Abstract
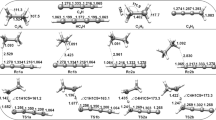
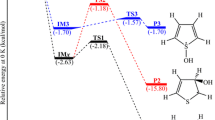
We present density functional theory (DFT) and complete basis set (CBS) calculations of the prototypical radical–radical reaction of ground–state atomic oxygen [O(3P)] with ethyl (C2H5) radicals. The respective reaction mechanisms and dynamics were investigated on the doublet potential energy surfaces using the DFT method and CBS model. In the title reaction, the barrierless addition of O(3P) to C2H5 led to the formation of energy-rich intermediates that underwent subsequent isomerization and decomposition to yield various products. The products predicted to be found were: H2CO + CH3, CH3CHO + H, c–CH2OCH2 + H, 1,3CH3COH + H, 1,3HCOH + CH3, CH2CHOH + H, C2H3 + H2O, and CH2CH2 + OH. In particular, unlike previous kinetic results, proposed to proceed only through the direct H-atom abstraction process, two distinctive pathways to the formation of CH2CH2 + OH were predicted to be in competition: direct, barrierless H-atom abstraction mechanism versus addition process. The competition was consistent with the recent crossed-beam investigations, and their microscopic dynamic characteristics are discussed at the molecular level.









Similar content being viewed by others
References
Choi JH (2006) Int Rev Phys Chem 25:613 (references therein)
Donalson DJ, Okuda IV, Sloan J (1995) J Chem Phys 193:37
Min Z, Quandt RW, Wong TH, Bersohn R (1999) J Chem Phys 111:7369
Lindner J, Loomis RA, Klaassen JJ, Leone SR (1998) J Chem Phys 108:1944
Reid JP, Marcy TP, Kuehn S, Leone SP (2000) J Chem Phys 113:4572
Slagle IR, Sarzynski D, Gutman D, Miller JA, Melius CF (1988) J Chem Soc Faraday Trans II 84:491
Hoyermann K, Olzmann M, Seeba J, Viskolcz B (1999) J Phys Chem A 103:5692
Hack W, Hoyermann K, Olzmann M, Zeuch T (2002) Proc Combust Inst 29:1247
Kwon HC, Park JH, Lee H, Kim HK, Choi YS, Choi JH (2002) J Chem Phys 116:2675
Park JH, Lee H, Kwon HC, Kim HK, Choi YS, Choi JH (2002) J Chem Phys 117:2017
Lee H, Joo SK, Kwon LK, Choi JH (2003) J Chem Phys 119:9337
Lee H, Joo SK, Kwon LK, Choi JH (2004) J Chem Phys 120:2215
Joo SK, Kwon LK, Lee H, Choi JH (2004) J Chem Phys 120:7976
Nam MJ, Youn SE, Li L, Choi JH (2005) J Chem Phys 123:211105
Nam MJ, Youn SE, Choi JH (2006) J Chem Phys 124:104307
Kwon LK, Nam MJ, Youn SE, Joo SK, Lee H, Choi JH (2006) J Chem Phys 124:204320
Youn SE, Ok YH, Choi JH (2008) Chem phys chem 9:1099
Park YP, Kang KW, Jung SH, Choi JH (2010) J Phys Chem A 114:4891
Park YP, Kang KW, Jung SH, Choi JH (2010) Phys Chem Chem Phys 12:7098
Kohn DW, Clauberg H, Chen P (1992) Rev Sci Instrum 63:4003
Sweeney GM, McKendrick KG (1997) J Chem Phys 106:9182
Sweeney GM, Watson A, McKendrick KG (1997) J Chem Phys 106:9172
Kleimermanns K, Luntz AC (1982) J Chem Phys 77:3533
Andresen P, Luntz AC (1980) J Chem Phys 72:5842
Knyazev VD (2001) J Phys Chem A 106:8741
Marcy TP, Diaz RR, Heard D, Leone SR, Harding LB, Klippenstein SJ (2001) J Phys Chem A 105:8361
Yagi K, Takayanagi T, Taketsugu T, Hirao K (2004) J Chem Phys 120:10395
Harding LB, Klippenstein SJ, Georgievskii Y (2005) Proc Combust Inst 30:985
Gupta A, Singh RP, Singh VB, Mishra BK, Sathyamuryhy N (2007) J Chem Sci 119:457
Yong Y, Weijun Z, Xiaoming G, Shixin P, Jie S, Wei H, Jun Q (2005) Chin J Chem Phys 18:515
Nam MJ, Youn SE, Choi JH (2006) ChemPhysChem 7:2526
Park JH, Lee H, Choi JH (2003) J Chem Phys 119:8966
Lee H, Nam MJ, Choi JH (2006) J Chem Phys 124:044311
Baulch DL, Bowman CT, Cobos CJ, Cox RA, Just Th, Kerr JA, Pilling MJ, Stocker D, Troe J, Tsang W, Walker RW, Warnatz J (2005) J Phys Chem Ref Data 34:757
Petersson GA, Al-Laham MA (1991) J Chem Phys 94:6081
Petersson GA, Tensfeldt TG, Montgomery JA Jr (1991) J Chem Phys 94:6091
Montgomery JA Jr, Ochtersk JW, Petersson GA (1994) J Chem Phys 101:5900
Baker J, Muir M, Andzelm J (1995) J Chem Phys 102:2063
Ochterski JW, Petersson GA, Montgomery JA Jr (1996) J Chem Phys 104:2598
Montgomery JA Jr, Frisch MJ, Ochterski JW, Petersson GA (1999) J Chem Phys 110:2822
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Zakrzewski VG, Montgomery JA Jr, Stratmann RE, Burant JC, Dapprich S, Millam JM, Daniels AD, Kudin KN, Strain MC, Farkas O, Tomasi J, Barone V, Cossi M, Cammi R, Mennucci B, Pomelli C, Adamo C, Clifford S, Ochterski J, Petersson GA, Ayala PY, Cui Q, Morokuma K, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Cioslowski J, Ortiz JV, Baboul AG, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Gomperts R, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Gonzalez C, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Andres JL, Gonzalez C, Head-Gordon M, Replogle ES, Pople JA (2003) Gaussian 03, revision B 01. Gaussian, Inc., Pittsburgh
Zhu RS, Park J, Lin MC (2005) Chem Phys Lett 408:25
Senosiain JP, Klippenstein SJ, Miller JA (2006) J Phys Chem A 110:6960
Xu ZF, Xu K, Lin MC (2009) ChemPhysChem 10:972
Liu GX, Ding YH, Li ZS, Fu Q, Huang XR, Sun CC, Tang AC (2002) Phys Chem Chem Phys 4:1021
Hammond GS (1955) J Am Chem Soc 77:334
Steinfeld JI, Francisco JS, Hase WL (1999) Chemical kinetics and dynamics. Prentice Hall, New Jersey
Baer T, Hase WL (1996) Unimolecular reaction dynamics. Oxford University, New York
Acknowledgments
This work was supported by National Research Foundation of Korea Grant funded by the Korean Government (2010-0014418) and Priority Research Centers Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (NRF20100020209).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jung, SH., Park, YP., Kang, KW. et al. A computational study of the radical–radical reaction of O(3P) + C2H5 with comparisons to gas-phase kinetics and crossed-beam experiments. Theor Chem Acc 129, 105–118 (2011). https://doi.org/10.1007/s00214-011-0903-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00214-011-0903-2