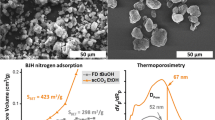
Abstract
The analysis of the porosity of materials is an important and challenging field in analytical chemistry. The gas adsorption and mercury intrusion methods are the most established techniques for quantification of specific surface areas, but unfortunately, dry materials are mandatory for their applicability. All porous materials that contain water and other solvents in their functional state must be dried before analysis. In this process, care has to be taken since the removal of solvent bears the risk of an incalculable alteration of the pore structure, especially for soft materials. In the present paper, we report on the use of small-angle X-ray scattering (SAXS) as an alternative analysis method for the investigation of the micro and mesopores within cellulose beads in their native, i.e., water-swollen state; in this context, they represent a typical soft material. We show that even gentle removal of the bound water reduces the specific surface area dramatically from 161 to 109 m2 g−1 in cellulose bead sample type MT50 and from 417 to 220 m2 g−1 in MT100. Simulation of the SAXS curves with a bimodal pore size distribution model reveals that the smallest pores with radii up to 10 nm are greatly affected by drying, whereas pores with sizes in the range of 10 to 70 nm are barely affected. The SAXS results were compared with Brunauer–Emmett–Teller results from nitrogen sorption measurements and with mercury intrusion experiments.

Volume-weighted pore size distribution of wet and dry cellulose beads derived from small-angle X-ray scattering experiments using a bimodal pore structure model







Similar content being viewed by others
References
O’Neil IL, Reichardt E (1951) Method of producing cellulose pellets. United States of America
Peska J, Stamberg J, Hradil J, Ilavsky M (1976) J Chromatogr 125:455
Luo XG, Zhang LN (2010) J Chromatogr A 1217:5922
Boeden HF, Pommerening K, Becker M, Rupprich C, Holtzhauer M, Loth F, Muller R, Bertram D (1991) J Chromatogr 552:389
Barrett EP, Joyner LG, Halenda PP (1951) J Am Chem Soc 73:373
Ravikovitch PI, Neimark AV (2001) Colloids Surface A 187:11
Lueking AD, Kim HY, Jagiello J, Bancroft K, Johnson JK, Cole MW (2009) J Low Temp Phys 157:410
Sonnauer A, Hoffmann F, Froba M, Kienle L, Duppel V, Thommes M, Serre C, Ferey G, Stock N (2009) Angew Chem Int Ed 48:3791
Brunauer S, Emmett PH, Teller E (1938) J Am Chem Soc 60:309
Emmerling A, Fricke J (1992) J Non-Cryst Solids 145:113
Porod G (1951) Kolloid Z Z Polym 124:83
Porod G (1952) Kolloid Z Z Polym 125:51
Smarsly B, Antonietti M, Wolff T (2002) J Chem Phys 116:2618
Stribeck N, Ruland W (1978) J Appl Crystallogr 11:535
Siemann U, Ruland W (1982) Colloid Polym Sci 260:999
Ruland W (2001) Carbon 39:323
Glatter O (1977) J Appl Crystallogr 10:415
Jemian PR, Allen AJ (1994) J Appl Crystallogr 27:693
Ilavsky J, Jemian PR (2009) J Appl Crystallogr 42:347
Thommes M, Smarsly B, Groenewolt M, Ravikovitch PI, Neimark AV (2006) Langmuir 22:756
Egger CC, du Fresne C, Raman VI, Schadler V, Frechen T, Roth SV, Muller-Buschbaum P (2008) Langmuir 24:5877
Acknowledgements
C. Wieland thanks BAM Federal Institute for Materials Research and Testing for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thünemann, A.F., Klobes, P., Wieland, C. et al. On the nanostructure of micrometer-sized cellulose beads. Anal Bioanal Chem 401, 1101–1108 (2011). https://doi.org/10.1007/s00216-011-5176-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-011-5176-z