Abstract
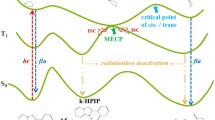
N-Hydroxypyridine-2(1H)-thione (N-HPT) is an important photochemical generator of hydroxyl radicals; however, it has been pointed out that N-HPT is not a specific precursor of hydroxyl radical. Photoionization of N-HPT competes with photochemical N–O bond cleavage in neutral aqueous solution. The possibility of a competitive reaction could be critical for studies using N-HPT as the radical precursor; therefore, the detailed behaviors of electronic excitation and ionization of N-HPT and its deprotonated anion, which is the dominant tautomer under neutral pH conditions, are studied using quantum chemical methods with the symmetry-adapted cluster-configuration interaction (SAC-CI) method and the polarizable continuum model (PCM). The detailed assignment of the UV–Vis spectra of N-HPT is provided, and the origin of the observed negative solvatochromism is found to be the charge transfer excitation between the sulfur and the pyridine ring. The photochemical N–O bond cleavage occurs via the conical intersections between the lowest π → π* and π → σ* states and between the π → σ* and ground state, when N-HPT dissociates into PyS· and ·OH radicals. The calculated ionization potentials of N-HPT and the deprotonated N-HPT anion are 5.75 and 4.67 eV in PCM water. This demonstrates that the charge transfer excitation energy between N-HPT and liquid water becomes significantly lower for the deprotonated anion in comparison with the neutral molecule. Even under mild photochemical conditions, photoinduced ionization of N-HPT may occur in neutral aqueous solution.






Similar content being viewed by others
References
Neihof RA, Bailey CA, Patouillet C, Hannan PJ (1979) Photodegradation of mercaptopyridine-N-oxide biocides. Arch Environ Contam Toxicol 8:355–368
Blatt J, Taylor SR, Kontoghiorghes GJ (1989) Comparison of activity of deferoxamine with that of oral iron chelators against human neuroblastoma cell lines. Cancer Res 49:2925–2927
Reeder NL, Xu J, Youngquist RS, Schwartz JR, Rust RC, Saunders CW (2011) The antifungal mechanism of action of zinc pyrithione. Br J Dermatol 165:9–12
Turner GA, Matheson JR, Li G-Z, Fei X-Q, Zhu D, Baines FL (2013) Enhanced efficacy and sensory properties of an anti-dandruff shampoo containing zinc pyrithione and climbazole. Int J Cosmet Sci 35:78–83
Epe B, Ballmaier D, Adam W, Grimm GN, Saha-Möller CR (1996) Photolysis of N-hydroxypyridinethiones: a new source of hydroxyl radicals for the direct damage of cell-free and cellular DNA. Nucleic Acids Res 24:1625–1631
Chaulk SG, Pezacki JP, MacMillan AM (2000) Studies of RNA cleavage by photolysis of N-hydroxypyridine-2(1H)-thione. A new photochemical footprinting method. Biochemistry 39:10448–10453
Mitroka S, Zimmeck S, Troya D, Tanko JM (2010) How solvent modulates hydroxyl radical reactivity in hydrogen atom abstractions. J Am Chem Soc 132:2907–2913
Chen C-H, Han R-M, Liang R, Fu L-M, Wang P, Ai X-C, Zhang J-P, Skibsted LH (2011) Direct observation of the β-carotene reaction with hydroxyl radical. J Phys Chem B 115:2082–2089
Reszka KJ, Chignell CF (1995) Photochemistry of 2-mercaptopyridines, part 3. EPR study of photoproduction of hydroxyl radicals by N-hydroxypyridine-2-thione using 5,5-dimethy-1-pyrroline N-oxide in aqueous solutions. Photochem Photobiol 61:269–275
Aveline BM, Kochevar IE, Redmond RW (1996) N-Hydroxypyridine-2(1H)-thione: not a selective generator of hydroxyl radicals in aqueous solution. J Am Chem Soc 118:289–290
Aveline BM, Kochevar IE, Redmond RW (1996) Photochemistry of the nonspecific hydroxyl radical generator, N-hydroxypyidine-2(1H)-thione. J Am Chem Soc 118:10113–10123
Lapinski L, Gerega A, Sobolewski AL, Nowak MJ (2008) Thioperoxy derivative generated by UV-induced transformation of N-hydroxypyridine-2(1H)-thione isolated in low-temperature matrixes. J Phys Chem A 112:238–248
Arnone M, Engels B (2007) Rational design of substituted N-alkoxypyridine-2(1H)thiones with increased stability against daylight. J Phys Chem A 111:3161–3165
Jankowiak A, Kaszynski P (2009) 4-Substituted 1-acyloxypyridine-2(1H)-thiones: experimental and computational studies of the substituent effect on electronic absorption spectra. J Org Chem 74:7441–7448
Nakatsuji H (1978) Cluster expansion of the wavefunction excited states. Chem Phys Lett 59:362–364
Nakatsuji H (1979) Electron correlations in ground and excited states by SAC (symmetry-adapted cluster) and SAC-CI theories. Chem Phys Lett 67:329–333
Nakatsuji H (1992) Electronic structures of ground, excited, ionized, and anion states studied by the SAC/SAC-CI theory. Acta Chim Acad Sci Hung 129:719–776
Fukuda R, Nakatsuji H (2008) Formulation and implementation of direct algorithm for the symmetry adapted cluster and symmetry adapted cluster-configuration interaction method. J Chem Phys 128:094105
Miertuš S, Scrocco E, Tomasi J (1981) Electrostatic interaction of a solute with a continuum. A direct utilization of ab initio molecular potentials for the prevision of solvent effects. Chem Phys 55:117–129
Tomasi J, Mennucci B, Cammi R (2005) Quantum mechanical continuum solvation models. Chem Rev 105:2999–3093
