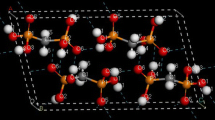
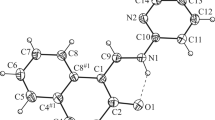
Abstract
The repulsive nature of the interaction between the cation radicals of the π-[terthiophene] 2+2 dimers, 1 2+ 2 , found in crystals has been concluded from B3LYP/6-31+G(d) calculations. Hence, the bonding component is weaker than the Coulombic repulsion, consistent to recent findings for [TTF]·+–[TTF]·+ interactions (TTF = tetrathiafulvalene). The existence of 1 2+ 2 dimers originates from the cation+–anion− electrostatic interactions, which exceeds the combined effect of the 1 .+–1 .+ plus (SbF6)−–(SbF6)− repulsions in 1 2(SbF6)2, similar to what is found for [TTF]·+–[TTF]·+ interactions in [TTF]2(ClO4)2 aggregates and in crystals. The long, multicenter bond in 1 2+ 2 is characterized as a 2e−/10c bond from an Atoms-in-molecules analysis.






Similar content being viewed by others
Notes
The MCQDPT/CASSCF(2,2) method performs multiconfigurational perturbation calculation on a multiconfigurational CASSCF(2,2) wavefunction using the MCQDPT method [24] provides an accurate evaluation of the dispersion component of the interaction energy and gives results similar to those obtained using the more popular CASPT2 method [25]. The (2,2) active space in these CASSCF(2,2), and MCQDPT/CASSCF(2,2) calculations was that resulting from combining the two SOMO orbitals of the fragments.
The closed-shell singlet state of the [TTF]2(ClO4)2 aggregate was also found to be −19.7 kcal/mol more stable than its fragmentation into two [TTF]+ and two (ClO4)− ions at the crystal geometry (the open-shell singlet and triplet states are also more stable than the fragments by −19.6 and −6.8 kcal/mol, respectively).
References
Hotta S, Waragai K (1993) Adv Mater 5:896. doi:10.1002/adma.19930051204
Nalwa HS (ed) (1997) Handbook of organic conductive molecules and polymers. Wiley, New York
Chang AC, Miller LL (1987) Synth Met 22:71
Zinger B, Mann KR, Hill MG, Miller LL (1992) Chem Mater 4:1113. doi:10.1021/cm00023a033
Hill MG, Mann KR, Miller LL, Penneau JF (1992) J Am Chem Soc 114:2728. doi:10.1021/ja00033a063
Miller LL, Yu Y, Gunic E, Duan R (1995) Adv Mater 7:547. doi:10.1002/adma.19950070606
Graf DD, Miller LL, Mann KR (1996) J Am Chem Soc 118:5480. doi:10.1021/ja960194b
Brocks G (2000) J Chem Phys 112:5353. doi:10.1063/1.481105
Yamazaki D, Nishinaga T, Tanino N, Komatsu K (2006) J Am Chem Soc 128:14470. doi:10.1021/ja065995l
Garcia-Yoldi I, Miller JS, Novoa JJ (2009) J Phys Chem A (in press)
Novoa JJ, Lafuente P, Del Sesto RE, Miller JS (2001) Angew Chem Int Ed 40:2540
Novoa JJ, Lafuente P, Del Sesto RE, Miller JS (2002) Angew Chem Int Ed 4:373
Del Sesto RE, Miller JS, Lafuente P, Novoa JJ (2002) Chem Eur J 8:4894
Miller JS, Novoa JJ (2007) Acc Chem Res 40:189
Novoa JJ, Ribas-Ariño J, Shum WW, Miller JS (2007) Inorg Chem 46:103
Garcia-Yoldi I, Mota F, Novoa JJ (2007) J Comput Chem 28:326
Garcia-Yoldi I, Miller JS, Novoa JJ (2007) J Phys Chem A 111:8020
Garcia-Yoldi I, Miller JS, Novoa JJ (2008) Phys Chem Chem Phys 10:4106
Lu J-M, Rosokha SV, Kochi JK (2003) J Am Chem Soc 125:12161
Jakowski J, Simons J (2003) J Am Chem Soc 125:16089
Jung Y, Head-Gordon M (2004) Phys Chem Chem Phys 6:2008
Becke AD (1993) J Chem Phys 98:5648
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785
Nakano H, Nakayama K, Hirao K, Dupuis M (1997) J Chem Phys 106:4912. doi:10.1063/1.473540
Roos BO, Andersson K, Fulscher MK, Malmqvist PA, Serrano-Andres L, Pierloot K, Merchan M (1996) Adv Chem Phys 93:219. doi:10.1002/9780470141526.ch5
Ditchfield R, Hehre WJ, Pople JA (1971) J Chem Phys 54:724
Frisch MJ et al (2004) Gaussian-03, Revision-C.02. Gaussian Inc., Wallingford
Boys SF, Bernardi F (1970) Mol Phys 19:553. doi:10.1080/00268977000101561
van Duijneveldt FB, de Rijdt JGCM, van Lenthe JH (1994) Chem Rev 94:1873. doi:10.1021/cr00031a007
Novoa JJ, Planas M, Whangbo MH (1994) Chem Phys Lett 225:240. doi:10.1016/0009-2614(94)00646-6
Bader RF (1990) Atoms in molecules. A quantum theory, Clarendon Press, Oxford
Acknowledgments
I.G. Yoldi and J.J. Novoa were supported by the Spanish Science and Education Ministry (BQU2002-04587-C02-02 and UNBA05-33-001, and Ph.D. grant to I.G. Yoldi) and the CIRIT (2001SGR-0044 and 2005-PEIR-0051/69). Computer time was also provided by CESCA and BSC. J.S. Miller was supported in part by the U. S. NSF (Grant No. 0553573), and the DOE (Grant No. DE FG 03-93ER45504). One of us would also express here his gratitude to Prof. S. Olivella for his guidance, support, and personal friendship in the early years of his scientific career.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Dedicated to Professor Santiago Olivella on the occasion of his 65th birthday and published as part of the Olivella Festschrift Issue.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yoldi, I.G., Miller, J.S. & Novoa, J.J. Long, multicenter bonding in π-[terthiophene] 2+2 dimers. Theor Chem Acc 123, 137–143 (2009). https://doi.org/10.1007/s00214-009-0543-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00214-009-0543-y