Abstract
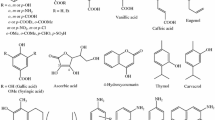
Chromone (4H-chromen-4-one, 4H-1-benzopyran-4-one) heterocyclic compound has been widely used in traditional medicine, which has anti-allergy, anti-inflammation, anti-diabetes, anti-tumor, antibacterial and other pharmacological effects. To estimate the scavenging activity on free radical of 3-styrylchromone derivatives, we considered the physical and chemical properties of the molecule structure, orbital, and thermodynamics. The antioxidant activity of 3-styrylchromone derivatives was studied by density functional theory (DFT) in gas and water. A series of quantum chemical parameters were calculated to describe molecular properties and clarify the radical scavenging mechanism. Our study suggests that the antioxidant activity of scavenging free radicals is largely affected by molecular structure, especially the number of hydroxyl groups, hydrogen bonds, and conjugation effects. 3-styrylchromone derivatives containing pyrogallol structure may have potential antioxidant activity. The antioxidant ability of catechol structure is greater than pyrogallol structure. 1,4-pyrrole also has a positive effect on free radical scavenging, but the scavenging effect of 2-OH and 5-OH introduced by antioxidants was not obvious. The excellent correlations between the structure and the mentioned DFT-based descriptors lead us to predict good antioxidants.





Similar content being viewed by others
Availability of data and material
All data generated or analyzed during this study are included in this article.
Code availability
All data, models, and code generated or used during the study appear in the submitted article.
References
M Dizdaroglu, P Jaruga, M Birincioglu, H Rodriguez (2002) Free radical-induced damage to dna: mechanisms and measurement 1,2 1this article is part of a series of reviews on “oxidative dna damage and repair.” the full list of papers may be found on the homepage of the journal. 2guest editor: miral dizdaroglu. Free Radical Biology and Medicine 32(11): 1102–1115
Knekt P, Kumpulainen J, Järvinen R, Rissanen H, Heliövaara M, Reunanen A, Hakulinen T, Aromaa A (2002) Flavonoid intake and risk of chronic diseases. Am J Clin Nutr 76(3):560–568
Gaspar A, Matos MJ, Garrido J, Uriarte E, Borges F (2014) Chromone: a valid scaffold in medicinal chemistry. Chem Rev 114:4960–4992
Abu-Hashem AA, Youssef MM (2011) Synthesis of new visnagen and khellin furochromone pyrimidine derivatives and their antiinflammatory and analgesic activity. Molecules 16:1956–1972
Keri RS, Budagumpi S, Pai RK, Balakrishna RG (2014) Chromones as a privileged scaffold in drug discovery: a review. Eur J Med Chem 78:340–374
Filipe P, Silva AMS, Morliere P, Brito CM, Patterson LK, Hug GL, Silva JN, Cavaleiro JAS, Maziere JC, Freitas JP, Santus R (2004) Polyhydroxylated 2-styrylchromones as potent antioxidants. Biochem Pharmacol 67(12):2207–2218
Momoi K, Sugita Y, Ishihara M, Satoh K, Kikuchi H, Hashimoto K, Yokoe I, Nishikawa H, Fujisawa S, Sakagami H (2005) Cytotoxic activity of styrylchromones against human tumor cell lines. Vivo 19(1):157
Conti C, Desideri N (2010) New 4h-chromen-4-one and 2h-chromene derivatives as anti-picornavirus capsid-binders. Bioorg Med Chem 18:6480–6488
Koichi T, Ryo I, Yoshiaki S (2014) Synthesis and biological evaluation of 3-styrylchromone derivatives as free radical scavengers and α-glucosidase inhibitors. Chem. Pharma. Bull. 62(8):815–816
Conti C, Desideri N (2010) New 4H-chromen-4-one and 2Hchromene derivatives as anti-picornavirus capsid-binders. Bioorg Med Chem 18:6480–6488
Shimada C, Uesawa Y, Ishii-Nozawa R, Ishihara M, Kagaya H, Kanamoto T, Sakagami H (2014) Quantitative structure–cytotoxicity relationship of 3-styrylchromones. Anticancer Res 34:5405–5411
Sonawane SA, Chavan VP, Shingare MS, Karale BK (2002) Synthesis of 3-methyl-4-[(1, 3-diphenyl-1H-pyrazol-4-yl) methylene]-1-ph enyl-pyrazolin-5 (4H)-ones and some 3-styrylchromones. Ind J Heterocycl Chem 12:65–66
Takao K, Ishikawa R, Sugita Y (2014) Synthesis and biological evaluation of 3-styrylchromone derivatives as free radical scavengers and α-glucosidase inhibitors. Chem Pharm Bull 62:810–815
Akmak E, In DZ (2020) A theoretical evaluation on free radical scavenging activity of 3-styrylchromone derivatives: the dft study. J Mol Model 26(5):1–11
Guo Q, Zhao BL, Shen SR, Hou JW, Hu JG, Xin WJ (1999) Biochim Biophys Acta 1427:13–23
Taubert D, Breitenbach T, Lazar A, Censarek P, Harlfinger S, Berkels R, Foti M, Piattelli M, Baratta MT, Ruberto G (1996) J Agric Food Chem 44:497–501
Thaipong K, Boonprakob U, Crosby K, Cisneros-Zevallos L, Byrne DH (2012) Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating antioxidant activity from guava fruit extracts. J Food Compos Anal 19(6–7):669–675
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas Ö, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09, Revision C 01. Gaussian Inc, Wallingford
Lee C, Yang W, Parr RG (1988) Development of the colle-salvetti correlation-energy formula into a functional of the electron density. Phys. Res. B 37(2):785–789
Becke AD (1993) Density-functional thermochemistry III The role of exact exchange. J Chem Phys 98(7):5648
