Abstract
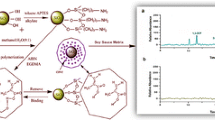
We report the use of nanospheres prepared by coating silica with molecularly imprinted polymer (MIP) for sulfamethoxazole (SMO). The resulting SiO2–SMO–MIP nanoparticles have highly improved imprinting and adsorption capacity, and can be used for separation and determination of sulfonamides in eggs and milk. In the synthesis, monodispersed SiO2 nanoparticles (Si–NP) of diameter 80 nm were amino-modified by reaction with 3-aminopropyltriethoxylsilane. The acryloyl monolayer was then grafted onto the amine-modified Si–NP. Finally, the MIP films were coated on to the surface of Si–NP by the copolymerization of the vinyl end groups with functional monomer (acrylamide), cross-linking agent (ethylene glycol dimethacrylate), initiator (azo-bis-isobutyronitrile), and template molecule (sulfamethoxazole). The resulting SiO2–SMO–MIP–NP were characterized by transmission electron microscopy, scanning electron microscopy, and Fourier transform infrared spectrometry. The adsorption properties were demonstrated by equilibrium rebinding experiments and Scatchard analysis. The results showed that the binding sites of the SiO2–SMO–MIP were highly accessible, and the maximum adsorption capacity of the SiO2–SMO–MIP for SMO was 20.21 mg g−1. The selectivity of the SiO2–SMO–MIP–NP obtained was elucidated by using SMO and structurally related sulfonamides. The results indicated that the SiO2–SMO–MIP had significant selectivity for SMO. The feasibility of removing SMO and sulfadiazine (SDZ) from food samples was proved by use of spiked milk and eggs. A method for the separation and determination of trace SMO and SDZ in milk and egg samples was developed, with recoveries ranging from 69.8% to 89.1%.

The nanosized and uniform silica-molecularly imprinted polymers (MIPs) for sulfamethoxazole (SMO) was described. The resulting SiO2/SMO-MIPs nanoparticles possess a highly improved imprinting effect and adsorption capacity, and can be applied in separation and determination of sulfonamides in the eggs and milk.








Similar content being viewed by others
References
Wang XL, Li K, Shi DS, Xiong N, Jin X (2007) J Agric Food Chem 55:2072–2078
Korpimäki T, Hagren V, Brockmann EC, Tuomola M (2004) Anal Chem 76:3091–3098
Furusawa N (2003) Anal Chim Acta 481:255–259
Stoev G, Michailova A (2000) J Chromatogr A 871:37–42
Preechaworapun A, Chuanuwatanakul S, Einaga Y, Grudpan K, Motomizu S, Chailapakul O (2006) Talanta 68:1726–1731
Chiavarino B, Crestoni ME, Marzio AD, Fornarini S (1998) J Chromatogr B 706:269–277
Reeves VB (1999) J Chromatogr B 723:127–137
Bogialli S, Curini R, Corcia AD, Nazzari M, Samperi R (2003) Anal Chem 75:1798–1804
Hows MEP, Perrett D, Kay J (1997) J Chromatogr A 768:97–104
Shao B, Dong D, Wu YN, Hu JY, Meng J, Tu XM, Xu SK (2005) Anal Chim Acta 546:174–181
Ramström O, Skudar K, Haines J, Patel P, Brüggemann O (2001) J Agric Food Chem 49:2105–2114
Baggiani C, Anfossi L, Giovannoli C (2007) Anal Chim Acta 591:29–39
Ye L, Haupt K (2004) Anal Bioanal Chem 378:1887–1897
Turiel E, Martín-Esteban A (2004) Anal Bioanal Chem 378:1876–1886
Tamayo FG, Turiel E, Martín-Esteban A (2007) J Chromatogr A 1152:32–40
