Abstract
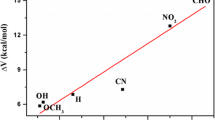
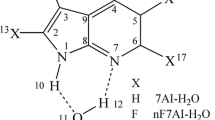
The substituted effect on the first excited-state proton transfer (ESPT) process in 2,7-diazaindole-H2O (2,7-DAI-H2O) complex in water was studied in detail at the TD-M06-2X/6–311 + G(d, p) level. The frontier molecular orbital, geometries, reaction mechanism and energies of ESPT process with different substituent have been analyzed. ESPT process in the title complex occurred concertedly but highly asynchronously no matter of the electronic nature of substituent. The absorption and fluorescence peaks, H-bond distances, asynchronicity of ESPT and barrier height were affected by the substituent. The Hammett’s substituent constant had linear correlation with the difference between the sum of N1−O11 and O11−N7 distances in the reactant and that in the TS and Mulliken charge of H3O+.






Similar content being viewed by others
References
Dybala-Defratyka A, Paneth P, Pu J, Truhlar D (2004) J Phys Chem A 108:2475
Han K, He G (2007) J Photochem Photobiol C: Photochem Rev 8:55
Pietrzak M, Shibl M, Broring M, Kuhn O, Limbach H (2007) J Am Chem Soc 129:296
Olsen S, Smith SC (2008) J Am Chem Soc 130:8677
Raymo FM, Bartberger MD, Houk KN, Stoddart JF (2001) J Am Chem Soc 123:9264
Cramer CJ, Truhlar DG (2008) Acc Chem Res 41:760
Zhao G, Han K (2007) J Phys Chem A 111:2469
Zhao G, Liu J, Zhou L, Han K (2007) J Phys Chem B 111:8940
Zhao G, Northrop B, Stang P, Han K (2010) J Phys Chem 114:3418
Zhao G, Han K (2010) Phys Chem Chem Phys 12:8914
Zhao G, Han K (2012) Acc Chem Res 45:404
Kungwan N, Kerdpol K, Daengngern R, Hannongbua S (2014) Barbatti M 133:1480
Savarese M, Brémond É, Adamo C, Rega N, Ciofini I (2016) Chem Phys Chem 17:1530
Wilbraham L, Savarese M, Rega N, Adamo C, Ciofini I (2015) J Phys Chem B 119:2459
Rini M, Magnes BZ, Pines E, Nibbering ETJ (2003) Science 301:349
Siwick BJ, Bakker HJ (2007) J Am Chem Soc 129:13412
Savarese M, Netti PA, Adamo C, Rega N, Ciofini I (2013) J Phys Chem B 117:16165
Wang Y, Liu W, Tang L, Oscar B, Han F, Fang C (2013) J Phys Chem A 117:6024
Raucci U, Savarese M, Adamo C, Ciofini I, Rega N (2015) J Phys Chem B 119:2650
Petrone A, Cimino P, Donati G, Hratchian HP, Frisch MJ, Rega N (2016) J Chem Theory Comput 12:4925
Zhou PW, Han KL (2018) Acc Chem Res 51:1681
Chiariello MG, Rega N (2018) J Phys Chem A 122:2884
Donati G, Petrone A, Caruso P, Rega Nadia (2018) Chem Sci 9: 1126
Amoruso G, Taylor VCA, Duchi M, Goodband E, Oliver TAA (2019) J Phys Chem B 123:4745
Negreie M, Bellefeuille SM, Whitham S, Petrich JW, Thornburg RW (1990) J Am Chem Soc 112:7419
Smirnov AS, English DS, Rich RL, Lane J, Teyton L, Schwabacher AW, Luo S, Thornburg RW, Petrich JW (1997) J Phys Chem B 101:2758
Negrerie M, Gai F, Bellefeuille SM, Petrich JW (1991) J Phys Chem 95:8663
Douhal A, Kim SK, Zewail AH (1995) Nature 378:260
Chachisvilis M, Fiebig T, Douhal A, Zewail AH (1998) J Phys Chem A 102:669
Fiebig T, Chachisvilis M, Manger M, Zewail AH, Douhal A, Garcia-Ochoa I, de La Hoz AA (1999) J Phys Chem A 103:7419
Moreno M, Douhal A, Lluch JM (2001) J Phys Chem A 105:3887
Guallar V, Batista VS, Miller WH (1999) J Chem Phys 110:9922
Kwon OH, Zewail AH (2007) Proc Natl Acad Sci USA 104:8703
Takeuchi S, Tahara T (1998) J Phys Chem A 102:7740
Catalán J, Prez P, del Valle JC, de Paz JLG, Kasha M (2002) Proc Natl Acad Sci USA 99:5799
Catalán J, Prez P, del Valle JC, de Paz JLG, Kasha M (2004) Proc Natl Acad Sci USA 101:419
Sakota K, Hara A, Sekiya H (2004) Phys Chem Chem Phys 6:32
