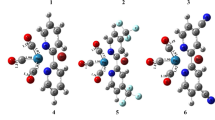
Abstract
Manganese tricarbonyl complexes are considered as promising compounds as they release carbon monoxide (CO) upon irradiation of light. Carbon monoxide-releasing molecules may supply a controlled amount of CO to the biological systems. Therefore, this research area is a hot topic in medicine, especially cancer treatment. The designing and analysis of carbon monoxide-releasing compounds can be done using various experimental and theoretical methods. We have performed density functional theory (DFT) calculations to identify and scrutinize such molecules to investigate the ability to release CO. In this report, we have taken Mn(I) tricarbonyl complexes that bear di(2-picolyl)amine unit with different kinds of electron-withdrawing nature ligands. We have used density functional theory (DFT) to optimize different complexes with the B3LYP/LANL2DZ basis set. DFT and time-dependent density functional theory (TD-DFT) calculations infer that the designed carbonyl complexes (2 to 8) will release CO efficiently. The calculated results also suggest the transfer of electron density from the electron-rich metal centers to π molecular orbitals of the ligand via strong metal-to-ligand charge transfer (MLCT) in the visible/near-IR region. The strong MLCT results weaken the metal-CO back bonding and promote the speedy CO-release.







Similar content being viewed by others
References
Toscani A, Hind C, Clifford M, Kim S-H, Gucic A, Woolley C, Saeed N, Rahman KM, Sutton JM, Castagnolo D (2021) Eur J Med Chem 213:113172
Turner JJ, George MW, Poliakoff M, Perutz RN (2022) Chem Soc Rev 51:5300–5329
Amorim AL, Guerreiro A, Glitz VA, Coimbra DF, Bortoluzzi AJ, Caramori GF, Braga AL, Neves A, Bernardes GJL, Peralta RA (2020) New J Chem 44:10892–10901
Motterlini R, Otterbein LE (2010) Nat Rev Drug Discovery 9:728–743
Foresti R, Bani-Hani MG, Motterlini R (2008) Intensive Care Med 34:649–658
Vincent JL (2003) Lancet 361:2068–2077
Cheng J, Jinming H (2021) ChemMedChem 16(24):3628–3634. https://doi.org/10.1002/cmdc.202100555
Heinemann SH, Hoshi T, Westerhausen M, Schiller A (2014) Chem Commun 50:3644–3660
Romao CC, Blattler WA, Seixas JD, Bernardes GJL (2012) Chem Soc Rev 41:3571–3583
Douglas CG, Haldane JS, Haldane JBS (1912) J Physiol 44:275–304
Haldane JB (1927) Biochem J 21:1068–1075
Mann BE (2012) Organometallics 31:5728–5735
Alberto R, Motterlini R (2007). Dalton Trans. https://doi.org/10.1039/b701992k
Shatzschneider U (2015) Br J Pharmacol 172:1638–1650
Askes SHC, Reddy GU, Wyrwa R, Bonnet S, Schiller A (2017) J Am Chem Soc 139:15292–15295
Reddy UG, Liu J, Gorls H, Askes SHC, Schiller A, Hoffmann P, Neugebauer U, Steinmetzer J, Kupfer S, Grafe S (2017) Chem Sci 8:6555–6560
Schatzschneider U (2011) Inorg Chim Acta 374:19–23
Zobi F (2013) Future. Med Chem 5:175–188
Compain JD, Bourrez M, Haukka M, Deronzier A, Chardon-Noblat S (2014) Chem Commun 50:2539–2542
Stamellou E, Storz D, Botov S, Ntasis E, Wedel J, Sollazzo S, Krämer BK, van Son W, Seelen M, Schmalz HG, Schmidt A (2014) Redox Biol 2:739–748
Betts JW, Roth P, Pattrick CA, Southam HM, La Ragione RM, Poole RK, Schatzschneider U (2020) Metallomics 10:1563–1575
Gessner N, Bäck AK, Knorr J, Nagel C, Marquetand P, Schatzschneider U, González L, Nuernberger P (2021) Phys Chem Chem Phys 23(42):24187–24199. https://doi.org/10.1039/D1CP03514
Benniston AC, Zeng L (2022) Dalton Trans 51:4202–4212
Fairlamb IJS, Lynam JM, Hirao T, Moriuchi T, Elsevier 2019,137–154
Aucott BJ, Eastwood JB, Hammarback LA, Clark IP, Sazanovich IV, Towrie M, Fairlamb IJS (2019) J M Lynam Dalton Trans 48:16426
Tinajero-Trejo M, Rana N, Nagel C, Jesse HE, Smith TW, Wareham LK, Hippler M, Schatzschneider U, Poole RK (2016) Antioxid Redox Signal 24:765–780
Nagel C, McLean S, Poole RK, Braunschweig H, Kramer T, Schatzschneider U (2014) Dalton Trans 43:9986–9997
Pfeiffer H, Sowik T, Schatzschneider U (2013) J Organomet Chem 734:17–24
Govender P, Pai S, Schatzschneider U, Smith GS (2013) Inorg Chem 52:5470–5478
Carrington SJ, Chakraborty I, Mascharak PK (2013) Chem Commun 49:11254–11256
Mohr F, Niesel J, Schatzschneider U, Lehmann CW, Anorg Z (2012) Allg Chem 638:543–546
Niesel J, Pinto A, Peindy HW, N’Dongo KM, Ott I, Gust R, Schatzschneider U (2008). Chem Commun. https://doi.org/10.1039/b719075a
Jiang Q, Xia Y, Barrett J, Mikhailovsky A, Wu G, Wang D, Shi P, Ford PC (2019) Inorg Chem 58:11066–11075
Rana N, Jesse HE, Tinajero-Trejo M, Butler JA, Tarlit JD, Milena L, Nagel C, Schatzschneider U, Poole RK (2017) Microbiology 163:1477
Betts J, Nagel C, Schatzschneider U, Poole R, La Ragione RM (2017) PLoS ONE 12:e186359
