Abstract
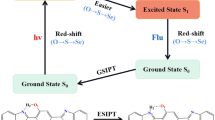
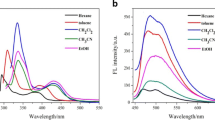
The effect of different substituents on the electronic spectra and excited state intramolecular hydrogen-bonding interactions of 2-(2-aminophenyl)benzothiazole (BTN) and its derivatives (BTN-1, BTN-2, BTN-3) were studied by using the density functional theory (DFT) and time-dependent DFT (TD-DFT) methods. The structural parameters, infrared vibrational frequencies, topological parameters, frontier molecular orbitals, absorption and emission spectra as well as potential energy curves in the ground (S0) and excited (S1) states were obtained. BTN and its derivatives are difficult to undergo ESIPT process since the reverse ESIPT barriers are about 6–7 kcal/mol lower than the corresponding forward ESIPT barriers. The experimental fluorescence emission wavelength of BTN is originated from its normal form, and is well reproduced theoretically. As the electron-donating ability of functional group increases from –CH3 to –OCH3, the absorption and fluorescence wavelengths red-shift more. The S0 and S1 energy gap is in the order of BTN > BTN-1 > BTN-2 > BTN-3, which is in good agreement with its red-shift values of absorption and emission peaks.







Similar content being viewed by others
References
Niu LY, Chen YZ, Zheng HR, Wu LZ, Tung CH, Yang QZ (2015) Chem Soc Rev 44:6143
Xuan W, Sheng C, Cao Y, He W, Wang W (2012) Angew Chem Int Ed 51:2282
Wang R (2012) Physiol Rev 92:791
Szabo C (2016) Nat Rev Drug Discov 15:185
Li L, Rose P, Moore PK (2011) Annu Rev Pharmacol Toxicol 51:169
Predmore BL, Kondo K, Bhushan S, Zlatopolsky MA, King AL, Aragon JP, Grinsfelder DB, Condit ME, Lefer DJ (2012) Am J Physiol Heart Circ Physiol 602:H2410
Szabó C (2007) Nat Rev Drug Discov 6:917
Yang G, Wu L, Jiang B, Yang W, Qi J, Cao K, Meng Q, Mustafa AK, Mu W, Zhang S, Snyder SH, Wang R (2008) Science 322:587
Lavu M, Bhushan S, Lefer DJ (2011) Clin Sci (Lond) 120:219
Kamoun P, Belardinelli MC, Chabli A, Lallouchi K, Chadefaux-Vekemans B (2003) Am J Med Genet A 116A:310
Environmental Health Criteria 19 (1981) Hydrogen sulfide, the international labor organization and the world health organization. WHO, Geneva, p 48. Sw, fr. 6.00
Lawrence NS, Davis J, Compton RG (2020) Talanta 52:771
Wardencki W (1998) J Chromatogr A 793:1
Mitchell TW, Savage JC, Gould DH (1993) J Appl Toxicol 13:389
Fogo JK, Popowsky M (1949) Anal Chem 21:732
Kroll AV, Smorchkov VI, Nazarenko AY (1994) Sensor Actuat B Chem 21:97
Garcı́a-Calzada M, Marbán G, Fuertes AB (1999) Anal Chim Acta 380:39
Choi MG, Cha S, Lee H, Jeon HL, Chang SK (2009) Chem Commun 47:7390
Choi MMF (1998) Analyst 123:1631
de Silva AP, Gunaratne HQN, Gunnlaugsson T, Huxley AJM, McCoy CP, Rademacher JT, Rice TE (1997) Chem Rev 97:1515
Carter KP, Young AM, Palmer AE (2014) Chem Rev 114:4564
Tang L, Zhou L, Liu A, Yan X, Zhong K, Liu X, Gao X, Li J (2021) Dyes Pigm 186:109034
Tang L, Zhou L, Yan X, Zhong K, Gao X, Li J (2020) J Photochem Photobiol A 387:112160
Li S, Cao D, Meng X, Hu Z, Li Z, Yuan C, Zhou T, Han X, Ma W (2020) J Photochem Photobiol A 392:112427
Chen L, Oh H, Wu D, Kim MH, Yoon J (2018) Chem Commun 54:2276
Karakuş E, Üçüncü M, Emrullahoğlu M (2016) Anal Chem 88:1039
Niu W, Fan L, Nan M, Li Z, Lu D, Wong MS, Shuang S, Dong C (2015) Anal Chem 87:2788
Montoya LA, Pluth MD (2014) Anal Chem 86:6032
Ren M, Deng B, Kong X, Zhou K, Liu K, Xu G, Lin W (2016) Chem Commun 52:6415
Yang Y, Yin C, Huo F, Zhang Y, Chao J (2014) Sensor Actuat B Chem 203:596
Kang J, Huo F, Yao Y, Yin C (2019) Dyes Pigm 171:107755
Wang J, Wen Y, Huo F, Yin C (2019) Sensor Actuat B Chem 297:126773