Sakota K, Sekiya H (2005) J Phys Chem A 109:2718
Sakota K, Sekiya H (2005) J Phys Chem A 109:2722
Sakota K, Okabe C, Nishi N, Sekiya H (2005) J Phys Chem A 109:5245
Catalän J, de Paz JLG (2005) J Chem Phys 123:114302
Takeuchi S, Tahara T (2007) Proc Natl Acad Sci USA 104:5285
Folmer DE, Wisniewski ES, Stairs JR, Castleman AW Jr (2000) J Phys Chem A 104:10545
Schowen RL (1997) Angew Chem Int Ed 36:1434
Kwon OH, Lee YS, Park HJ, Kim Y, Jang DJ (2004) Angew Chem Int Ed 43:5792
Avouris P, Yang LL, El-Bayoumi MA (1976) Photochem Photobiol 24:211
Collins ST (1983) J Phys Chem 87:3202
Chou PT, Martinez ML, Cooper WC, McMorrow D, Collin ST, Kasha M (1992) J Phys Chem 96:5203
Chapman CF, Maroncelli M (1992) J Phys Chem 96:8430
Chen Y, Rich RL, Gai F, Petrich JW (1993) J Phys Chem 97:1770
Ross JB, Szabo AG, Hogue CW (1997) Methods Enzymol 278:151
Rich RL, Smirnov AV, Schwabacher AW, Petrich JW (1995) J Am Chem Soc 117:11850
Hoesl MG, Larregola M, Cui H, Budisa N (2010) J Pept Sci 16:589
Chen Y, Gai F, Petrich JW (1994) J Phys Chem 98:2203
Shen JY, Chao WC, Liu C, Pan HA, Yang HC, Chen CL, Lan YK, Lin LJ, Wang JS, Lu JF, Chou SCW, Tang KC, Chou PT (2013) Nat Commun 4:2611
Chou PT, Chi Y (2007) Chem Eur J 13:380
Liu Y, Tang Z, Wang Y, Tian J, Fei X, Cao F, Li GU (2017) Spec Acta A Mol Biomol Spec 187:163
Fang H (2019) Spec Acta A Mol Biomol Spec 214:152
Chen KY, Hsieh CC, Cheng YM, Lai CH, Chou PT (2006) Chem Commun 13:4395
Hsieh CC, Cheng YM, Hsu CJ, Chen KY, Chou PT (2008) J Phys Chem A 112:8323
Hristova S, Dobrikov G, Kamounah FS, Kawauchi S, Hansen PE, Deneva V, Nedeltcheva D, Antonov L (2015) RSC Adv 5:102495
Li CZ, Yang YG, Ma C, Liu YF (2016) RSC Adv 6:5134
Marciniak H, Hristova S, Deneva V, Kamounah FS, Hansen PE, Lochbrunner S, Antonov L (2017) Phys Chem Chem Phys 19:26621
Yi JC, Fang H (2018) Struct Chem 29:1341
Frisch MJ, Truck GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09, Rev. D01, Gaussian, Inc, Wallingford CT.
Zhao Y, Truhlar DG (2008) Theor Chem Acc 120:215
Cancès E, Mennucci B, Tomasi J (1997) J Chem Phys 107:3032
Cossi M, Barone V, Mennucci B (1998) Chem Phys Lett 286:253
Mennucci B, Tomasi J (1997) J Chem Phys 106:5151
Tanner C, Manca C, Leutwyler S (2003) Science 302:1736
Fang WH (1999) J Am Chem Soc 103:5567
Tanner C, Manca C, Leutwyler S (2005) J Chem Phys 122:204326
Ashfold MNR, Cronin B, Devine AL, Dixon RN, Nix MGD (2006) Science 312:1637
Mohammed OF, Pines D, Nibbering ETJ, Pines E (2007) Angew Chem Int Ed 46:1458
Hansch C, Leo A, Taft RW (1991) Chem Rev 91:165
Limbach HH, Pietrzak M, Benedict H, Tolstoy PM, Golubev NS, Denisov GS (2004) J Mol Struct 706:115
Limbach HH, Lopez JM, Kohen A (2006) Philos Trans R Soc B 361:1399
Limbach HH (2007) In hydrogen-transfer reactions. Schowen RL, Klinman JP, Hynes JT, Limbach HH (eds). Wiley, Weinheim, Chapter 6, pp 135–221.
Brown ID (1992) Acta Cryst B 48:553
Dos A, Schimming V, Tosoni S, Limbach HH (2008) J Phys Chem B 112:15604
Garrett BC, Truhlar DG (1979) J Am Chem Soc 101:4534
Johnston HS (1966) Gas phase reaction rate theory. Ronald Press, New York, pp 1–362
Limbach HH, Schowen KB, Schowen RL (2010) J Phys Org Chem 23:586
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fang, H. A theoretical study on water-assisted excited state double proton transfer process in substituted 2,7-diazaindole-H2O complex. Theor Chem Acc 139, 139 (2020). https://doi.org/10.1007/s00214-020-02655-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-020-02655-3