Abstract
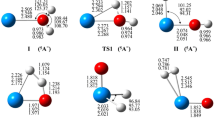
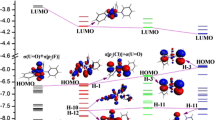
The reaction mechanism of uranium atom with acetonitrile molecule has been systematically studied on the quintet and triplet spin-state potential energy surfaces (PESs) at B3LYP level of density functional theory. Reaction site prediction and bonding evolution were analyzed using different methods. Crossing seams and possible spin inversion processes between different PESs are discussed by means of spin–orbit coupling (SOC) calculations. The results show that there are three crossing points in the reaction, which appear in the process of capturing hydrogen atom. Larger SOC constant (1545.80 cm−1) and intersystem crossing (ISC) probability (<P ISC> = 0.72) between quintet and triplet indicate that the effective ISC would occur in the vicinity of the minimum energy crossing points.






Similar content being viewed by others
References
Lyon J-T, Andrews L (2007) Inorg Chem 46:4799–4808
Cho H-G, Andrews L, Vlaisavljevich B et al (2009) Organometallics 28:5623–5632
Si Y, Zhang W, Zhao Y (2012) J Phys Chem A 116:2583–2590
Niu W, Zhang H, Li P, Gao T (2015) Int J Quantum Chem 115:6–18
Wang X-L, Wang Y-C, Li S et al (2016) J Phys Chem A 120:5457–5463
Shaik S, Danovich D, Fiedler A, Schwarz H (1995) Helv Chim Acta 78:1393–1407
Shaik S, De-Visser S-P, Ogliaro F, Schwarz H, Schröder D (2002) Curr Opin Chem Biol 6:556–567
Schröder D, Shaik S, Schwarz H (2000) Acc Chem Res 33:139–145
Shaik S (2013) Int J Mass Spectrom 354:5–14
Cho H-G, Andrews L (2015) Eur J Inorg Chem 2015:4379–4387
Cho H-G, Andrews L (2012) Organometallics 31:6095–6105
Li Q, Qiu Y-X, Chen X-Y, Schwarz W-E, Wang S-G (2012) Phys Chem Chem Phys 14:6833–6841
Li Q, Chen X-Y, Qiu Y-X, Wang S-G (2012) J Phys Chem A 116:5019–5025
Xiao Y, Ji W-X, Chen X-Y et al (2014) Dalton Trans 43:9508–9517
Jin Y, Wang Y, Ji D (2013) Comput Theor Chem 1011:75–81
Ji D, Wang Y, Jin Y et al (2012) Int J Quantum Chem 112:3685–3690
Cho H-G, Andrews L (2011) Organometallics 31:535–544
Su M-D, Chu S-Y (1999) J Am Chem Soc 121:4229–4237
Harvey J-N (2006) Annu Rep Prog Chem Sect C: Phys Chem 102:203–226
Lee C, Yang W, Parr R-G (1988) Phys Rev B 37:785
Raghavachari K, Trucks G-W (1989) J Chem Phys 91:1062–1065
Becke A-D (1988) Phys Rev A 38:3098
Küchle W, Dolg M, Stoll H et al (1994) J Chem phys 100:7535–7542
Andrae D, Haeussermann U, Dolg M et al (1990) Theoret Chim Acta 77:123–141
Scuseria G-E, Schaefer H-F III (1989) J Chem Phys 90:3700–3703
Purvis G-D III, Bartlett R-J (1982) J Chem Phys 76:1910–1918
Dunning Jr T-H (1989) J Chem Phys 90:1007–1023
Yoshizawa K, Shiota Y, Yamabe T (1999) J Chem Phys 111:538–545
Harvey J-N, Poli R, Smith K-M (2003) Coord Chem Rev 238:347–361
Harvey J-N, Aschi M, Schwarz H, Koch W (1998) Theor Chem Acc 99:95–99
Granovsky A-A, www http://classic.chem.msu.su/gran/gamess/index.html. Schmidt. MW
Gaenko A, DeFusco A, Varganov S-A, Martínez T-J, Gordon M-S (2014) J Phys Chem A 118:10902–10908
Danovich D, Marian C-M, Neuheuser T, Peyerimhoff S-D, Shaik S (1998) J Phys Chem A 102:5923–5936
Isobe H, Yamanaka S, Kuramitsu S, Yamaguchi K (2008) J Am Chem Soc 130:132–149
Volkov MV, Ostrovsky VN (2004) J Phys B-At Mol Opt 37:4069
Goodrow A, Bell AT, Head-Gordon M (2009) J Phys Chem C 113:19361–19364
Kaliakin DS, Zaari RR, Varganov SA (2015) J Phys Chem A 119:1066–1073
Lu T, Chen F (2012) J Mol Graph Model 38:314–323
Becke A-D, Edgecombe K-E (1990) J Chem Phys 92:5397–5403
Alikhani M-E, Michelini M-C, Russo N, Silvi B (2008) J Phys Chem A 112:12966–12974
Bader R (1990) A Quantum Theory. Oxford, England, Clarendon
Lu T, Chen F (2012) J Comput Chem 33:580–592
Cremer D, Kraka E (1984) Angew Chem Int Ed 23:627–628
Di Santo E, Michelini M-C, Russo N (2009) Organometallics 28:3716–3726
De Almeida K, Ramalho T, Neto J, Santiago R, Felicíssimo V, Duarte H (2013) Organometallics 32:989–999
Du J, Sun X, Chen J, Zhang L, Jiang G (2014) Dalton Trans 43:5574–5579
Danovich D, Shaik S (1997) J Am Chem Soc 119:1773–1786
Kennedy C-J, Siviloglou G-A, Miyake H, Burton W-C, Ketterle W (2013) Phys Rev Lett 111:225301
Cimas A, Rayón V-M, Aschi M, Barrientos C, Sordo J-A, Largo A (2005) J Chem Phys 123:114312
Cimas A, Rayón V, Barrientos C, Aschi M, Sordo J, Largo A (2006) Chem Phys 328:45–52
Acknowledgements
We acknowledge financial support from the National Natural Science Foundation of China (Grant No. 21263023) and support from the Supercomputing Center of Gansu Province.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, X., Wang, Y., Li, S. et al. Activation of acetonitrile by gas-phase uranium: bond structure analysis and spin–flip reaction mechanism. Theor Chem Acc 136, 107 (2017). https://doi.org/10.1007/s00214-017-2137-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-017-2137-4