Elsayed S, de la Torre C, Santos-Figueroa LE, Marín-Hernández C, Martínez-Máñez R, Sancenón F, Costero AM, Gil S, Parra M (2015) Sensor Actuat B Chem 207:987
Jiang Y, Wu Q, Chang X (2014) Talanta 121:122
Wan Q, Song Y, Li Z, Gao X, Ma H (2013) Chem Commun 49:502
Wu Z, Li Z, Yang L, Han J, Han S (2012) Chem Commun 48:10120
Wang R, Yu F, Chen H, Wang L, Zhang W (2012) Chem Commun 48:11757
Wen Y, Huo F, Wang J, Yin C (2019) J Mater Chem B 7:6855
Zhang W, Huo F, Yin C (2019) Org Lett 21:5277
Qu X, Li C, Chen H, Mack J, Guo Z, Shen Z (2013) Chem Commun 49:7510
Hou F, Huang L, Xi P, Cheng J, Zhao X, Xie G, Shi Y, Cheng F, Yao X, Bai D, Zeng Z (2012) Inorg Chem 51:2454
Padalkar VS, Seki S (2016) Chem Soc Rev 45:169
Kwon JE, Park SY (2011) Adv Mater 23:3615
Han KL, Zhao GJ (2011) Hydrogen Bonding and Transfer in the Excited State. Wiley
Waluk J (2000) Conformational analysis of molecules in excited states. Wiley
Zhao J, Ji S, Chen Y, Guo H, Yang P (2012) Phys Chem Chem Phys 14:8803
Guan H, Zhang A, Li P, Xia L, Guo F (2019) ACS Omega 4:9113
Wu C, Hu X, Gu B, Yin P, Su W, Li Y, Lu Q, Zhang Y, Li H (2018) Anal Methods 10:604
Lu Y, Dong B, Song W, Kong X, Mehmood AH, Lin W (2019) Anal Methods 11:3301
Liu J, Chen X, Zhang Y, Gao G, Zhang X, Hou S, Hou Y (2018) New J Chem 42:12918
Kim JK, Bong SY, Park R, Park J, Jang DO (2022) Spectrochim Acta A Mol Biomol Spectrosc 278:121333
Santos GC, Rocha IO, Stefanello FS, Copetti JPP, Tisoco I, Martins MAP, Zanatta N, Frizzo CP, Iglesias BA, Bonacorso HG (2022) Spectrochim Acta A Mol Biomol Spectrosc 269:120768
Jia L, Liu Y (2020) Spectrochim Acta A Mol Biomol Spectrosc 242:118719
Hisham S, Tajuddin HA, Chee CF, Hasan ZA, Abdullah Z (2019) J Lumin 208:245
Frisch MJ, Truck GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09, Rev. D. 01.Gaussian, Inc, Wallingford CT
Cancès E, Mennucci B, Tomasi J (1997) J Chem Phys 107:3032
Cossi M, Barone V, Mennucci B, Tomasi J (1998) Chem Phys Lett 286:253
Padilla P, Vega C (1997) J Chem Phys 106:1997
Yanai T, Tew DP, Handy NC (2004) Chem Phys Lett 393:51
Zhao Y, Truhlar DG (2008) Theor Chem Acc 120:215
Lee CT, Yang WT, Parr RG (1988) Phys Rev B 37:785
Adam C, Barone V (1999) J Chem Phys 110:6158
Perdew JP, Chevary JA, Vosko SH, Jackson KA, Pederson MR, Singh DJ, Fiolhais C (1992) Phys Rev B 46:6671
Salzne U, Aydin A (2011) J Chem Theory Comput 7:2568
Lu T, Chen F (2012) J Comput Chem 33:580
Li Y, Wang L, Guo X, Zhang J (2015) J Comput Chem 36:2374
Tseng HW, Liu JQ, Chen YA, Chao CM, Liu KM, Chen CL, Lin TC, Hung CH, Chou YL, Lin TC, Wang TL, Chou PT (2015) J Phys Chem Lett 6:1477
Zhao GJ, Han KL (2008) Biophys J 94:38
Zhao J, Yao H, Liu J, Hoffmann MR (2015) J Phys Chem A 119:681
Yu F, Li P, Wang B, Han K (2013) J Am Chem Soc 135:7674
Stasyu AJ, Cyrański MK, Gryko DT, Solà M (2015) J Chem Theory Comput 11:1046
Kasha M (1950) Discuss Faraday Soc 9:14
Funding
This work did not receive project support.
Author information
Authors and Affiliations
Contributions
Data curation, writing—original draft preparation, visualization and investigation were performed by ZZ. Conceptualization, methodology, software, supervision, validation, writing—reviewing and editing were performed by HF. All authors approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, Z., Fang, H. Uncovering the effects of different substituents on excited state hydrogen-bonding interaction and fluorescent behavior in BTN system: a TD-DFT insight. Theor Chem Acc 142, 109 (2023). https://doi.org/10.1007/s00214-023-03054-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-023-03054-0