Cammi R, Fukuda R, Ehara M, Nakatsuji H (2010) Symmetry-adapted cluster and symmetry-adapted cluster-configuration interaction method in the polarizable continuum model: theory of the solvent effect on the electronic excitation of molecules in solution. J Chem Phys 133:024104
Fukuda R, Ehara M, Nakatsuji H, Cammi R (2011) Nonequilibrium solvation for vertical photoemission and photoabsorption processes using the symmetry-adapted cluster-configuration interaction method in the polarizable continuum model. J Chem Phys 134:104109
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2010) Gaussian 09, revision B. 01. Gaussian, Wallingford
Cancès E, Mennucci B, Tomasi J (1997) A new integral equation formalism for the polarizable continuum model: theoretical background and applications to isotropic and anisotropic dielectrics. J Chem Phys 107:3032–3041
Mennucci B, Cancès E, Tomasi J (1997) Evaluation of solvent effects in isotropic and anisotropic dielectrics and in ionic solutions with a unified integral equation method: theoretical bases, computational implementation, and numerical applications. J Phys Chem B 101:10506–10517
Cancès E, Mennucci B (1998) New applications of integral equation methods for solvation continuum models: ionic solutions and liquid crystals. J Math Chem 23:309–326
Dunning TH Jr (1989) Gaussian basis sets for use in correlated molecular calculations. I. The atoms boron through neon and hydrogen. J Chem Phys 90:1007–1023
Kendall RA, Dunning TH Jr, Harrison RJ (1992) Electron affinities of the first-row atoms revisited. Systematic basis sets and wave functions. J Chem Phys 96:6796–6806
Woon DE, Dunning TH Jr (1993) Gaussian basis sets for use in correlated molecular calculations. III. The second row atoms, Al–Ar. J Chem Phys 98:1358–1371
Fukuda R, Ehara M, Cammi R (2014) Electronic excitation spectra of molecules in solution calculated using the symmetry-adapted cluster-configuration interaction method in the polarizable continuum model with perturbative approach. J Chem Phys 140:064114
Fukuda R, Ehara M (2014) Effects of perturbation-selection and orbital dependence for the SAC-CI calculations in valence excitations of medium-size molecules. J Comput Chem 35:2163–2176
Nakatsuji H, Hirao K (1981) Cluster expansion of the wave function. Electron correlation in singlet and triplet excited states, ionized states, and electron attached states by SAC and SAC-CI theories. Int J Quantum Chem 20:1301–1313
Jones RA, Katritzky AR (1960) N-oxides and related compounds. Part XVII. The tautomerism of mercapto- and acylamino-pyridine 1-oxides. J Chem Soc 2937–2942. doi:10.1039/JR9600002937
Daly AM, Mitchell EG, Sanchez DA, Block E, Kukolich SG (2011) Microwave spectra and gas phase structural parameters for N-hydroxypyridine-2(1H)-thione. J Phys Chem A 115:14526–14530
Sobolewski AL, Domcke W, Dedonder-Lardeux C, Jouvet C (2002) Excited-state hydrogen detachment and hydrogen transfer driven by repulsive 1πσ* states: a new paradigm for nonradiative decay in aromatic biomolecules. Phys Chem Chem Phys 4:1093–1100
Ashfold MNR, Cronin B, Devine AL, Dixon RN, Nix MGD (2006) The role of πσ* excited states in the photodissociation of heteroaromatic molecules. Science 312:1637–1640
Ashfold MNR, King GA, Murdock D, Nix MGD, Oliver TAA, Sage AG (2010) πσ* excited states in molecular photochemistry. Phys Chem Chem Phys 12:1218–1238
Thapa B, Schlegel HB (2015) Calculations of pK a’s and redox potentials of nucleobases with explicit waters and polarizable continuum solvation. J Phys Chem A 119:5134–5144
Mulliken RS (1952) Molecular compounds and their spectra. II. J Am Chem Soc 74:811–824
McGlynn SP (1958) Energetics of molecular complexes. Chem Rev 58:1113–1156
Hastings SH, Franklin JL, Schiller JC, Matsen FA (1952) Molecular complexes involving iodine. J Am Chem Soc 75:2900–2905
Grand D, Bernas A, Amouyal E (1979) Photoionization of aqueous indole; conduction band edge and energy gap in liquid water. Chem Phys 44:73–79
do Couto PC, Cabral BJC, Canuto S (2006) Electron binding energies of water cluster: implications for the electronic properties of liquid water. Chem Phys Lett 429:129–135
Acknowledgments
This work was supported by a Grant-in-Aid for Scientific Research from the Japan Society for the Promotion of Science (JSPS), Japan, and MEXT (Ministry of Education Culture, Sports, Science and Technology, Japan) program “Elements Strategy Initiative to Form Core Research Center.” Some of the computations were performed at the Research Center for Computational Science, Okazaki, Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published as part of the special collection of articles “Charge Transfer Modeling in Chemistry”.
Rights and permissions
About this article
Cite this article
Fukuda, R., Ehara, M. Electronic excitation and ionization behavior of N-hydroxypyridine-2(1H)-thione and its deprotonated anion in a polarizable medium studied using quantum chemical computations. Theor Chem Acc 135, 105 (2016). https://doi.org/10.1007/s00214-016-1870-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-016-1870-4