Abstract
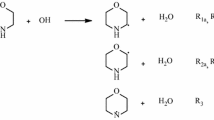
The reaction of aniline with hydrogen atom is investigated herein using the hybrid meta-DFT functional of BB1 K. Hydrogen atom is found to preferentially add at an ortho position. However, the fate of the o-(C6H5NH2)H adduct is found to be solely the deactivation of the initial addition channel. The rate constant for the abstraction channel (C6H5NH2 + H → C6H5NH + H2) is fitted by the expression 1.10 × 10−11 exp(−4,200/T) cm3 molecule−1 s−1. Our calculated rate constant for the abstraction channel agrees very well with the available experimental measurements. Satisfactory agreement is found between calculated and experimental measurements for the displacement channel (C6H5NH2 + H → C6H6 + NH2). Our detailed analysis for the corresponding displacements in toluene and phenol suggests that the three systems exhibit similar behavior with regard to the relative importance of abstraction and displacement channels.










Similar content being viewed by others
References
Aniline Fact Sheet (CAS No. 62-53-3) (1994) Pollution prevention and toxics. Washington, DC
Jones CR, Liu Y-Y, Sepai O, Yan H, Sabbioni G (2005) Environ SciTechnol 40:387
Calaf RE, Pena J, Paytubi S, Blount BC, Posada de la Paz M, Gelpi E, Abian J (2001) Anal Chem 73:3828
Ferge T, Maguhn J, Hafner K, Muhlberger F, Davidovic M, Warnecke R, Zimmermann R (2005) Environ Sci Technol 39:1393
Atkinson R, Tuazon EC, Wallington TJ, Aschmann SM, Arey J, Winer AM, Pitts JN (1987) Environ Sci Technol 21:64
Patil SS, Shinde VM (1988) Environ Sci Technol 22:1160
Yang K, Wu W, Jing Q, Zhu L (2008) Environ Sci Technol 42:7931
He YZ, Cui JP, Mallard WG, Tsang WJ (1988) J Phys Chem 92:1510
Frisch MJT, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Zakrzewski VG, Montgomery JA, Stratmann RE, Burant JC, Dapprich S, Millam JM, Daniels RE, Kudin KN, Strain MC, Farkas O, Tomasi J, Barone VCM, Cammi R, Mennucci B, Pomelli C, Adamo C, Clifford S, Ochterski J, Petersson GA, Ayala PY, Cui Q, Morokuma K, Salvador P, Dannenberg JJ, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Cioslowski JO, Baboul AG, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Gomperts R, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Andres JL, Gonzalez CM, Head-Gordon M, Replogle ES, Pople JA (2001) Gaussian 03; revision A. 11th edn. Gaussian, Inc, Pittsburgh
Zhao Y, Lynch BJ, Truhlar DGJ (2004) Phys Chem A 108:2715
Montgomery JA, Ochterski JW, Petersson GA (1994) J Chem Phys 101:5900
Kohn W, Sham LJ (1965) Phys Rev A 140:1133
Zhao Y, Gonzalez-Garcia N, Truhlar DG (2005) J Phys Chem A 109:2012
McClurg RB, Flagan RC, Goddard WA (1997) J Chem Phys 106:6675
Mokrushin V, Bedanov V, Tsang W, Zachariah M, Knyazev V (2002) ChemRate; version 1, 19th edn. NIST, Gaithersburg
Eyring HJ (1935) Chem Phys 3:107
Eckart C (1930) Phys Rev 35:1303
Duncan WT, Bell RL, Truong TN (1998) J Comput Chem 19:1309
Fukuyo M, Hirotsu K, Higuchi T (1982) Acta Cryst B 38:640
Alcolea Palafox M, Meléndez F (1999) J Mol Struct: THEOCHEM 493:171
Larsen NW, Hansen EL, Nicolaisen FM (1976) Chem Phys Lett 43:584
Halpern RM, Ramachandran BR, Glendening ED (2007) J Chem Edu 84:1067
Hussein AP, Lielmezs J, Aleman H (1985) Thermochimica Acta 86:209
Evans JC (1960) Spectrochim Acta 428
Michael JV, Sutherland JW, Klemm RJ (1986) Phys Chem 90:497
Good WD, Smith NK (1969) J Chem Eng Data 14:102
da Silva G, Chen C-C, Bozzelli JW (2006) Chem Phys Lett 424:42
He YZ, Mallard WG, Tsang W (1988) J Phys Chem 92:2196
Demissy M, Lesclaux R (1980) J Am Chem Soc 102:2897
Zhu L, Bozzelli JW (2003) J Phys Chem A 107:3696
Altarawneh M, Dlugogorski BZ, Kennedy EM, Mackie JC (2010) Combust Flame 157:1325
Robaugh D, Tsang WJ (1988) J Phys Chem 80:4159
Ellis C, Scott MS, Walker RW (2003) Combust Flame 132:291
Zhang QZ, Qu XH, Xu F, Shi XY, Wang WX (2009) Environ Sci Technol 43:4105
Li S, Zhang Q, Wang W (2006) Chem Phys Lett 428:262
Xu F, Wang H, Zhang QZ, Zhang RX, Qu XH, Wang WX (2010) Environ Sci Technol 44:1399
Acknowledgments
This study has been supported by a grant of computing time from the Australian Centre of Advanced Computing and Communications (ac3).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
214_2011_940_MOESM1_ESM.doc
Supporting Information Available: Calculated total energies, zero-point energies, Cartesian coordinates, moments of inertia, and vibrational frequencies of all structures. (DOC 67 kb)
Rights and permissions
About this article
Cite this article
Batiha, M., Altarawneh, M., Alsofi, A. et al. Theoretical study on the reaction of hydrogen atoms with aniline. Theor Chem Acc 129, 823–832 (2011). https://doi.org/10.1007/s00214-011-0940-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00214-011-0940-x