Zhao Y, Truhlar DG (2008) How well can new-generation density functionals describe the energetics of bond-dissociation reactions producing radicals? J Phys Chem A 112(6):1095–1099
Cances E, Mennucci B, Tomasi J (1997) A new integral equation formalism for the polarizable continuum model: theoretical background and applications to isotropic and anisotropic dielectrics. J Chem Phys 107:3032–3041
Szeląg M, Mikulski D, Molski M (2012) Quantum-chemical investigation of the structure and the antioxidant properties of α-lipoic acid and its metabolites. J Mol Model 18:2907–2916
Wright JS, Johnson ER, Dilabio GA (2001) Predicting the activity of phenolic antioxidants: theoretical method, analysis of substituent effects, and application to major families of antioxidants. J Am Chem Soc 123(6):1173–1183
Stepanic V, Troselj KG, Lucic B, Markovic Z, Amic D (2013) Bond dissociation free energy as a general parameter for flavonoid radical scavenging activity. Food Chem 141(2):1562–1570
Xue Y, ZhengY AL, Dou Y, Liu Y (2014) Density functional theory study of the structure-antioxidant activity of polyphenolic deoxybenzoins. Food Chem 151:198–206
Wright JS, Johnson ER, DiLabio GA (2001) Predicting the activity of phenolic antioxidants: theoretical method, analysis of substituent effects, and application to major families of antioxidants. J Am Chem Soc 123:1173–1183
Klein E, Rimarcik J, LukesV, (2009) DFT/B3LYP study of the O-H bond dissociation enthalpies and proton affinities of para-and metasubstituted phenols in water and benzene. Acta Chim Slov 2:37–51
Parker VD (1992) Homolytic bond (HA) dissociation free energies in solution. Applications of the standard potential of the (H+/H. bul.) couple. J Am Chem Soc 114:7458–7462
Bizarro MM, Cabral BC, Dos Santos RB, Simões JM (1999) Substituent effects on the O-H bond dissociation enthalpies in phenolic compounds: agreements and controversies+ erratum. Pure Appl Chem 71:1249–1256
Janak JF (1978) Proof that∂ e∂ n i= ε in density-functional theory. Phys Rev B 18:7165
Parr RG, Yang W (1989) Density functional theory of atoms and molecules. Oxford University Press, New York
Pearson RG (1997) Chemical hardness: applications from molecules to solids. Wiley-VCH, Weinheim
Pearson RG (1973) Hard and soft acids and bases. Dowden Hutchinson and Ross, Stroudsberg
Parr RG, Pearson RG (1983) J Am Chem Soc 105:7512–7516
Pearson RG (1988) Absolute electronegativity and hardness: application to inorganic chemistry. Inorg Chem 27:734–740
Parr RG, Szentpaly LV, Liu S (1999) J Am Chem Soc 121:1922–1924
Sıdır I, Sıdır YG, Kumalar M, Taşal E (2010) Ab initio Hartree-Fock and density functional theory investigations on the conformational stability, molecular structure and vibrational spectra of 7-acetoxy-6-(2,3-dibromopropyl)-4,8-dimethylcoumarin molecule. J Mol Struct 964:134–151
Barakat A, Al-Majid AM, Soliman SM, Mabkhot YN, Ali M, Ghabbour HA, Fun HK, Wadood A (2015) Synthesis and antibacterial and antifungal activities of n-(tetra-o-acetyl-β-d-glucopyranosyl)thiosemicarbazones of substituted 4-formylsydnones. Chem Cent J 9:1–15
Mukai K, Oka W, Watanabe K, Egawa Y, Nagaoka S-i, Terao J (1997) Kinetic study of free-radical-scavenging action of flavonoids in homogeneous and aqueous triton x–100 micellar solutions. J Phys Chem A 101:3746–3753
Liu Q, Trotter J, Zhang J, Peters MM, Cheng H, Bao J, Han X, Weeber EJ, Bu G (2010) Neuronal lrp1 knockout in adult mice leads to impaired brain lipid metabolism and progressive, age-dependent synapse loss and neurodegeneration. J Neuroence 30(50):17068–17078
Wang G, Xue Y, An L, Zheng Y, Dou Y, Zhang L, Liu Y (2015) Theoretical study on the structural and antioxidant properties of some recently synthesised 2,4,5-trimethoxy chalcones. Food Chem 171:89–97
Acknowledgements
This work is supported by the team of syngas catalytic conversion of Shaanxi University of Technology, the project of education department of Shaanxi province (20JK0565), Natural Science Basic Research Program of Shaanxi (Program No.2021JQ-751), the Shaanxi Province Education Ministry Research Foundation (19JS010), the Project of Shaanxi University of Technology (SLGKY2009).
Funding
This work is supported by the team of syngas catalytic conversion of Shaanxi University of Technology, the project of education department of Shaanxi province (20JK0565), Natural Science Basic Research Program of Shaanxi (Program No.2021JQ-751), the Shaanxi Province Education Ministry Research Foundation (19JS010), the project of Shaanxi University of Technology (SLGKY2009).
Author information
Authors and Affiliations
Contributions
Pei Huang contributed to the conception of the study and performed the experiment; Lingxia Jin contributed significantly to analysis and manuscript preparation; Jiufu Lu performed the data analyses and wrote the manuscript; Yanhong Gao and Shaobo Guo helped perform the analysis with constructive discussions.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Huang, P., Jin, LX., Lu, JF. et al. A theoretical study of radical scavenging antioxidant activity of 3-styrylchromone derivatives using DFT based on quantum chemical descriptors. Theor Chem Acc 140, 48 (2021). https://doi.org/10.1007/s00214-021-02754-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-021-02754-9