guan SH, Sun JH, Guo DA (2007) J Chromatogr B 846:32–41
FDA (2000) Guidance for Industry. Botanical drug products (draft guidance). FDA, Rockville
EMEA (2006) Guidance on quality of herbal medicinal products. European Medicines Agency, London
State Food and Drug Administration of China (2002) Requirements for studying fingerprints of traditional Chinese medicine injection. SFDA, Beijing
Xie B, Gong T, Tang M, Mi D, Zhang X, Liu J, Zhang Z (2008) J Pharm Biomed Anal 48:1261–1266
Xu CJ, Liang YZ, Chau FT, Heyden YV (2006) J Chromatogr A 1134(1–2):253–259
Xu L, Han X, Qi Y, Xu Y, Yin L, Peng J, Liu K, Sun C (2009) Anal Chim Acta 633:136–148
Li Y, Wu T, Zhu J, Wan L, Yu Q, Li X, Cheng Z, Guo C (2010) J Pharm Biomed Anal 52:597–602
Duckstein SM, Stintzing FC (2011) Anal Bioanal Chem 401:677–688
Wei H, Sun L, Tai Z, Gao S, Xu W, Chen W (2010) Anal Chim Acta 662(1):97–104
Han J, Ye M, Guo H, Yang M, Wang BR, Guo DA (2007) J Pharm Biomed Anal 44:430–438
Zhang W, Saif MW, Dutschman GE, Li X, Lam W, Bussom S, Jiang Z, Ye M, Chu E, Cheng YC (2010) J Chromatogr A 1217(37):5785–5793
He WY, Gao RM, Li XQ, Jiang JD, Li YH (2010) Yao Xue Xue Bao 45:395–398
Li XQ, Sun XH, Cai S, Ying XX, Li FM (2009) Yao Xue Xue Bao 44(8):895–904
Han J, Ye M, Xu M, Sun J, Wang B, Guo D (2007) J Chromatogr B 848:355–362
National Pharmacopoeia Committee (2010) Chinese pharmacopoeia, vol 1. China Medical Science Press, Beijing
Liu EH, Liu Q, Chu C, Li P (2011) J Sep Sci 34:2566–2575
Bai Y, Zhang W, Wang X (2010) Zhongguo Zhong Yao Za Zhi 35:166–168
Yang X, Han FM, Cheng ZY, Chen Y (1999) Se Pu 17:573–575
ICH (1996) Guidance for Industry. Q2B validation of analytical procedures: methodology. ICH, Rockville
Ohmishi M, Morishita H, Iwahashi H (1994) Phytochemistry 36:579–583
Sakuma H, Matsushima S, Munakata S, Sugawara S (1982) Agric Biol Chem 46:1311–1317
Paganga G, Miller N, Rice-Evans CA (1999) Free Radic Res 30:153–162
Yan X, Suzuki M, Ohnishi-Kameyama M, Sada Y, Nakanishi T, Nagata T (1999) J Agric Food Chem 47:4711–4713
Yu J, Vasanthan T, Temelli F (2001) J Agric Food Chem 49:4352–4358
Deyama T, Ikawa T, Kigagawa S, Nishibe S (1987) Chem Pharm Bull 35:1785–1789
Schram K, Miketova P, Slanina J, Humpa O, Taborska E (2004) J Mass Spectrom 39:384–395
Ye M, Yan Y, Guo DA (2005) Rapid Commun Mass Spectrom 19:1469–1484
Löffler C, Sahm A, Wray V, Czygan FC, Proksch P (1995) Biochem Syst Ecol 23:121–128
Song Y, Li SL, Wu MH, Li HJ, Li P (2006) Anal Chim Acta 564:211–218
Li XQ, Sun XH, Cai S, Ying XX, Li FM (2009) Yao Xue Xue Bao 44:895–904
Horvath CR, Martos PA, Saxena PK (2005) J Chromatogr A 1062:199–207
Fabre N, Rustan I, de Hoffmann E, Quetin-Leclercq J (2001) J Am Soc Mass Spectrom 12:707–715
Acknowledgments
The authors acknowledge support for this work by a Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions, the Xuzhou Natural Science Foundation of China (no. XX10A052), and Jiangsu Aosaikang Foundation for Clinical Pharmacy (201012).
Author information
Authors and Affiliations
Corresponding author
Additional information
Hui Chen and Xu Chen contributed equally to this work.
Rights and permissions
About this article
Cite this article
Chen, H., Chen, X., Han, Q. et al. A new strategy for quality control and qualitative analysis of Yinhuang preparations by HPLC-DAD-MS/MS. Anal Bioanal Chem 404, 1851–1865 (2012). https://doi.org/10.1007/s00216-012-6281-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-012-6281-3