W. Huber, R. Linder, J. Niesel, U. Schatzschneider, B. Spingler, P. C. Kunz, (2012) Eur. J. Inorg. Chem., 3140–3146.
Kottelat E, Ruggi A, Zobi F (2016) Dalton Trans 45:6920–6927
Marques AR, Kromer L, Gallo DJ, Penacho N, Rodrigues SS, Seixas JD, Bernardes GJL, Reis PM, Otterbein SL, Ruggieri RA, Goncalves ASG, Goncalves AML, De Matos MN, Bento I, Otterbein LE, Blattler WA, Romao CC (2012) Organometallics 31:5810–5822
Ji X, Damera K, Zheng Y, Yu B, Otterbein LE, Wang B (2016) J Pharm Sci 105:406–416
Yuan Z, Yang X, Wang B (2021) Chem Sci 12:13013–13020
Romanski S, Kraus B, Guttentag M, Schlundt W, Rucker H, Adler A, Neudorfl J-M, Alberto R, Amslinger S, Schmalz H-G (2012) Dalton Trans 41:13862–13875
Aucott BJ, Ward JS, Andrew SG, Milani J, Whitwood AC, Lynam JM, Parkin A, Fairlamb IJS (2017) Inorg Chem 56:5431–5440
Fairlamb IJS, Duhme-Klair AK, Lynam JM, Moulton BE, O’Brien CT, Sawle P, Hammad J, Motterlini R (2006) Bioorg Med Chem Lett 16:995–998
I. J. S. Fairlamb, J. M. Lynam, B. E. Moulton, I. E. Taylor, A. K. Duhme-Klair, P. Sawle and R. Motterlini, (2007). Dalton Trans. 3603–3605.
Atkin AJ, Williams S, Sawle P, Motterlini R, Lynam JM, Fairlamb IJS, Atkin AJ, Williams S, Sawle P, Motterlini R, Lynam JM, Fairlamb IJS (2009) Dalton Trans 38:3653–3656
Zhang WQ, Atkin AJ, Thatcher RJ, Whitwood AC, Fairlamb IJS, Lynam JM (2009) Dalton Trans 38:4351–4358
Zhang WQ, Whitwood AC, Fairlamb IJS, Lynam JM (2010) Inorg Chem 49:8941–8952
Ji X, De La Cruz LKC, Pan Z, Chittavong V (2017) Chem Commun 53:9628–9631
Motterlini R, Mann BE, Scapens DA (2005) Expert Opin. Investig Drug 14:1305–1318
Reddy GU, Axthelm J, Hoffmann P, Taye N, Gläser S, Görls H, Hopkins SL, Plass W, Neugebauer U, Bonnet S et al (2017) J Am Chem Soc 139:4991–4994
Sakla R, Jose DA (2021) Inorg Chim Acta 516:120134
Mitra A, Jana G, Pal R, Gaikwad P, Sural S, Chattaraj PK (2021) Theoret Chem Acc 140:1–12
R. D. Dennington, T. A. Keith, J. M. Millam, (2008) GaussView 5.0.8, Gaussian
O’Boyle NM, Tenderholt AL, Langner KM (2008) J Comput Chem 29:839–845
Lu T, Chen F (2012) J Comput Chem 33:580–592
Cotton FA, Kraihanzel CS (1962) J Am Chem Soc 84:4432–4438
A. M. Mansour., Polyhedron.2016, 99, 109.
Mansour AM (2013) Inorg Chim Acta 436:394
Chakraborty I, Carrington SJ, Mascharak PK (2014) Acc Chem Res 47:2603–2611
Monari A, Assfeld X, Baley M, Gros PC (2011) J Phys Chem 115(15):3596–3603
Wang HT, Taufany F, Nachimuthu S, Jiang JC (2014) J Comput Aided Mol Des 28:565–575
Sakla R, Singh A, Kaushik R, Kumar P, Jose DA (2019) Inorg Chem 58:10761–10768
Li Y, Li H, Zhao X, Chen M (2010) J Phys Chem A 114:6972–6977
Duan YA, Geng Y, Li HB, Jin JL, Wu Y, Su ZM (2013) J Comput Chem 34:1611–1619
Beenken WJ, Pullerits T (2004) J Phys Chem B 108:6164–6169
Acknowledgements
Ajeet Singh acknowledges to Science and Engineering Research Board (SERB) New Delhi for financial support (Ref. no. CRG/2019/001032) under core research grant.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Author information
Authors and Affiliations
Contributions
RS and DAJ conceived the study. SKY and RK performed and analyze the DFT calculated data. RS, SKY, AS participated in interpretation and drafting the manuscript. AS and DAJ supervised the study. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Seth, R., Jose, D.A., Yadav, S.K. et al. Quest of new molecular frameworks for photoinduced carbon monoxide-releasing molecules: a computational prospective. Theor Chem Acc 141, 79 (2022). https://doi.org/10.1007/s00214-022-02937-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-022-02937-y