Wulff G (2002) Chem Rev 102:1–27
Haupt K, Mosbach K (2000) Chem Rev 100:2495–2504
Lanza F, Sellergren B (2001) Chromatographia 53:599–611
Sun XL, He XW, Zhang YK, Chen LX (2009) Talanta 79:926–934
Zheng N, Li YZ, Chang WB, Wang ZM, Li TJ (2002) Anal Chim Acta 452:277–283
Zheng N, Li YZ, Wen MJ (2004) J Chromatogr A 1033:179–182
Liu XJ, Ouyang CB, Zhao R, Shangguan DH, Chen Y, Liu GQ (2006) Anal Chim Acta 571:235–241
Valtchev M, Palm BS, Schiller M, Steinfeld U (2009) J Hazard Mater 170:722–728
Chen ZY, Zhao R, Shangguan DH, Liu GQ (2005) Biomed Chromatogr 19:533–538
Davies MP, Biasi VD, Perrett D (2004) Anal Chim Acta 504:7–14
de Prada Guzmán-Vázquez A, Martínez-Ruiz P, Reviejo AJ, Pingarrón JM (2005) Anal Chim Acta 539:125–132
He JX, Wang S, Fang GZ, Zhu HP, Zhang Y (2008) J Agric Food Chem 56:2919–2925
Su SF, Zhang M, Li BL, Zhang HY, Dong XC (2008) Talanta 76:1141–1146
Guo LY, Jiang XM, Yang CL, Zhang HX (2008) Anal Bioanal Chem 391:2291–2298
Yilmaz E, Haupt K, Mosbach K (2000) Angew Chem Int Ed 39:2115–2118
Bossi A, Piletsky SA, Piletska EV, Righetti PG, Turner APF (2001) Anal Chem 73:5281–5286
Li Y, Yang HH, You QH, Zhuang ZX, Wang XR (2006) Anal Chem 78:317–320
Barahona F, Turiel E, Cormack PAG, Martí-Esteban A (2010) J Polym Sci A Polym Chem 48:1058–1066
Lee HY, Kim BS (2009) Biosens Bioelectron 25:587–591
Sulitzky C, Rückert B, Hall AJ, Lanza F, Unger K, Sellergren B (2002) Macromol 35:79–91
Titirici MM, Sellergren B (2006) Chem Mater 18:1773–1779
Sellergren B, Rückert B, Hall AJ (2002) Adv Mater 14:1204–1208
Rückert B, Hall AJ, Sellergren B (2002) J Mater Chem 12:2275–2280
Carter SR, Rimmer S (2004) Adv Funct Mater 14:553–561
Silvestri D, Borrelli C, Giusti P, Cristallini C, Ciardelli G (2005) Anal Chim Acta 542:3–13
Lu CH, Zhou WH, Han B, Yang HH, Chen X, Wang XR (2007) Anal Chem 79:5457–5461
Gao DM, Zhang ZP, Wu MH, Xie CG, Guan GJ, Wang DP (2007) J Am Chem Soc 129:7859–7866
Wang X, Wang LY, He XW, Zhang YK, Chen LX (2009) Talanta 78:327–332
Li L, He XW, Chen LX, Zhang YK (2009) Chem Asian J 4:286–293
Lu Q, Chen XM, Li N, Luo J, Jiang HJ, Chen LN, Hu Q, Du SH, Zhang ZP (2010) Talanta 81:959–966
Stöber W, Fink A, Bohn EJ (1968) J Colloid Interface Sci 26:62–69
Philipse AP, Vrij AJ (1989) J Colloid Interface Sci 128:121–136
Lanza F, Hall AJ, Sellergren B, Bereczki A, Horvai G, Bayoudh S, Cormack PAG, Sherrington DC (2001) Anal Chim Acta 435:91–106
Aerts MML, Beek WMJ (1998) J Chromatogr 435:97–112
Acknowledgements
The authors are grateful for financial support from 863 High Technology Project of China (2007AA10Z432, 2006AA10Z438), the National Basic Research Program of China (no. 2007CB914100), the National Natural Science Foundation of China (nos 20935001, 20875050), and the National Natural Science Foundation of Tianjin (no. 10JCZDJC17600).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Gao, R., Zhang, J., He, X. et al. Selective extraction of sulfonamides from food by use of silica-coated molecularly imprinted polymer nanospheres. Anal Bioanal Chem 398, 451–461 (2010). https://doi.org/10.1007/s00216-010-3909-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-010-3909-z